Graphene‑Based Materials for Bone Regeneration: Opportunities, Challenges, and Clinical Perspectives
Abstract
Recent years have witnessed remarkable advances in the biomedical use of graphene family materials. Their nanoscale dimensions, enormous surface area, intrinsic photoluminescence, and inherent antibacterial properties position them as powerful tools for bone tissue engineering, drug and gene delivery, and bio‑sensing. In this review, we synthesize current progress, critically assess biocompatibility, and discuss the feasibility of graphene derivatives for bone regeneration, highlighting both promising applications and outstanding safety concerns.
Introduction
Patients suffering severe maxillofacial infections, trauma, tumours, or congenital deformities often present with jaw‑bone defects that require prolonged healing. Although bone has a natural capacity for repair [1, 2], the limited self‑regeneration of large or critical‑size defects remains a major clinical challenge [3]. Current therapies—autograft, allograft, and xenograft—each carry drawbacks such as donor‑site morbidity, infection risk, or insufficient osteoinductivity [4–7]. Consequently, bone tissue engineering and regenerative medicine are intensifying efforts to develop substitutes that match the mechanical and biological properties of native bone [8, 9].
Graphene family materials (GFMs)—graphene, graphene oxide (GO), reduced graphene oxide (rGO), carboxylated graphene (CXYG), and graphene quantum dots (GQDs)—have emerged as promising candidates for bone repair. Their exceptional mechanical strength, electrical conductivity, and surface chemistry enable scaffold fabrication, cell‑directed differentiation, and antimicrobial activity [20–23]. However, long‑term biocompatibility and osteogenic potential must be rigorously evaluated before clinical translation.
Challenges in Determining the Bio‑Safety of Graphene Family Materials
In Vitro Cytotoxicity
Prior to clinical trials, GFMs must undergo comprehensive cytotoxicity testing [38]. Bare graphene is hydrophobic and prone to agglomeration, leading to protein adsorption and potential immunogenicity [34, 40, 41]. Chemical functionalization (oxidation, reduction, or introduction of functional groups) enhances hydrophilicity and modulates biological responses. Studies reveal that rGO is less cytotoxic than GO, largely due to lower oxidative stress induced by a reduced density of oxygenated groups [31].
Dose‑dependence is a recurrent theme. For instance, GO concentrations ≥50 µg/mL reduce viability in A549 lung carcinoma cells [43], while 10 µg/mL inhibits proliferation of bone mesenchymal stem cells (BMSCs) but 0.1 µg/mL promotes it [44]. Similar trends appear with MC3T3‑E1 preosteoblasts, where rGO is tolerated below 62.5 µg/mL but becomes detrimental above 100 µg/mL [23]. Carboxylated graphene and GQDs display low toxicity at low doses [34, 46]. Thus, concentration is a key, but not sole, determinant of cytocompatibility.
Shape and size further influence toxicity. Graphene nano‑onions (GNOs) exhibit higher CD_50 values than nanoribbons (GONRs) or nanoplatelets (GONPs), indicating shape‑dependent cytotoxicity [47]. Comparisons with multi‑walled carbon nanotubes (MWNTs) show that flat graphene sheets are less disruptive to cell membranes, whereas tubular MWNTs penetrate membranes more readily, causing acute toxicity [48–50].
Size‑dependent uptake mechanisms have been reported. Smaller GO flakes (≈31 nm) induce apoptosis due to higher cellular internalization, while larger flakes (≈81 nm) mainly adsorb to membranes and are less toxic [51]. Larger sheets (≈860 nm) engage phagocytosis, whereas smaller ones (~420 nm) enter via clathrin‑mediated endocytosis [33]. Across studies, a general trend emerges: nano‑sized GFMs are safer for biomedical use [54], yet precise size thresholds vary among research groups.
In Vivo Biodistribution and Toxicity
Animal studies largely corroborate in vitro findings. Zebrafish embryos exposed to small GO flakes exhibit reduced viability, whereas larger flakes do not [53]. In mice, intravenously injected GO accumulates in liver, lung, and spleen, inducing chronic hepatitis and lung fibrosis; PEGylation mitigates these effects [55]. Pulmonary exposure of GO also causes severe lung injury, but pristine graphene or Pluronic‑dispersed GO are less toxic [56]. Subcutaneous implantation of graphene foams (GFs) or GO foams (GOFs) in rats shows no systemic toxicity after 7 months, with only localized granuloma formation [40]. These results underscore the importance of dosage, functionalization, and delivery route in determining in vivo safety.
Despite these insights, long‑term biodistribution and degradation pathways remain under‑characterized. Continued research must clarify how GFMs interact with complex biological milieus and establish robust safety profiles.
Antibacterial Activity of Graphene Family Materials
Infection control is critical for bone regeneration. GFMs exhibit antibacterial properties through multiple mechanisms: direct membrane disruption by sharp edges, electron‑transfer‑induced oxidative stress, and ROS generation [60–63]. While the precise contributions of edge versus surface chemistry remain debated, the synergistic effects of functional groups and sharp nanosheets consistently reduce bacterial viability. Further investigation into physicochemical determinants will enable rational design of antimicrobial bone grafts.
Graphene Family Materials Mediate Cells into Osteogenic Differentiation and Promote Bone Regeneration In Vivo
Graphene and its derivatives have repeatedly shown the ability to support cell attachment, proliferation, and spontaneous osteogenic differentiation across a range of stem cell sources, including dental pulp, bone marrow, periodontal ligament, and fibroblasts [20, 64–70]. Researchers have explored GFMs as scaffolds, coatings, membrane additives, and drug delivery platforms (Fig. 2), each strategy harnessing their high surface area, mechanical reinforcement, and bioactive surface chemistry.
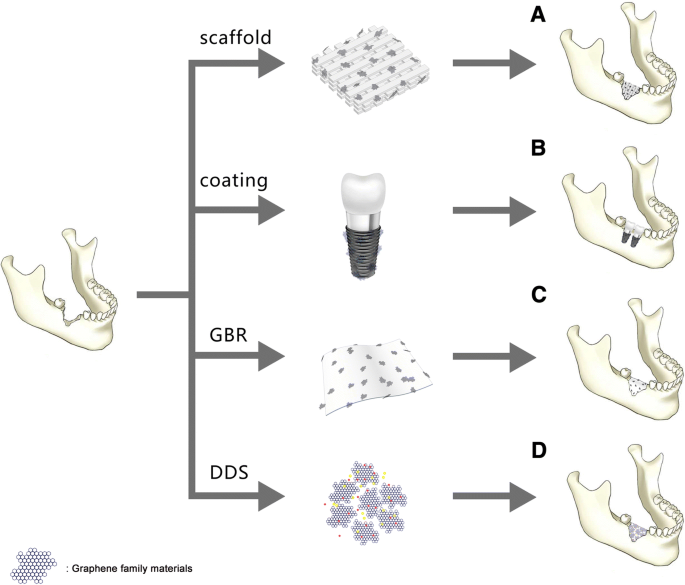
A. Scaffold or reinforcement; B. Surface coating; C. Guided bone membrane additive; D. Drug delivery system.
Scaffold Reinforcement
3D graphene foams (3DGp) support human mesenchymal stem cell (hMSC) viability and osteogenic differentiation [66]. Combining GFMs with calcium‑phosphate or polymeric scaffolds enhances mechanical properties and osteoinductivity. For example, adding 2 wt% GO to poly‑caprolactone increases tensile strength from 1.61 MPa to 3.50 MPa and modulus from 7.01 MPa to 15.15 MPa [80].
Calcium‑Phosphate Composites
Hydroxyapatite (HA) reinforced with GO or rGO improves fracture toughness by >200 % and promotes hMSC osteogenesis via ALP activity and gene expression [89–91]. β‑tricalcium phosphate (β‑TCP) combined with GO activates the Wnt/β‑catenin pathway, enhancing in vitro osteogenic markers and in vivo bone formation in rabbit calvarial defects [85]. Calcium‑phosphate cement (CPC) integrated with GO‑Cu nanocomposites upregulates HIF‑1α, VEGF, and BMP‑2, leading to superior angiogenesis and osteogenesis in rat models [99].
Chitosan Composites
Chitosan (CS) is hydrophilic, osteoconductive, and hemostatic. CS/GO composites (3 wt% GO) exhibit improved water retention, porosity, and ALP activity, translating to enhanced bone formation in vivo [103–104]. Tricomponent CS/GO/HA scaffolds further elevate tensile strength (up to 10 MPa) and promote osteogenic differentiation [2, 87].
Synthetic Polymers
Collagen scaffolds reinforced with GO increase elastic modulus from ~15 kPa to ~39 kPa without compromising cell viability, thereby enhancing MSC osteogenesis through mechanotransduction pathways [107]. Similar improvements are observed with other polymers such as PLA, PLGA, and PCL when combined with GFMs.
Surface Coatings
Graphene coatings on polymeric or metallic substrates accelerate stem cell osteogenic differentiation, matching or surpassing BMP‑2 induction [20, 68, 111]. Titanium (Ti) surfaces modified with GO or rGO exhibit superior hydrophilicity (contact angles 14–20° vs 60° for Ti), increased ALP activity, and enhanced bone‑forming gene expression [117–120]. 3D porous graphene coatings on Ti further improve osseointegration in rat calvarial models [128]. Chemical vapor deposition (CVD) offers scalable, substrate‑independent graphene coatings that preserve surface chemistry and support protein immobilization [122].
Guided Bone Regeneration Membranes
Non‑resorbable GO‑Ti membranes enhance osteogenic cell attachment and bone fill in rat calvarial defects [128]. Resorbable collagen or chitosan membranes supplemented with GO improve mechanical stiffness, reduce deformability, and accelerate bone regeneration [129, 130].
Drug Delivery Systems
GFMs’ high surface area and π‑π stacking capacity enable efficient loading of hydrophobic drugs, growth factors, and antibiotics. For bone regeneration, GO‑loaded simvastatin releases over 30 days, enhancing BMP‑2 and VEGF expression [147]. Dexamethasone (DEX) conjugated to rGO‑Ti surfaces shows sustained release and promotes osteogenic differentiation [143]. Bone morphogenetic protein‑2 (BMP‑2) adsorbed onto GO‑coated Ti retains bioactivity, leading to robust bone formation in murine calvarial defects [132]. Dual delivery of BMP‑2 and antibiotics (e.g., vancomycin) via GO‑PDA‑Ti scaffolds provides antibacterial protection and osteogenesis [149].
Conclusions
Graphene family materials hold significant promise for bone regeneration due to their antibacterial activity, mechanical reinforcement, and ability to induce osteogenic differentiation. Yet, biocompatibility remains a critical hurdle. Cytotoxicity depends on functionalization, concentration, size, and shape; flat, low‑concentration GFMs exhibit the best safety profile. Long‑term in vivo studies are needed to elucidate biodistribution and degradation pathways. When carefully engineered, GFMs can be integrated as scaffolds, coatings, membranes, or drug carriers, offering a multifaceted platform for next‑generation bone repair.
Abbreviations
- ALP:
Alkaline phosphatase
- BMP-2:
Bone morphogenetic protein‑2
- BMSC:
Bone mesenchymal stem cells
- CPC:
Calcium phosphate cements
- CVD:
Chemical vapor deposition
- CXYG:
Carboxyl graphene
- DEX:
Dexamethasone
- ECM:
Extracellular matrix
- GBR:
Guided bone regeneration
- GelMS:
Gelatin microspheres
- GNOs:
Graphene nano‑onions
- GO:
Graphene oxide
- GONPs:
Graphene oxide nanoplatelets
- GONRs:
Graphene oxide nanoribbons
- GQDs:
Graphene quantum dots
- HA:
Hydroxyapatite
- HUVEC:
Human umbilical vein endothelial cells
- MC3T3-E1:
A murine pre‑osteoblastic cell line
- MWNTs:
Multi‑walled carbon nanotubes
- PCL:
Poly‑caprolactone
- PDA:
Polydopamine
- PEG:
Polyethylene glycol
- PLGA:
Poly‑glycolic acid
- rGO:
Reduced graphene oxide
- ROS:
Reactive oxygen species
- SIM:
Simvastatin
- SP:
Substance P
- SPS:
Spark plasma sintering
- Ti:
Titanium
- Van:
Vancomycin
- VEGF:
Vascular endothelial growth factor
- β‑TCP:
β‑Tricalcium phosphate
Nanomaterials
- Graphene‑Based Loudspeakers and Earphones: Ultra‑Low‑Power, High‑Fidelity Sound
- Nanoparticle-Based Cancer Therapy: Advances, Mechanisms, and Clinical Translation
- TiO₂ Nanofluids: Applications, Challenges, and Future Directions – Part 2
- Assessing the Biosafety and Antibacterial Efficacy of Graphene and Graphene Oxide for Orthopedic Implant Applications
- Graphene Oxide-Delivered TIMP‑1 Enhances Skin Regeneration Over 40 Days
- Hybrid Graphene/WO₃ and Graphene/CeOx Electrodes for High‑Performance Supercapacitors
- Enhanced Photocatalytic Degradation of Oxytetracycline by WO3/Graphene Nanocomposites and Device Analysis of Photo‑Induced Doping Mechanisms
- Controlling Terahertz and Near‑Infrared Transmission in FeCl₃‑Intercalated Graphene and WS₂ Thin Films for Advanced THz‑TDS Applications
- Graphene Metasurface for Dual-Function Generation and Steering of Vortex Waves
- Engineering High-Quality Graphene/TMD Heterostructures: Fabrication and Spectral Analysis



