Tuning Graphene Morphology via Ion‑Enhanced Cathodic Plasma Exfoliation for High‑Performance Supercapacitors
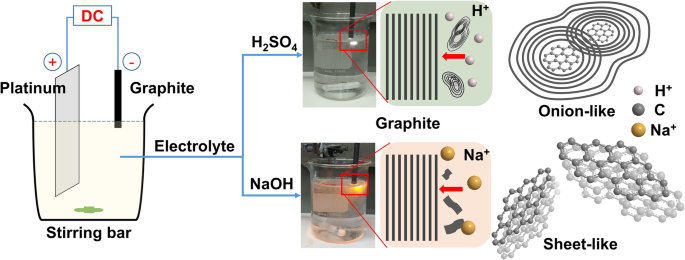
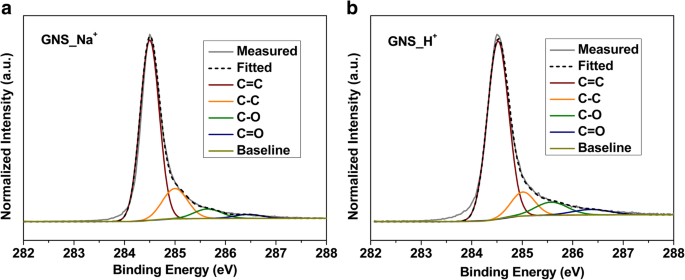
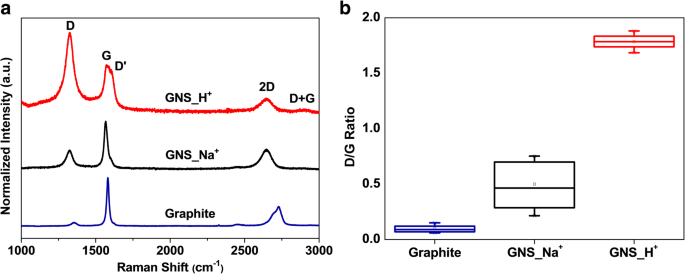
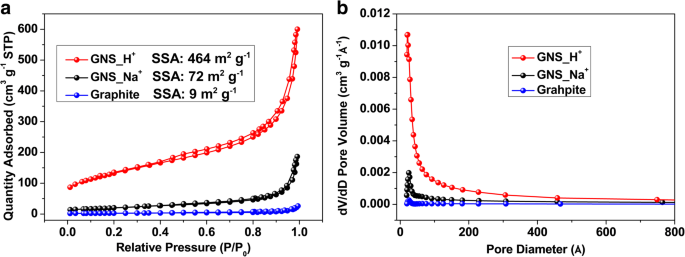
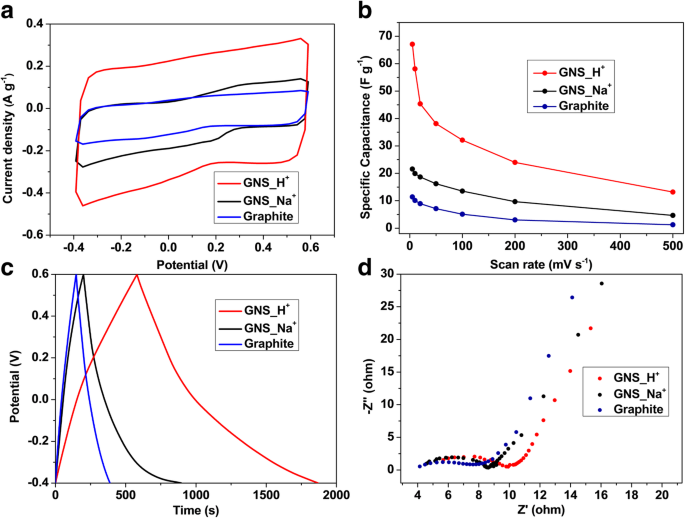
We report a facile, ambient‑temperature cathodic plasma exfoliation approach that uses electrolyte ions to steer the architecture of graphene nanosheets (GNS). By selecting either aqueous NaOH or H₂SO₄, we obtain distinct sheet‑like or onion‑like GNS, respectively, as verified by SEM and TEM imaging. The onion‑like GNS display a remarkably high specific surface area of 464 m² g⁻¹ and a specific capacitance of 67.1 F g⁻¹ at 5 mV s⁻¹ in 1 M NaCl—substantially higher than the sheet‑like GNS (72 m² g⁻¹, 21.6 F g⁻¹). These findings demonstrate that electrolyte ion selection can be exploited to tailor graphene morphology for enhanced energy‑storage performance. The rapid depletion of fossil fuels and growing environmental concerns have spurred intensive research into advanced energy‑storage technologies, including batteries and supercapacitors. Supercapacitors, in particular, are prized for their high power density and long cycle life, and are categorized into pseudocapacitors—where Faradaic reactions dominate—and electrical double‑layer capacitors (EDLCs), which rely on ion adsorption onto high‑surface‑area electrodes. Two‑dimensional materials such as graphene and transition‑metal dichalcogenides (TMDCs) have emerged as promising electrode candidates owing to their exceptional electrical conductivity, mechanical flexibility, and tunable surface chemistry. While high‑quality graphene can be produced via mechanical, epitaxial, or chemical vapor deposition methods, these approaches are costly and require sophisticated equipment. In contrast, solution‑phase exfoliation techniques—ball‑milling, arc‑discharging, and electrochemical methods—offer scalable routes to produce powdery graphene with high specific surface area. However, conventional electrochemical exfoliation often suffers from re‑aggregation of graphene layers due to π‑stacking, limiting the accessible surface area and, consequently, the capacitance. Recent efforts have focused on structural engineering strategies, such as sacrificial‑template synthesis and aerogel fabrication, to mitigate layer restacking. Nonetheless, these processes are typically multi‑step, time‑consuming, and involve hazardous reagents. There remains a clear need for a simple, one‑step, green synthesis that can produce graphene with controllable morphology and high surface area under ambient conditions. In this work, we introduce a cathodic plasma exfoliation protocol that leverages the ion composition of the electrolyte to dictate the resulting GNS architecture. By tuning the electrolyte between NaOH and H₂SO₄, we achieve sheet‑like or onion‑like morphologies, respectively. We systematically characterize the structural, compositional, and electrochemical properties of the resulting materials, highlighting their potential for high‑performance supercapacitor electrodes. All reagents were used as received: sodium hydroxide (NaOH) and sulfuric acid (H₂SO₄) from Sigma–Aldrich; graphite rods (Ø 7 mm, 70 mm) and 99.5 % graphite powder from Toyo Tanso; platinum plates (200 mm × 30 mm × 0.7 mm) from Guang Yi Eleciron Chemical Equipment. Cathodic plasma exfoliation was conducted with a graphite rod as the cathode and a platinum plate as the anode, submerged at depths of 10–20 mm and 60–120 mm, respectively, in 2 M H₂SO₄ or 4 M NaOH solutions. A 60 V potential was applied using a LinVac Tech power supply. GNS were collected by vacuum filtration (0.2 µm Millipore nylon), washed, and dried at 70 °C for 3 h. For further characterization, the GNS were dispersed in ethanol (0.3 mg mL⁻¹) and centrifuged (2000 rpm, 30 min) to remove unexfoliated graphite. The samples derived from H₂SO₄ and NaOH are denoted GNS_H⁺ and GNS_Na⁺, respectively. Field‑emission SEM (Hitachi SU‑8010) and high‑resolution TEM (JEOL ARM200F) were employed to assess morphology and layer thickness. Raman spectroscopy (HORIBA LabRAM HR, 632.8 nm) quantified defect density via I_D/I_G ratios. Nitrogen adsorption–desorption (Micromeritics ASAP 2020) provided BET surface areas and BJH pore size distributions. X‑ray photoelectron spectroscopy (XPS, ULVAC PHI Quantera SXM) determined surface elemental composition. X‑ray diffraction (Bruker D2, Cu Kα, λ = 0.1542 nm) confirmed crystallographic changes. Electrochemical performance was evaluated in 1 M NaCl using a three‑electrode configuration: saturated calomel reference, platinum counter, and graphite paper working electrode. Electrode inks comprised 90 wt % active material, 10 wt % PVDF, and NMP, cast on graphite paper and dried at 80 °C overnight. Cyclic voltammetry (CV) was performed from –0.4 V to +0.6 V; specific capacitance (C) was calculated via C = ∫I dv/(2 v Δm ΔV). Electrochemical impedance spectroscopy (EIS) spanned 100 mHz–100 kHz with 5 mV amplitude. Figure 1 illustrates the cathodic plasma exfoliation setup and the resulting morphologies obtained with NaOH (sheet‑like) and H₂SO₄ (onion‑like) electrolytes. The low cathode–anode surface area ratio (1:10) generates a high current density at the submerged cathode, producing rapid water vaporization and plasma formation that exfoliates graphene sheets. The ionic species in the electrolyte influence the plasma chemistry: Na⁺ ions promote mechanical exfoliation, whereas H⁺ ions, due to their smaller size, facilitate bond cleavage and lead to the formation of onion‑like structures. Schematic representations of the experimental setup and the cathodic plasma exfoliation processes using NaOH and H₂SO₄ as electrolytes, along with digital images of the experiments FESEM images (Figure 2a,b) reveal smooth, layered sheets for GNS_Na⁺ and a broccoli‑like, multi‑layered morphology for GNS_H⁺. TEM and HRTEM confirm that GNS_Na⁺ consists of ~4‑layer graphene sheets, whereas GNS_H⁺ displays concentric, onion‑like shells 4–10 nm in diameter with 4–6 graphene layers. Fast Fourier transform (FFT) patterns show hexagonal symmetry in both samples, indicating preserved lattice structure. Thickness statistics (Additional File 1: Figure S1a) show that ~87 % of GNS_H⁺ and ~74 % of GNS_Na⁺ are ≤6 layers, underscoring the uniformity advantage conferred by H⁺ ions. a, b FESEM images of a GNS_Na⁺ and b GNS_H⁺; c, d bright‑field TEM images and e, f HRTEM images with corresponding FFT images of c, e GNS_Na⁺ and d, f GNS_H⁺ XPS survey spectra (Additional File 1: Figure S2a) indicate only C and O, with GNS_H⁺ showing a higher O content (6.1 at.%) than GNS_Na⁺ (2.1 at.%). Deconvoluted C 1s peaks reveal sp² (284.5 eV), sp³ (285.1 eV), C–O (285.7 eV), and C=O (287.1 eV) contributions; the elevated oxidation of GNS_H⁺ aligns with the increased defect density inferred from Raman spectroscopy. High‑resolution C 1s XPS spectra of a GNS_Na⁺ and b GNS_H⁺ Raman spectra (Figure 4a) display D (1325 cm⁻¹), G (1571 cm⁻¹), and 2D (2648 cm⁻¹) bands. The I_D/I_G ratios of 0.08 (graphite), 0.46 (GNS_Na⁺), and 0.79 (GNS_H⁺) confirm that both exfoliated samples possess increased defect density, with GNS_H⁺ exhibiting the highest level of disorder—consistent with its onion‑like architecture and higher oxidation state. a Raman spectra and b box charts of the intensity ratios of the D- and G-bands of graphite, GNS_Na⁺, and GNS_H⁺ Nitrogen adsorption–desorption isotherms (Figure 5a) reveal H3 hysteresis loops, indicative of mesoporosity. BET surface areas are 9 m² g⁻¹ (graphite), 72 m² g⁻¹ (GNS_Na⁺), and 464 m² g⁻¹ (GNS_H⁺). BJH analysis (Figure 5b) shows dominant pore sizes of 2.5 nm (graphite), 14.9 nm (GNS_Na⁺), and 9.2 nm (GNS_H⁺), with GNS_H⁺ possessing the largest pore volume (0.928 cm³ g⁻¹), a key factor for efficient ion transport. a Typical N₂ adsorption/desorption isotherms and b BJH pore size distribution of graphite, GNS_Na⁺, and GNS_H⁺ Electrochemical testing (Figure 6a) shows quasi‑rectangular CV curves for all samples, indicative of EDLC behavior. Specific capacitances (Table 1) are 11.4 F g⁻¹ (graphite), 21.6 F g⁻¹ (GNS_Na⁺), and 67.1 F g⁻¹ (GNS_H⁺) at 5 mV s⁻¹. The onion‑like GNS_H⁺ outperforms its sheet‑like counterpart across scan rates up to 500 mV s⁻¹ (Figure 6b), thanks to its higher surface area and pore volume. Galvanostatic charge/discharge (Figure 6c) yields symmetrical triangular profiles with negligible IR drops, confirming low internal resistance. EIS (Figure 6d) shows Rs values of 3.5 Ω (graphite), 3.9 Ω (GNS_Na⁺), and 4.6 Ω (GNS_H⁺) and charge‑transfer resistances (R_ct) of 3.5, 3.9, and 4.6 Ω, respectively, demonstrating that the onion‑like structure does not compromise conductivity. Cycle life tests (Additional File 1: Figure S4) reveal >90 % capacitance retention after 1000 cycles at 100 mV s⁻¹, underscoring the durability of both GNS variants. a Cyclic voltammograms of the various samples, recorded at 5 mV s⁻¹. b Specific capacitances of the samples, determined at scan rates from 5 to 500 mV s⁻¹. c Galvanostatic charge/discharge curves recorded at 0.1 A g⁻¹. d Nyquist plots of the samples measured from 100 mHz to 100 kHz; inset: corresponding expanded high‑frequency region of the plots We have demonstrated that the ion composition of the electrolyte in cathodic plasma exfoliation governs the morphology of graphene nanosheets. NaOH yields planar, sheet‑like GNS, while H₂SO₄ produces onion‑like GNS with a surface area of 464 m² g⁻¹ and a specific capacitance of 67.1 F g⁻¹. The superior electrochemical performance of the onion‑like GNS stems from their high surface area, abundant mesoporosity, and preserved crystallinity, which together enable efficient ion access and rapid charge transport. This one‑step, ambient‑temperature method offers a scalable route to tailor graphene architectures for high‑performance supercapacitors and other energy‑storage applications. Brunauer–Emmett–Teller Cyclic voltammogram Electrical double‑layer capacitor Field‑emission scanning electron microscopy Fast Fourier transform Full width at half maximum Graphene nanosheets Graphene nanosheets produced using H₂SO₄ Graphene nanosheets produced using NaOH Sulfuric acid Potassium hydroxide Sodium chloride Sodium hydroxide N‑Methyl‑2‑pyrrolidone Transmission electron microscopy X‑ray photoelectron spectroscopy X‑ray diffractionAbstract
Introduction
Methods
Materials
Characterization
Electrochemical Measurements
Results and Discussion

Conclusion
Abbreviations
Nanomaterials
- IBM Breakthrough: Controlling a Single Copper Atom’s Magnetism via NMR
- Graphene‑Polymer Composites for High‑Performance Supercapacitors: A Comprehensive Review
- Hybrid Graphene/WO₃ and Graphene/CeOx Electrodes for High‑Performance Supercapacitors
- Highly Compressible Graphene/Polyaniline Aerogel: Superelasticity Meets 713 F g⁻¹ Capacitance for All‑Solid‑State Supercapacitors
- High‑Quality Few‑Layer Graphene Produced by a Simple Needle‑Valve Hydrodynamic Exfoliation
- Cryogenic Cycling Rejuvenates Zr₅₀Cu₄₀Al₁₀ Bulk Metallic Glass: The Role of Casting Temperature on Microstructure and Properties
- Enhanced Red Upconversion in Single β‑NaYF4:Yb/Er Microcrystals Doped with Mn²⁺: Tunable Multicolor Emission and Energy‑Transfer Dynamics
- Predicting Surface Impedance of Metasurface–Graphene Hybrid Structures in the Terahertz Regime
- High-Performance Supercapacitor Electrodes from Hierarchically Porous Carbon Derived from Lignosulfonate Biowaste
- Controlled Low‑Temperature Growth of WSe₂ on Graphene for Efficient p‑Type Transistors



