Efficient Chlorin e6 Uptake and Enhanced Energy Transfer in PS–PPO–PNIPAM Hybrid Nanoparticles
Abstract
Polystyrene (PS) nanoparticles doped with diphenyloxazole (PPO) and capped with a cross‑linked poly‑N‑isopropylacrylamide (PNIPAM) shell form PS‑PPO‑PNIPAM hybrid nanosystems (NS). Fluorescence studies of chlorin e₆ added to these NS demonstrate efficient electronic excitation energy transfer (EEET) from both the PS core and the embedded PPO to the chlorin e₆ acceptor. EEET efficiency increases sharply within the first hour after chlorin e₆ addition, indicating that PNIPAM continues to incorporate the sensitizer during this time. Heating the PS‑PPO‑PNIPAM‑chlorin e₆ NS from 21 °C to 39 °C further enhances EEET, consistent with the thermally induced collapse of the PNIPAM network that shortens donor‑acceptor distances. Nonetheless, only a modest fraction of free chlorin e₆ associates with PNIPAM, suggesting that additional optimisation is warranted.
Background
The primary limitation of photodynamic therapy (PDT) for cancer is the shallow penetration of the excitation light into tissue. X‑ray–driven photodynamic therapy (X‑PDT) offers a solution, as X‑rays penetrate deeply and can activate sensitizers that generate singlet oxygen upon X‑ray excitation. Key to X‑PDT is efficient electronic excitation energy transfer (EEET) between a scintillating component and a sensitizer. Strategies to maintain optimal donor–acceptor proximity include chemical conjugation, electrostatic attraction, surfactant layers, or polymer shells. In previous work, we examined EEET in PS‑PPO–chlorin e₆ nanoparticles, where chlorin e₆ was attached to PS‑PPO via a sodium dodecyl sulfate shell.
Poly‑N‑isopropylacrylamide (PNIPAM) is a stimuli‑responsive polymer that undergoes a conformational transition above its lower critical solution temperature (LCST) of 32 °C, becoming more hydrophobic and shrinking. For cross‑linked PNIPAM, the LCST is higher, allowing temperature‑driven size changes at physiological temperatures. We fabricated PS‑PPO nanoparticles enveloped by a cross‑linked PNIPAM shell, yielding PS‑PPO‑PNIPAM hybrid NS. We investigated whether this shell can anchor chlorin e₆ and whether the PNIPAM’s temperature‑dependent collapse can reduce donor‑acceptor distances, thereby boosting EEET and tumor‑cell killing efficacy.
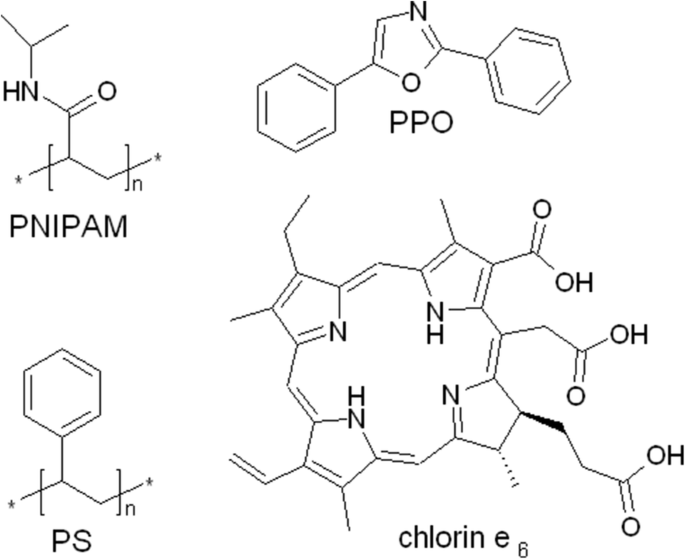
Structures of the components of the PS‑PPO‑PNIPAM hybrid nanosystem and chlorin e₆
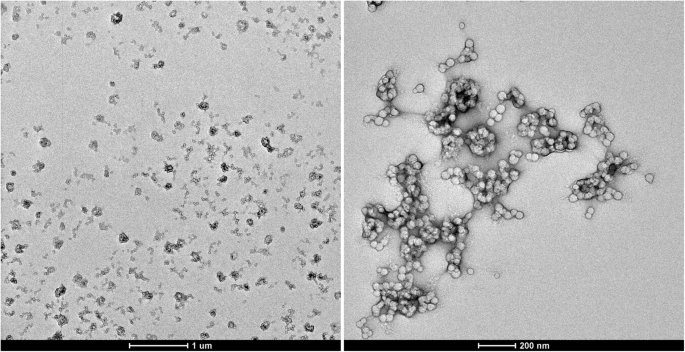
TEM images of the obtained PS‑PPO‑PNIPAM hybrid nanosystems with lower (left) and higher (right) magnification
Experimental
Materials
Styrene (ST, Ukraine) of p.a. quality was purified via standard methods before polymerization. N‑Isopropylacrylamide (NIPAM, Sigma‑Aldrich Inc.), N,N′‑methylenebisacrylamide (BIS, Sigma‑Aldrich Inc.), potassium persulfate (K₂S₂O₈, KPS, Ukraine), sodium phosphate monobasic dehydrate (NaH₂PO₄ × 2H₂O, Ukraine), and anionic surfactant sodium dodecyl sulfate (SDS, Sigma‑Aldrich Inc.) were reagent grade and used without further purification. Chlorin e₆ (Frontier Scientific Inc.) was kindly supplied by T.Y. Ohulchanskyy (Institute for Lasers, Photonics and Biophotonics, State University of New York at Buffalo). A 50 mM Tris‑HCl buffer (pH 7.2) served as solvent.
Synthesis and Characterization of Nanosystems
PS‑co‑PNIPAM core nanoparticles doped with PPO were first prepared by microemulsion polymerization. Briefly, 0.2 g NIPAM, 0.2 g SDS, and 0.01 g NaH₂PO₄ × H₂O were dissolved in 90 g H₂O. Subsequently, 0.09 g PPO was dissolved in 1.8 g styrene and added dropwise over 30 min. The mixture was stirred at 700 rpm, purged with Ar for 30 min, and then heated to 70 °C. 0.01 g K₂S₂O₈ dissolved in 1 mL H₂O initiated polymerization. After 4 h at 70 °C, a cross‑linking step was performed by adding 0.69 g NIPAM and 0.06 g BIS in aqueous solution, followed by 3 h at 70 °C and 1 h at 90 °C. The product was cooled to room temperature and dialyzed for 48 h against a cellulose membrane (MWCO 3500 Da).
Transmission electron microscopy (TEM) images (Fig. 2) confirm that the hybrid NS consist of several PS‑PPO cores bound by a cross‑linked PNIPAM network.
Spectral Measurements and Sample Preparation
Absorption spectra were recorded with a Specord M40 spectrophotometer (Carl Zeiss, Germany). Fluorescence excitation and emission spectra were obtained using a Cary Eclipse fluorimeter (Varian, Australia). All measurements were performed in 1 × 1 cm quartz cells at room temperature. The PS‑PPO‑PNIPAM solution was diluted 100‑fold in Tris‑HCl buffer. A 10 mM chlorin e₆ stock in DMF was diluted to 1 mM in buffer, and 2 µM final concentration of chlorin e₆ was added to the NS solution (DMF ≤ 0.02 %). Fluorescence excitation and emission spectra were collected at 0, 5, 10, 20, 40, 60, 80, and 100 min after addition, with saturation reached after ≈ 80 min.
For temperature‑dependent studies, the NS solution with chlorin e₆ was placed in a thermostatted holder (T = 23 °C). After ≈ 88 min of saturation, the sample was heated to 39 °C (above the LCST of cross‑linked PNIPAM). Fluorescence excitation spectra (chlorin e₆ emission at 680 nm) were recorded at intervals after heating. The conformational transition of PNIPAM began ≈ 3 min after heating, with coagulation occurring after ≈ 18 min—reversible upon cooling. Experiments were repeated three times with consistent results.
Results and Discussion
Absorption, fluorescence excitation, and emission spectra of the PS‑PPO‑PNIPAM NS in 50 mM Tris‑HCl buffer (pH 7.2) are shown in Fig. 3. The absorption profile displays styrene (≈ 260 nm) and PPO (≈ 307 nm) bands. Fluorescence excitation at 250 nm (styrene absorption) yields PPO emission at 367 nm, confirming near‑complete EEET from styrene to PPO. The excitation spectrum of PPO (380 nm emission) further illustrates the contribution of the styrene band. Vibrational structure in the PPO emission, absent in aqueous PPO, confirms its incorporation into the PS matrix.
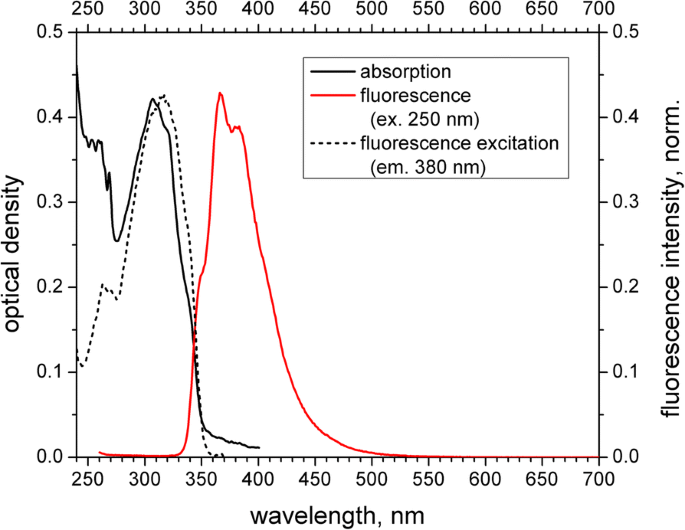
Absorption (black solid line), fluorescence excitation (emission at 380 nm, normalized; black short‑dashed line), and emission (excitation at 250 nm, normalized; red solid line) spectra of the PS‑PPO‑PNIPAM hybrid nanosystems in 50 mM Tris‑HCl buffer (pH 7.2)
Upon addition of chlorin e₆ (2 µM), its absorption spectrum remains largely unchanged (Fig. 4), except for a slight reduction in peak optical density. In contrast, chlorin e₆ addition induces a pronounced quenching of PPO fluorescence, which intensifies over about one hour (Fig. 5). This quenching results from both EEET (donor‑to‑acceptor energy transfer) and reabsorption (donor emission overlapping acceptor absorption). Initially, reabsorption dominates, but as time progresses, EEET becomes the primary contributor.
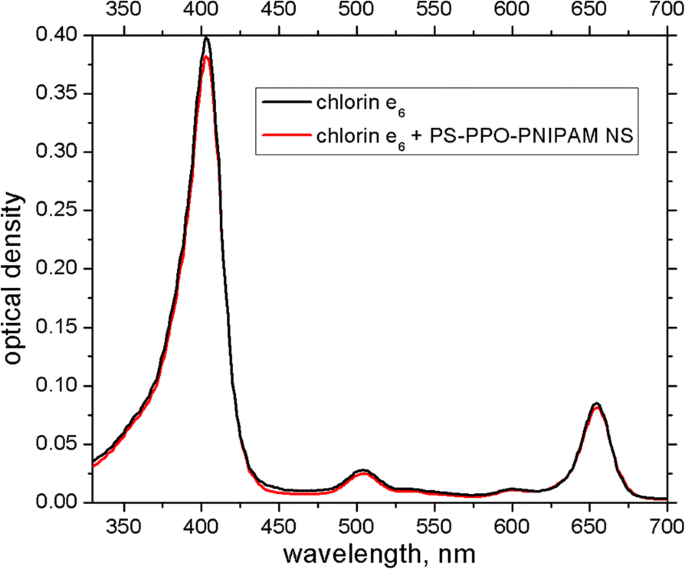
Absorption spectra of chlorin e₆ (2 µM) free (black line) and in the presence of PS‑PPO‑PNIPAM hybrid nanosystems (red line) in 50 mM Tris‑HCl buffer, pH 7.2
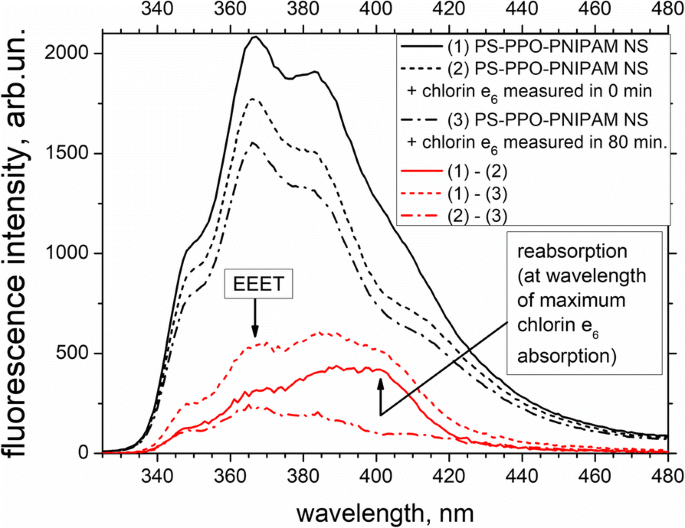
Fluorescence spectra of PS‑PPO‑PNIPAM hybrid NS free (black solid line) and after addition of chlorin e₆ measured at 0 min (black short‑dashed line) and 80 min (black dash‑dotted line). Excitation at 250 nm. Differences (red lines) illustrate the relative contributions of reabsorption and EEET to quenching.
Chlorin e₆ emission appears upon PS excitation at 250 nm, shifting to longer wavelengths over time and reaching saturation after ≈ 60 min (Fig. 6). This red‑shift indicates the influence of the PNIPAM environment on bound chlorin e₆ molecules. Excitation at chlorin e₆’s own Soret band (400 nm) produces only minor changes in emission, underscoring that a small fraction of chlorin e₆ remains free in solution.
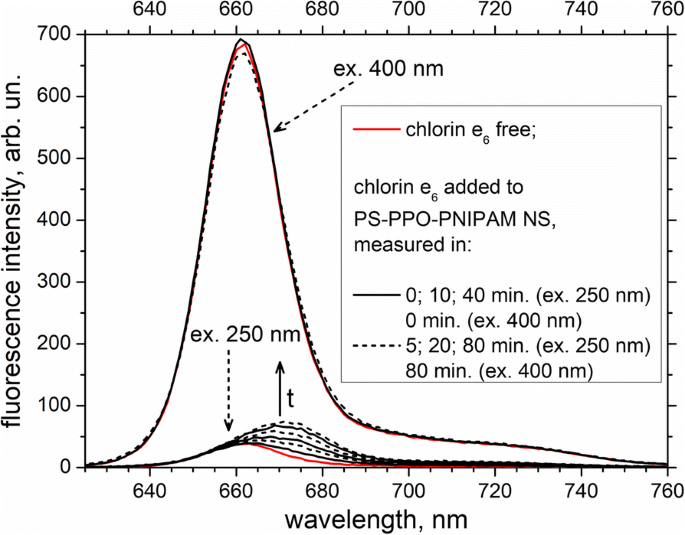
Fluorescence spectra of chlorin e₆ free and added to PS‑PPO‑PNIPAM hybrid NS (measured at 0, 5, 10, 20, 40, and 80 min after addition). Excitation wavelengths: 250 nm (solid arrow) and 400 nm (short‑dashed arrows). 50 mM Tris‑HCl buffer, pH 7.2, was used.
The excitation spectrum of chlorin e₆ (emission at 680 nm) shows a weak PS‑PPO band initially that intensifies over time (Fig. 7), reflecting increased EEET from the donor to the acceptor. The PS‑PPO band remains weak in the excitation spectrum recorded at chlorin e₆ emission (660 nm) even after 100 min, confirming that most energy transfer occurs via the PS‑PPO donor.
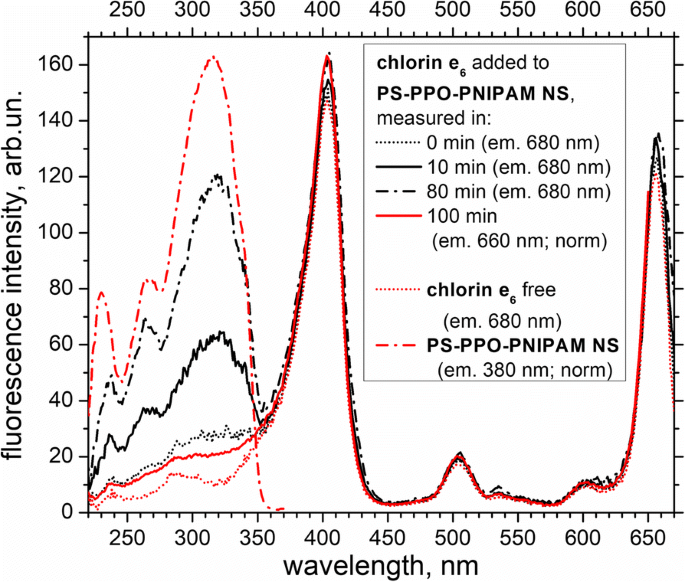
Fluorescence excitation spectra of chlorin e₆ free and added to PS‑PPO‑PNIPAM hybrid NS (measured at 0, 10, 80, and 100 min after addition), and PS‑PPO‑PNIPAM NS itself. Emission wavelengths: chlorin e₆ at 660 nm (normalized intensity) and 680 nm; PS‑PPO‑PNIPAM NS at 380 nm. 50 mM Tris‑HCl buffer, pH 7.2, was used.
Collectively, these results demonstrate that chlorin e₆ penetrates the PNIPAM network, enabling EEET from PS and PPO to the sensitizer. EEET efficiency rises during the first hour of incubation and can be further enhanced by heating the system above the LCST of cross‑linked PNIPAM, which contracts the polymer network and shortens donor‑acceptor distances.
Previous studies have shown that PS‑PPO nanoparticles emit PPO fluorescence under X‑ray excitation, suggesting that the EEET observed here under UV illumination would also operate under X‑ray irradiation. However, detecting X‑ray‑induced emission typically requires high NP concentrations; incorporating heavy‑atom–containing moieties could increase X‑ray absorption and improve sensitivity.
In the temperature‑dependent experiment (Fig. 8‑9), heating from 23 °C to 39 °C (post‑saturation) first caused a modest drop in the I_ex320/I_ex404 ratio (from 0.90 to 0.85), possibly reflecting reduced chlorin e₆–PNIPAM affinity. Subsequently, the ratio rose to 1.02, coinciding with increased light scattering—indicative of PNIPAM collapse and closer donor‑acceptor proximity, thereby boosting EEET.
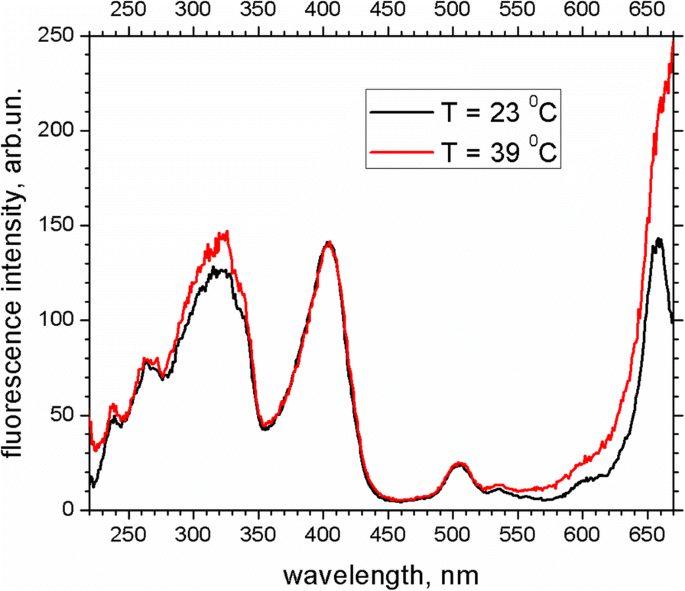
Fluorescence excitation spectra (normalized at 404 nm) of chlorin e₆ added to PS‑PPO‑PNIPAM hybrid NS at 23 °C (black line) and at 39 °C (red line, 14 min after heating). Emission at 680 nm; 50 mM Tris‑HCl buffer, pH 7.2.
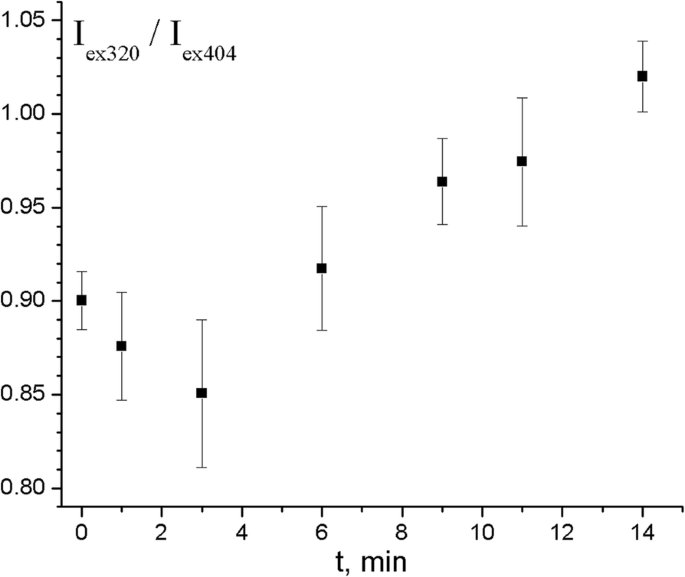
Time dependence of the I_ex320/I_ex404 ratio for chlorin e₆ added to PS‑PPO‑PNIPAM hybrid NS after heating to 39 °C. Error bars reflect spectral noise.
In summary, the PS‑PPO‑PNIPAM hybrid NS facilitate chlorin e₆ uptake and EEET, with efficiency increasing over ~1 h of incubation and further rising upon heating above LCST. Nevertheless, only a limited fraction of chlorin e₆ binds to PNIPAM, highlighting the need for future optimization.
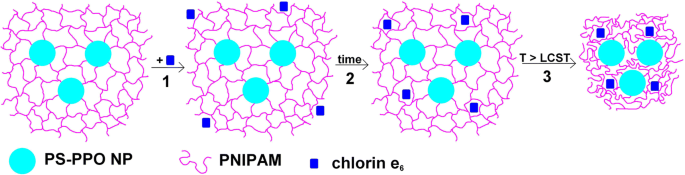
Proposed scheme of processes in PS‑PPO‑PNIPAM hybrid NS with chlorin e₆: (1) initial uptake, (2) ~1 h penetration and EEET build‑up, (3) temperature‑induced PNIPAM collapse and further EEET enhancement.
Conclusions
Fluorescence analysis confirms that chlorin e₆ is incorporated into PS‑PPO‑PNIPAM hybrid NS and that EEET from the PS matrix and embedded PPO to chlorin e₆ occurs. Uptake saturates after roughly one hour. Heating the system from 21 °C to 39 °C further boosts EEET, consistent with the thermally driven collapse of the cross‑linked PNIPAM shell, which brings donor and acceptor closer together. Although only a modest amount of chlorin e₆ remains bound to PNIPAM, these findings support the potential of such hybrid NS in X‑ray‑driven photodynamic therapy and warrant further studies to enhance sensitizer loading.
Abbreviations
- EEET:
Electronic excitation energy transfer
- LCST:
Lower critical solution temperature
- NS:
Nanosystem
- PNIPAM:
Poly‑N‑isopropylacrylamide
- PPO:
Diphenyloxazole
- PS:
Polystyrene
- TEM:
Transmission electron microscopy
Nanomaterials
- Hybrid Supercapacitors: A Unified Solution for Advanced Energy Storage
- Impact of Shell Thickness on Energy Transfer in Eu‑Doped Core–Shell ZnSe/ZnS Quantum Dots
- Electronic Structure and Transport in InSe Nanoribbons: Edge Effects and Hydrogen Passivation
- Highly Sensitive and Selective Hg²⁺ Detection via FRET between CdSe Quantum Dots and g‑C₃N₄ Nanosheets
- Hybrid Verapamil‑Dextran Nanostructured Lipid Carriers: Statistically Optimized Formulation for Enhanced Cellular Uptake and Sustained Release
- Hydrothermal Synthesis of 19 nm Sc₂O₃:Er³⁺,Yb³⁺ Nanoparticles with 4× Superior Red Upconversion Luminescence
- Predicting Surface Impedance of Metasurface–Graphene Hybrid Structures in the Terahertz Regime
- Enhancing Transfer Charge in Knit-Structured Triboelectric Nanogenerators via Surface Morphology Analysis
- Targeted NIR-Responsive PNIPAM‑Pyrrole Nanocomposites for Synergistic Chemo‑Photothermal Breast Cancer Therapy
- Effective PCB Heat Management: Techniques for Optimal Thermal Performance



