Fluorescent Soybean‑Derived Nano‑Biomass Dots via Ultrasonic Extraction for Sensitive Fe3+ Detection
Abstract
Soybean biomass, a renewable feedstock, has been converted into luminescent nano‑biomass dots (NBDs) through a heating‑free ultrasonic extraction strategy. The resulting amorphous particles possess an average diameter of 2.4 nm and emit bright blue fluorescence with a quantum yield of 16.7 %. Cytotoxicity assays demonstrate complete biocompatibility up to 800 µg mL-1. Importantly, the NBD fluorescence is highly responsive to Fe3+, enabling detection with a limit of 2.9 µM—well below the U.S. EPA threshold for drinking water. This green platform offers a scalable, low‑toxicity probe for environmental and biomedical applications.
Background
Luminescent nanomaterials have revolutionised fields ranging from light‑emitting diodes to bioimaging and metal‑ion sensing [1,2,3,4,5,6]. Conventional quantum dots (QDs) deliver outstanding optical properties but suffer from inherent toxicity [13,14]. Consequently, there is a strong drive to identify greener, sustainable luminescent platforms.
Biomass—organic material generated via photosynthesis—offers a renewable source for nanomaterial synthesis. Edible biomass, rich in sugars and proteins, can be transformed into nanodots with minimal environmental impact, ensuring high biocompatibility for biological and environmental uses [15,16]. To date, most biomass‑derived fluorescent nanodots have required high‑temperature carbonisation, limiting scalability and preserving the native structure of the feedstock [22].
Fluorescent nano‑biomass dots (NBDs) have yet to be reported as a green, efficient probe for Fe3+ detection. Fe3+ plays a vital role in haemoglobin synthesis but excessive levels lead to oxidative damage and organ failure, underscoring the need for sensitive, environmentally friendly sensors [24].
Here, we demonstrate the first ultrasonic‑extracted soybean‑derived NBDs with bright blue emission, a 16.7 % quantum yield, and excellent biocompatibility. Their fluorescence is selectively quenched by Fe3+, providing a detection limit of 2.9 µM.
Methods
Materials
Analytical‑grade reagents were used without further purification. Soybeans were sourced from a local supermarket, washed, and stored at room temperature. Calcium chloride, manganese chloride, cupric chloride, cobaltous chloride, lead nitrate, chromium nitrate, ferric chloride, ferrous chloride, cadmium chloride, mercury dichloride, sodium chloride, and zinc chloride were purchased from Aladdin Ltd. (Shanghai, China) and Sinopharm Chemical Reagent Co., Ltd. (Shanghai, China).
Synthesis of NBDs
One hundred soybean seeds were rinsed with an ethanol–water mixture to remove surface impurities. They were then immersed in 50 mL distilled water and subjected to ultrasonic agitation for 2 h. The solution’s colour shifted from transparent to dark yellow, indicating nanoscale fragmentation. Following two 3‑minute centrifugation steps at 7,000 rpm, the supernatant was filtered through a 0.22 µm membrane to remove aggregates. The filtrate was frozen at –5 °C for 6 h, lyophilised at –50 °C for 12 h, and the resulting powder was re‑dispersed in water to yield the final NBDs.
Characterization
X‑ray diffraction (XRD) was performed on an X′ Pert Pro diffractometer using a Cu‑Kα source. Transmission electron microscopy (TEM) and high‑angle annular dark‑field scanning TEM (HAADF‑STEM) were carried out on a JEM‑2010 microscope. Fluorescence and UV‑Vis absorption spectra were recorded on an F‑7000 fluorescence spectrophotometer and a UH4150 spectrophotometer, respectively. Fourier‑transform infrared (FTIR) spectra were collected with a Thermo Scientific Nicolet iS10 FTIR spectrometer. X‑ray photoelectron spectroscopy (XPS) was conducted on a Thermo Fisher Scientific ESCALAB 250Xi equipped with an Al‑Kα source.
Photoluminescence Quantum Yield Measurement
Quantum yield (QY) was measured using an F‑9000 spectrofluorometer coupled with an integrating sphere. An aqueous NBD solution (absorptance <0.1) was excited at 370 nm, and emission was recorded from 430 to 450 nm. Reference spectra of pure water were acquired under identical conditions, and QY was calculated using the integrated fluorescence software.
Cellular Toxicity Test
MTT assays evaluated NBD cytotoxicity. HeLa cells were seeded in 96‑well plates, incubated for 72 h in RPMI‑1640 with 10 % fetal bovine serum, and exposed to varying NBD concentrations. Cell viability was assessed by measuring absorbance at 570 nm.
Detection of Fe3+
A 1 mL aliquot of NBD solution (3 g L-1) was mixed with 1 mL of Fe3+ solution at different concentrations. After a 1‑minute incubation at room temperature, fluorescence spectra were recorded under 370 nm excitation.
Results and Discussion
Morphology and Chemical Composition
The ultrasonic extraction process (Scheme 1) yields nearly spherical NBDs with diameters ranging from 1 to 3 nm (average 2.4 nm). TEM images (Fig. 1a,b) confirm their amorphous nature, as no lattice fringes are observed. HAADF‑STEM and elemental mapping (Fig. 1d–g) reveal a composition dominated by carbon, nitrogen, and oxygen. Solid‑state 13C NMR (Fig. 1h) shows signals at 164 and 170 ppm, indicative of C=O bonds and sp2 carbon, respectively.
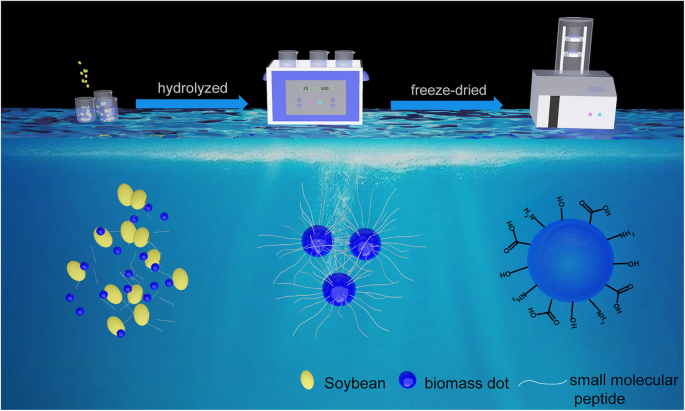
Schematic illustration of the preparation process of the NBDs from soybeans
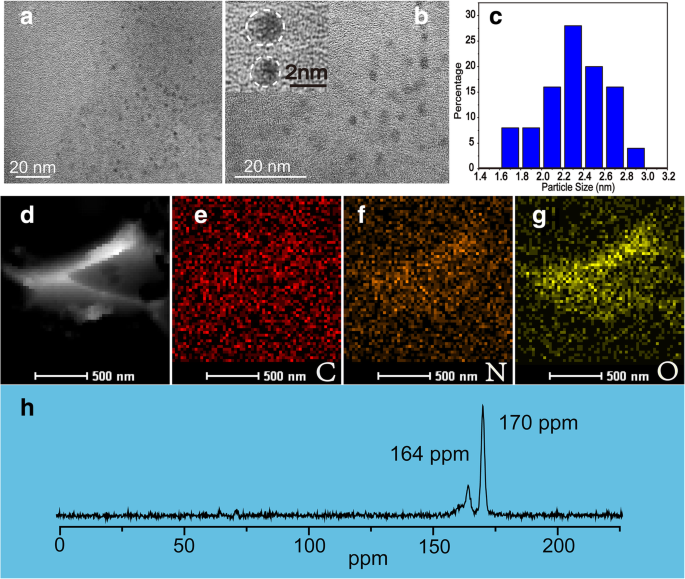
TEM images of the NBDs (a) and (b). c The particle size distribution of the NBDs. HAADF image (d) and corresponding elemental distribution mapping of carbon (e), nitrogen (f), and oxygen (g)
XRD (Fig. 2a) displays a broad peak at ~21.5° and a shoulder at ~41.0°, characteristic of amorphous carbon. FTIR spectra (Fig. 2b) show O–H/N–H stretching (~3380 cm-1), C–H stretching (~2906 cm-1), and C=O stretching (~1650 cm-1). Notably, the ~1750 cm-1 band, attributed to soybean lipids, disappears after ultrasonic extraction, indicating removal of insoluble lipids. XPS (Fig. 2c–f) confirms a composition of 64.33 % C, 32.34 % O, and 2.72 % N, with functional groups including C=O, C–O/C=N, C–C/C=C, pyridinic N, pyrrolic N, C–OH/C–O–C, and C=O. These surface groups confer hydrophilicity and stability in aqueous media.
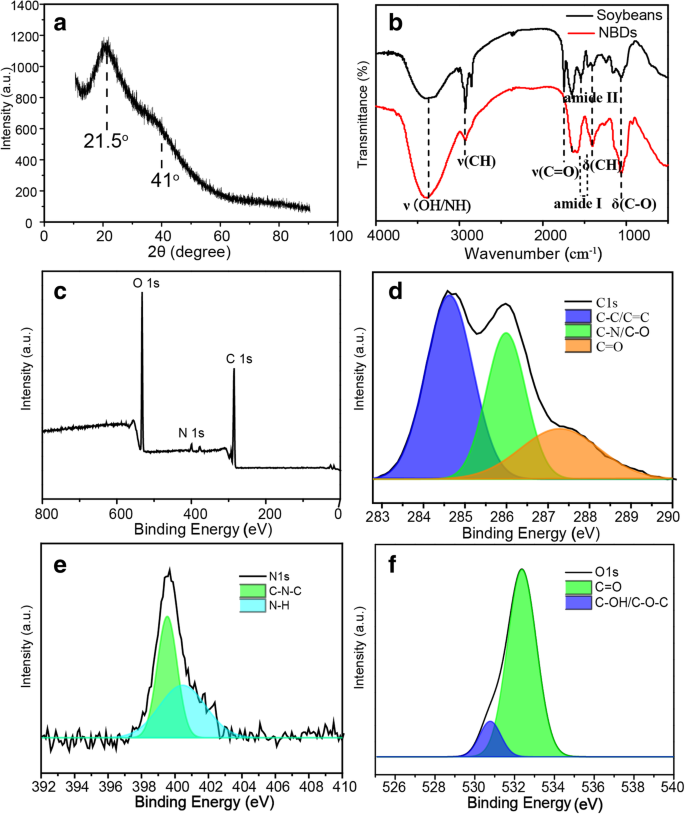
a XRD pattern of the NBDs. b FTIR spectra of the soybean and NBDs. c XPS survey spectrum of the NBDs. High‑resolution XPS spectra of C 1s (d), N 1s (e), and O 1s (f)
The formation mechanism involves ultrasonic disruption of soybean particles into nanometric fragments, hydrolysis of proteins into peptides and amino acids, and surface functionalisation of the nano‑dots with abundant –OH, –C=O, and –NH groups, which are the primary contributors to fluorescence.
Optical Properties
The NBDs exhibit excitation‑dependent fluorescence: as the excitation wavelength is varied from 320 to 520 nm, the emission peak red‑shifts, enabling spectral tuning (Fig. 3a). In aqueous solution, the NBDs are transparent under ambient light and fluoresce blue under UV illumination (inset, Fig. 3b). PLE spectra (Additional file 1: Fig. S3) reveal an excitation window of 360–420 nm. UV–Vis absorption (Fig. 3b) shows peaks at 270 nm (π–π* of C–C/C=C) and 330 nm (n–π* of C=O/N). Photoluminescence intensity decreases monotonically with temperature (Fig. 3c), consistent with increased non‑radiative recombination. Photostability tests indicate >90 % retention after 6 h of UV exposure, and the NBDs remain stable across 20–80 °C (Fig. 3d).
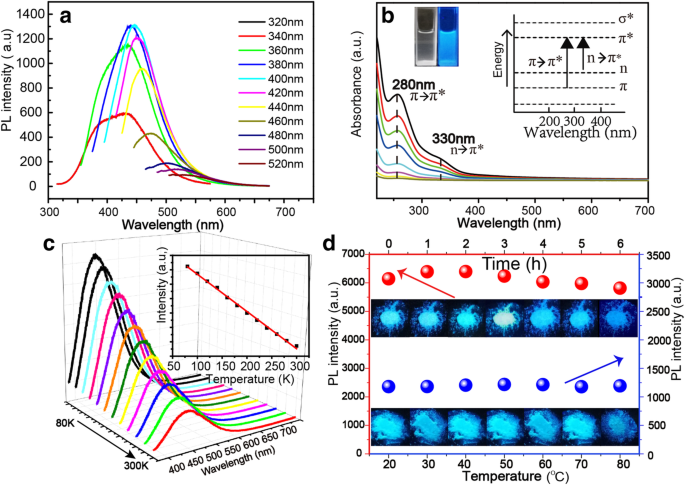
a Fluorescence spectra of the NBDs with excitation wavelength changes from 320 to 520 nm. b UV‑Vis absorption spectra of the NBDs. c Fluorescence spectra of the NBDs at different temperature, the inset is the plot of the fluorescence intensity of the NBDs as a function of temperature. d Fluorescence intensity and images of the NBD powders under the illumination of a 365‑nm lamp for different duration and those of the NBD powders at different measuring temperature
Cytotoxicity Evaluation
MTT assays with HeLa cells (Fig. 4) show negligible cytotoxicity: cell viability remains >90 % even at 800 µg mL-1 of NBDs. In contrast, two chemically synthesized carbon dots exhibit viability of 70 % and 67 % at the same concentration, underscoring the superior biocompatibility of biomass‑derived NBDs.
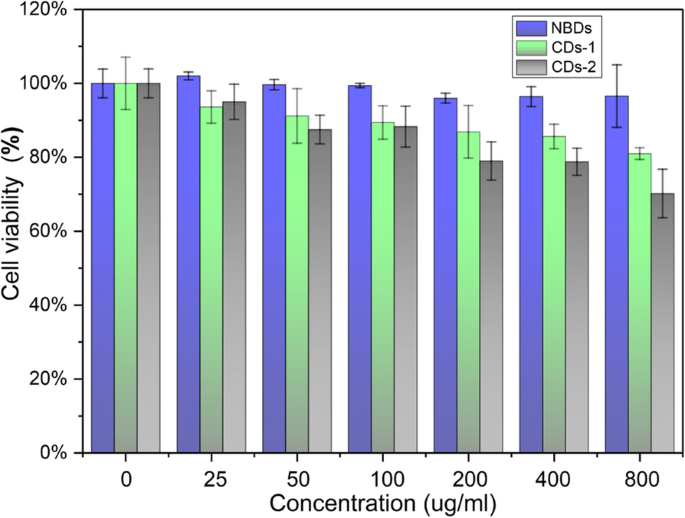
Viability of HeLa cells after 72 h of incubation with different concentrations of NBDs and CDs
Sensing Properties of the NBDs Toward Fe3+
Fluorescence quenching by Fe3+ is pronounced (Fig. 5a). A linear relationship between F0/F and Fe3+ concentration (0–30 µM) yields an R2 of 0.99 (Fig. 5b). The Stern‑Volmer equation gives KSV = 0.0072 µM-1, and the limit of detection (LOD) is 2.9 µM—below the U.S. EPA drinking‑water limit of 5.37 µM [24]. Selectivity tests (Fig. 5c) confirm negligible response to Ca2+, Cd2+, Co2+, Cu2+, Cr3+, Fe2+, Hg2+, Mn2+, Na+, Pb2+, and Zn2+, while Fe3+ produces the most significant quenching. Visual inspection under UV light (Fig. 5d) demonstrates a clear colour change, enabling naked‑eye detection.
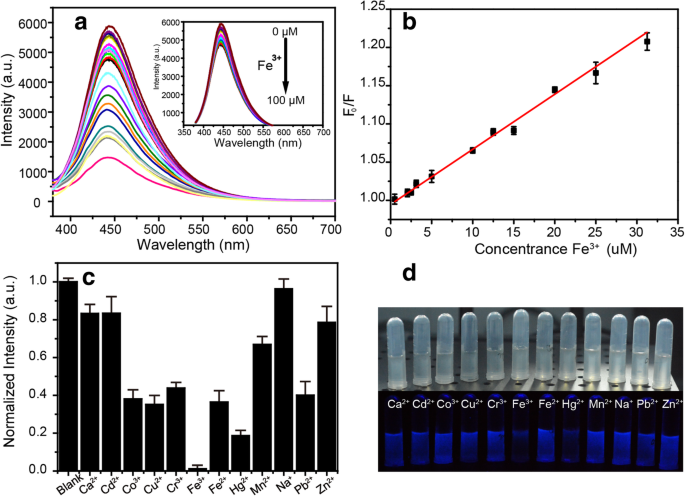
a PL spectra of the NBDs in the presence of different concentrations of Fe3+. b Calibration curve of the sensor as a function of Fe3+ concentration. c Fluorescence intensities of the NBDs in the presence of different ions. d Photographic images of the NBDs solution with different metal ions under indoor and UV illumination
Quenching Mechanism
UV‑Vis absorption spectra (Additional file 1: Fig. S8) show no shift upon Fe3+ addition, indicating structural integrity. Fluorescence lifetime measurements (Fig. 6a) reveal a reduction in decay time, suggesting photo‑induced electron transfer from the NBD LUMO to Fe3+ d‑orbitals. The quenching is attributed to strong coordination between Fe3+ and surface –OH/–NH/–COOH groups, with Fe3+ acting as a Lewis acid that facilitates electron transfer (Fig. 6b). This selective interaction accounts for the pronounced sensitivity towards Fe3+ over other metal ions.
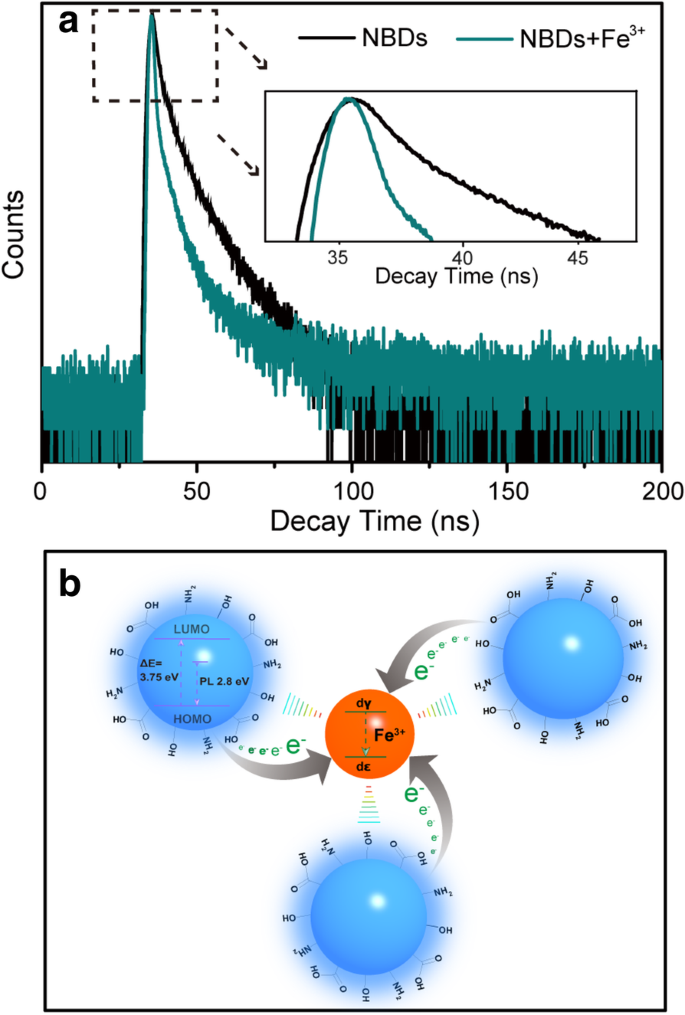
a Fluorescence decay traces of the NCDs in the absence and presence of Fe3+ under excitation at 370 nm and emission at 445 nm. b Schematic illustration for the possible fluorescence quenching mechanism of the NBDs in the presence of Fe3+ ions
Conclusion
We have established a green, heating‑free ultrasonic method to produce soybean‑derived NBDs with bright blue emission (16.7 % QY) and exceptional biocompatibility (100 % viability at 800 µg mL-1). Their fluorescence is selectively quenched by Fe3+, delivering a detection limit of 2.9 µM, well below regulatory thresholds. These findings position NBDs as promising probes for biomedical imaging and environmental monitoring.
Abbreviations
- FTIR:
Fourier‑transform infrared
- HAADF‑STEM:
High‑angle annular dark‑field scanning transmission electron microscopy
- LOD:
Limit of detection
- NBDs:
Nano‑biomass dots
- NMR:
Nuclear magnetic resonance
- PL:
Photoluminescence
- QDs:
Quantum dots
- QY:
Quantum yield
- TEM:
Transmission electron microscopy
- UES:
Ultrasonic extraction strategy
- USEPA:
United States Environmental Protection Agency
- XPS:
X‑ray photoelectron spectroscopy
- XRD:
X‑ray diffraction
Nanomaterials
- Fluorescent Nanomaterials: Advances, Synthesis, and Biomedical Applications
- Phosphine‑Free Synthesis of Reabsorption‑Suppressed ZnSe/CdS/ZnS Core–Shell Quantum Dots and Their Application in Sensitive CRP Detection
- Water‑Soluble Antimony Sulfide Quantum Dots: Efficient Synthesis and Superior Photo‑Electric Performance
- A Recyclable Magnetic‑Silica Hybrid Probe for Fe³⁺ Detection and Live‑Cell Imaging
- High-Sensitivity Graphene Oxide Fluorescent Aptasensor for Rapid Detection of CCRF-CEM Leukemia Cells
- High‑Quality In₂S₃ Quantum Dots Synthesized at Room Temperature for Ultra‑Sensitive UV Photodetectors
- Carbon Dots: Advanced Fluorescent Platforms for Sensitive Metal‑Ion Detection
- Laminarin-Based Nanoparticles Deliver Protoporphyrin IX for Enhanced Photodynamic Therapy: Cellular Uptake, ROS Generation, and In Vivo Efficacy
- Affordable Green Fluorescent Carbon Dots: Enhanced Bioimaging & Targeted Drug Delivery
- Advancing Lithium‑Ion Batteries: Binder‑Free Electrode Technology for Higher Energy Density



