One‑Step Acoustic Levitation Enables Rapid Synthesis of Single‑Atom Platinum Nanoclusters and Pt‑Decorated Microporous La₂O₃ for Catalysis
Abstract
Traditional preparation of supported noble‑metal catalysts typically requires pre‑treatments or complex impregnation protocols to stabilize metal species on a support. Here we introduce a simple, one‑step acoustic levitation approach that yields both single‑atom Pt in solution and highly dispersed Pt nanoclusters on microporous La₂O₃, without any prior modification of the oxide. The levitation process produces a container‑free, uniform acoustic field that suppresses heterogeneous nucleation on vessel walls and promotes rapid nucleation of ultrafine Pt particles. The resulting Pt‑La₂O₃ composites exhibit strong Pt–La–O interfacial bonding and abundant oxygen defects, making them promising candidates for heterogeneous oxidation reactions.
Background
Since the first isolated single‑atom Pt catalyst on FeOx was reported by Qiao et al. [1], the field of single‑atom catalysis has expanded rapidly. Down‑scaling Pt nanoparticles to clusters or single atoms can dramatically increase the catalytic surface area and activity, yet the high surface free energy of such species often leads to sintering under realistic reaction conditions [2, 3]. Conventional strategies to stabilize isolated atoms include defect engineering on reducible oxides [4], coordination with unsaturated Al3+ sites on Al₂O₃ [5], and the use of buffer layers such as Au or Ag on 3D substrates [6, 7]. However, selecting an optimal support that balances stability, dispersion, and loadability remains a major challenge. Acoustic levitation offers a novel route to circumvent these limitations. By levitating the precursor solution within a high‑frequency acoustic field, the liquid is suspended in a container‑free environment, eliminating wall‑induced nucleation and enabling rapid, uniform particle growth. In this study, we demonstrate that acoustic levitation can generate monodispersed Pt nanoclusters and even single‑atom Pt directly in solution, and that these species can be deposited onto microporous La₂O₃ in a single step without any pretreatment.
Results and Discussion
High‑resolution transmission electron microscopy (HRTEM) revealed the distribution of Pt species synthesized by acoustic levitation. In the levitated Pt sol, 10 % of the particles were isolated single atoms, 2 % were sub‑0.5 nm clusters, 25 % were 0.5–1.0 nm clusters, 46 % were 1.0–2.0 nm particles, and 17 % exceeded 2.0 nm. By contrast, a conventional NaBH₄ reduction produced only particles larger than 2.0 nm (100 %) and no single atoms or sub‑nanometer clusters (Figure 1).
Figure 1e shows isolated Pt atoms in the levitated sample, confirming the method’s ability to generate single‑atom sites. The reduced interaction between Pt atoms in the levitated environment likely suppresses sintering, allowing the formation of these ultrafine species [8].
The average Pt diameter in the levitated colloid depends only weakly on the initial H₂PtCl₆ concentration. At 0.00625 g L⁻¹, particles were uniformly 1.65 ± 0.29 nm, with clear (111) lattice fringes (d ≈ 0.26 nm) (Figure 2). No aggregation was observed across the concentration range, highlighting the effectiveness of acoustic levitation in maintaining monodispersity.
When the levitated Pt solution was deposited onto La₂O₃, the resulting Pt/La₂O₃ maintained a mean diameter of ~2.0 nm (Figure 3a). The particles were strongly adsorbed and often irregular in shape, indicating a significant Pt–La–O interaction. HRTEM images showed Pt particles partially buried within the oxide surface, suggesting that the La₂O₃ support actively shapes the Pt morphology and promotes defect formation (Figure 3b–c).
Comparative studies with ultrasonication‑only and conventional colloidal deposition methods revealed that levitation produced smaller, more irregular Pt particles with fewer free‑standing clusters, whereas other methods yielded larger, more spherical particles (Figures 3d–h). The container‑free levitation environment reduces heterogeneous nucleation on vessel walls and enhances acoustic streaming, leading to rapid mass transfer and controlled crystallization [14–16].
By combining levitation with the microporous La₂O₃ support (synthesized hydrothermally from d‑glucose and lanthanum nitrate at 180 °C for 20 h [24]), we achieved a one‑step synthesis of Pt/porous La₂O₃ with an average Pt size of ~2.2 nm and a loading of 1.01 wt% (Figure 4). The porous framework facilitates high surface area and efficient dispersion of Pt nanoclusters.
X‑ray photoelectron spectroscopy (XPS) confirmed the presence of both metallic Pt (Pt 4f7/2 at 71.27 eV) and oxidized Pt species (72.67 eV), indicating surface Pt–OH and PtOx formation. The O 1s spectrum showed lattice oxygen (531.74 eV), adsorbed oxygen (532.44 eV), and electrophilic oxygen species (533.34 eV), consistent with a defect‑rich surface that can facilitate electrophilic oxidation reactions [10, 11].
Overall, acoustic levitation provides a clean, scalable route to fabricate highly dispersed Pt nanoclusters and single‑atom Pt, as well as Pt‑decorated microporous La₂O₃, with strong interfacial bonding and abundant active sites.
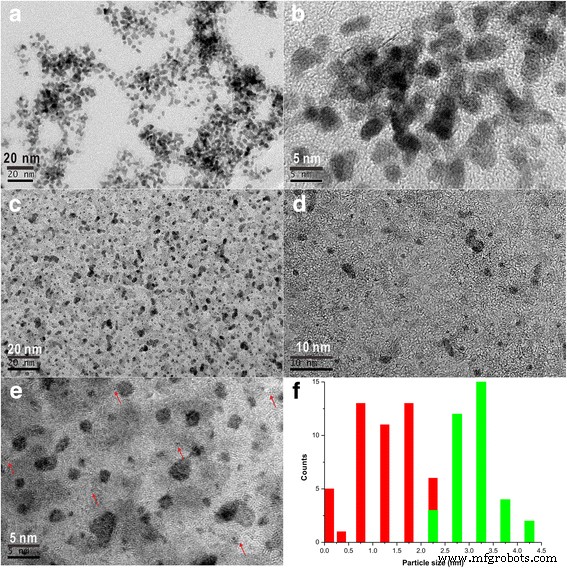
HRTEM images of a, b Pt nanoparticles prepared by NaBH₄ reduction (0.5 g L⁻¹) without levitation, c–e Pt clusters prepared by acoustic levitation (0.5 g L⁻¹), and f particle size distributions (green = NaBH₄, red = levitation).
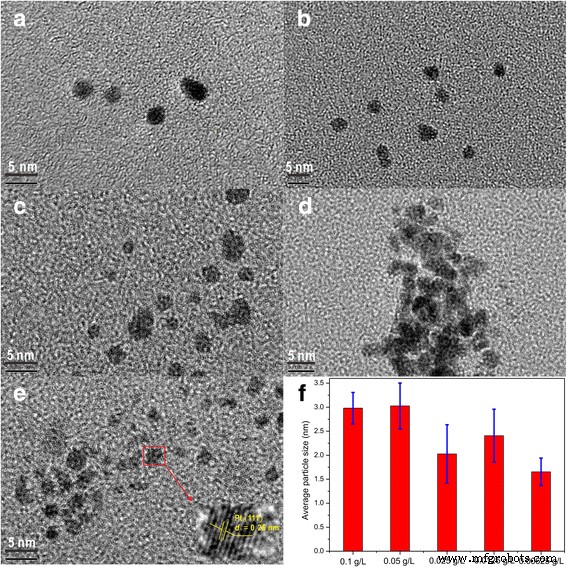
HRTEM images of a–e Pt nanoparticles prepared by levitation at varying concentrations (0.1, 0.05, 0.025, 0.0125, 0.00625 g L⁻¹) and f average particle size vs. concentration.

HRTEM images of Pt nanoparticles supported on La₂O₃: a, b ultrasonication‑acoustic levitation, c, d acoustic levitation, e, f conventional colloidal deposition, g, h ultrasonication‑only deposition.

a, b HRTEM of Pt nanoparticles on La₂O₃ prepared by ultrasonication‑acoustic levitation (0.00625 g L⁻¹).

a, b HRTEM of Pt nanoparticles on La₂O₃ prepared by acoustic levitation (0.00625 g L⁻¹).
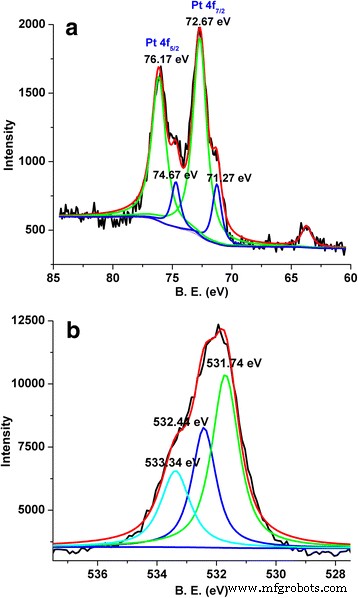
XPS spectra of a Pt 4f and b O 1s for Pt/La₂O₃.
Conclusions
We have demonstrated that acoustic levitation can rapidly produce single‑atom Pt and uniformly dispersed Pt nanoclusters in solution, and that these species can be deposited onto microporous La₂O₃ in a single, pretreatment‑free step. The levitation process induces strong Pt–La–O interfacial interactions, converting Pt from three‑dimensional particles to two‑dimensional layers on the oxide surface and generating oxygen defects that are beneficial for catalytic oxidation. This technique offers a scalable, contamination‑free platform for fabricating high‑performance, surface‑engineered Pt catalysts.
Methods
The acoustic levitator consisted of a 30 kHz emitter and reflector. Liquid samples were levitated by the acoustic radiation force generated by the nonlinear ultrasound field [8].
Preparation of Pt Sol
PVA (polyvinyl alcohol) was added to an aqueous H₂PtCl₆ solution (0.5, 0.1, 0.05, 0.025, 0.0125, 0.00625 g L⁻¹) under vigorous stirring at room temperature. The solution was levitated for several seconds, followed by injection of NaBH₄ (0.005 mol L⁻¹) to reduce Pt and form the sol.
Preparation of Pt/Microporous La₂O₃
The La₂O₃ support was synthesized hydrothermally from d‑glucose and lanthanum nitrate (G:M = 1:1.85) at 180 °C for 20 h [24]. This oxide was added to H₂PtCl₆ (0.5 or 0.00625 g L⁻¹) with or without ultrasonic dispersion, then levitated. Subsequent NaBH₄ injection yielded Pt/La₂O₃ with a 1.01 wt% Pt loading.
Materials Characterization
Morphology was examined with a JEOL JEM‑2100 TEM. Samples were drop‑cast onto carbon grids and dried. Particle sizes were measured from TEM images. X‑ray photoelectron spectroscopy (Perkin‑Elmer, ESCA PHI 5400) was used to determine the oxidation states of Pt and O in the composites.
Abbreviations
- 2D:
Two‑dimensional
- 3D:
Three‑dimensional
- HRTEM:
High‑resolution transmission electron microscopy
- PVA:
Polyvinyl alcohol
- UV:
Ultraviolet
- XPS:
X‑ray photoelectron spectroscopy
Nanomaterials
- Efficient Synthesis and Optical Characterization of Sub‑Micron Selenium Nanocrystals and Nanorods
- Controlled Hydrothermal Synthesis of ZnO Nanocrystals for Enhanced Inverted Polymer Solar Cells
- Pure Copper Nanoparticles via Wood Template: Synthesis, Characterization, and Antibacterial Performance
- Enhanced Supercapacitor Performance via Polyaniline‑Coated Nitrogen‑Doped Ordered Mesoporous Carbon Composites
- CuGeO3 Nanowires: A High‑Capacity, Stable Anode for Advanced Sodium‑Ion Batteries
- One-Pot Synthesis of Binary and Ternary Metal Sulfide Nanocrystals via Metal‑Thiolate Decomposition
- Multiferroic ABO3 Transition Metal Oxides: Harnessing Coupled Ferroelectric and Magnetic Phenomena
- Co‑Doped 3d–4f Metal Polyoxometalates: Controlled Synthesis, Morphology, and Photoluminescence Properties
- CNC Machining: Precision Fabrication of Materials and Components
- High-Performance Metal CNC Manufacturing: Precision, Efficiency, and Cost Savings



