Resveratrol-Loaded Solid Lipid Nanoparticles Enhance Insulin Sensitivity by Modulating SNARE Protein Expression in Adipose and Muscle Tissues of Type 2 Diabetic Rats
Abstract
In this study we formulated a resveratrol (RES)-loaded solid lipid nanoparticle (SLN‑RES) designed to improve insulin resistance by upregulating the SNARE protein complex in a rat model of type 2 diabetes. The optimized SLN‑RES displayed an average diameter of 248 nm, a zeta potential of –16.5 mV, and an entrapment efficiency of 79.9 %. In vitro release profiling revealed an initial burst followed by sustained release under physiological conditions. Infrared spectroscopy confirmed successful incorporation of RES into the lipid core, while transmission electron microscopy demonstrated spherical, non‑aggregated particles. Oral SLN‑RES administration prevented weight loss and achieved superior hypoglycemic effects compared with free RES. Serum oxidative‑stress markers returned to baseline levels following SLN‑RES treatment. Gene expression analysis showed a significant down‑regulation of synaptosomal‑associated protein 23 (Snap23), syntaxin‑4 (Stx4), and vesicle‑associated membrane protein 2 (Vamp2) in muscle tissue, with comparable effects in adipose tissue. Collectively, SLN‑RES represents a promising therapeutic platform for targeting insulin resistance via modulation of key SNARE proteins.
Presentation of the Hypothesis
Encapsulating RES within a lipid core nanoparticle enhances its physicochemical stability and intestinal absorption, leading to greater bioavailability. We hypothesize that SLN‑RES will reduce insulin resistance more effectively than free RES, owing to its antioxidant potency and improved delivery.
Testing the Hypothesis
Spectroscopic analyses were performed to confirm encapsulation efficiency. The second objective was to evaluate SLN‑RES effects on oxidative‑stress biomarkers and its hypoglycemic efficacy in an STZ‑induced type 2 diabetic rat model.
Implications of the Hypothesis
Improved stability and intestinal uptake increase oral RES bioavailability, leading to higher concentrations in target tissues and enhanced hypoglycemic action compared with free RES.
Background
Type 2 diabetes mellitus (T2DM) is characterized by systemic insulin resistance, largely driven by impaired GLUT 2 and GLUT 4 trafficking in muscle and adipose tissues. SNARE proteins—Snap23, Stx4, and Vamp2—anchor GLUT 4 vesicles to the plasma membrane, and their down‑regulation accelerates insulin resistance. Numerous plant‑derived compounds, including resveratrol (RES), have demonstrated metabolic benefits; however, oral RES suffers from poor absorption and rapid degradation. Nanomedicine approaches, particularly solid lipid nanoparticles (SLNs), offer a strategy to enhance RES stability and bioavailability. Prior studies indicate that RES encapsulation in SLNs improves neuroprotective outcomes and suggests potential for metabolic applications. Here, we develop and characterize RES‑loaded SLNs and assess their impact on glycemic control, oxidative stress, and SNARE protein expression in diabetic rats.
Materials and Methods
Materials
High‑purity trans‑resveratrol (>99 %) was sourced from Mega Resveratrol (USA). Streptozotocin (STZ) and nicotinamide (NA) were purchased from Sigma‑Aldrich (Germany). Hydrogenated soybean lecithin (S100) and hydrogenated palm oil (S154) were provided by Lipoid KG and Condea, respectively. TRIzol reagent and cDNA synthesis kit were obtained from Invitrogen (USA). Serum insulin ELISA kits were from Bio‑Equip (China). All other reagents were analytical grade.
Preparation of SLN‑RES
SLN‑RES was prepared via a modified hot homogenization‑ultrasonication method. Briefly, 40 mg S100 and 1 g sorbitol were dissolved in 15 ml distilled water and heated to 70 °C. Simultaneously, 100 mg S154, 70 mg S100, and RES (30–70 mg) were melted at 70 °C; 5 ml chloroform was added to create the organic phase. Rapid injection into the preheated aqueous phase under 1,000 rpm stirring removed chloroform. The suspension was sonicated (2 min) and then injected into 2 °C cold water under continuous stirring (1,000 rpm) for 2 h to solidify the lipid matrix. The resulting powder was washed twice by centrifugation, and free drug was quantified to calculate entrapment efficiency (EE). The effect of RES loading on particle size, PDI, zeta potential, and EE was evaluated (Table 1).
Characterization of SLN‑RES
Dynamic light scattering measured mean diameter, zeta potential, and PDI using a Malvern Zetasizer Nano‑ZS after a 1/30 dilution in water.
Entrapment Efficiency
EE (%) = [(total drug – free drug)/total drug] × 100. Free RES was quantified by UV‑Vis spectroscopy at 310 nm.
In Vitro Drug Release Study
SLN‑RES was dispersed in PBS (pH 7.4) and simulated gastric fluid (pH 1.2). At 0.5, 1, 2, 4, 6, and 8 h, aliquots were withdrawn, filtered (0.24 µm), and free RES measured.
Transmission Electron Microscopy
Particle morphology was examined on a ZEISS TEM grid, revealing spherical shapes and minimal aggregation.
Fourier Transform Infrared Spectroscopy
FTIR spectra (400–4,000 cm⁻¹) were obtained using a PerkinElmer instrument. Samples were mixed with KBr to form pellets.
Animal Study Design
Twenty male Wistar rats (200–250 g) were housed under standard conditions (25 ± 2 °C, 12‑h light/dark). T2DM was induced via a single intraperitoneal injection of STZ (65 mg/kg) and NA (110 mg/kg). Rats with fasting blood glucose >150 mg/dl were considered diabetic. After one week, rats received daily oral gavage of either 10 mg/kg RES or SLN‑RES (10 mg/kg RES content) for 4 weeks. Groups (n = 5) included healthy control (HC), diabetic control (DC), RES, and SLN‑RES. At study end, animals were weighed, anesthetized (ketamine/xylazine), and blood collected by cardiac puncture. Serum and tissues (skeletal muscle, visceral adipose) were harvested and stored at –80 °C for analysis.
Measurement of Fasting Blood Sugar
FBS was measured colorimetrically using a Pars Azmun kit.
Measurement of Serum Insulin
Serum insulin was quantified via a Bio‑Equip ELISA kit. HOMA‑IR was calculated using the standard formula.
Total Antioxidant Capacity
TAC was determined by the Fe³⁺/Fe²⁺ – TPTZ assay, measuring the blue complex absorbance.
Total Thiol Group
SH groups were measured with DTNB, producing a yellow chromophore.
Lipid Peroxidation Assay
MDA levels were assessed via the thiobarbituric acid reactive substances (TBARS) method using 1,1,3,3‑tetraethoxypropane as standard.
Total Oxidant Status
TOS was measured by the Fe³⁺/xylenol orange complex, calibrated with H₂O₂ and expressed as μM H₂O₂ eq/L.
Quantitative Reverse Transcription Polymerase Chain Reaction
mRNA from frozen tissues was extracted with TRIzol. cDNA synthesis used the RevertAid kit. qRT‑PCR quantified Snap23, Stx4, and Vamp2, normalizing to 18s rRNA. The 2⁻ΔΔCt method calculated relative expression. Experiments were triplicated.
Statistical Analysis
Data were expressed as mean ± SD. One‑way ANOVA (SPSS 16) assessed group differences; p < 0.05 was considered significant.
Results and Discussion
Physicochemical Characterization of SLN‑RES
Increasing the drug‑to‑lipid ratio did not significantly alter particle size or PDI, suggesting multilayer phospholipid formation. The mean size (~250 nm) is optimal for intestinal absorption while avoiding hepatic and splenic clearance. A PDI of 0.4 indicates a moderately heterogeneous population, yet zeta potential of –16.5 mV confers sufficient electrostatic stability. The 79.9 % EE confirms efficient encapsulation, and the SEM/TEM images confirm a smooth, spherical morphology with minimal aggregation.
In Vitro Release Profile
Release studies (Figure 1) showed ~70 % RES release after 6 h at pH 7.4 and after 1 h at pH 1.2, reflecting an initial burst followed by sustained release driven by drug–lipid interactions and diffusion gradients.
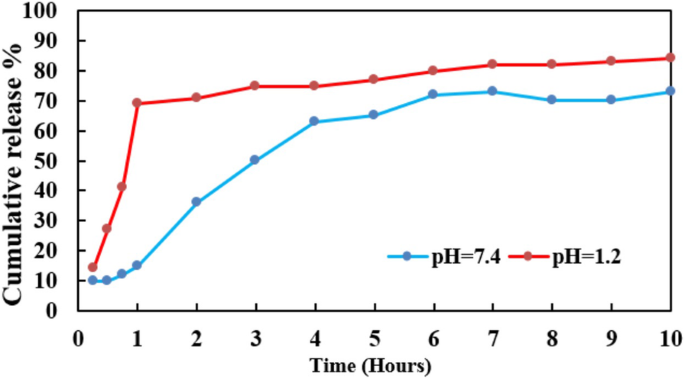
In vitro release profiles of RES from SLN‑RES in acidic and physiological conditions.
Morphology Evaluation
TEM (Figure 2) confirmed spherical particles averaging 20–30 nm, with occasional rod‑shaped forms but negligible aggregation, consistent with the high zeta potential and low PDI.

Transmission electron micrograph of SLN‑RES. Bar = 100 nm.
FTIR Validation of RES Loading
FTIR spectra (Figure 3) showed characteristic RES peaks (e.g., O‑H stretch at 3290 cm⁻¹, C=C stretch at 965 cm⁻¹) preserved in SLN‑RES, confirming successful encapsulation. Shifts in lecithin peaks indicated hydrogen‑bond formation and core loading.
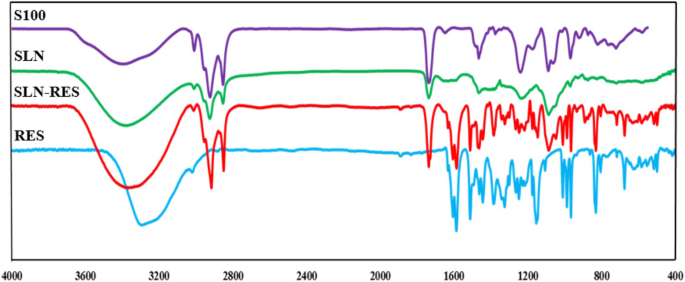
FTIR spectrum of SLN, SLN‑RES, S100, and pure RES.
Impact on Body Weight, Glycemia, and Insulin Sensitivity
Diabetic controls (DC) exhibited significant weight loss compared to healthy controls (HC). RES prevented weight loss, whereas SLN‑RES restored body weight to near HC levels (Table 2). Fasting blood glucose and HOMA‑IR improved in both RES and SLN‑RES groups, with SLN‑RES achieving values closer to HC. Serum insulin levels rose only in the SLN‑RES group, indicating enhanced insulin sensitivity.
Oxidative Stress Parameters
Diabetes markedly reduced TAC and SH levels while elevating MDA and TOS. SLN‑RES normalized TAC, MDA, and TOS, whereas free RES only partially restored TAC and failed to reduce MDA, underscoring the importance of improved bioavailability.
SNARE Gene Expression in Adipose Tissue
Both RES and SLN‑RES up‑regulated Snap23, Stx4, and Vamp2 in adipose tissue relative to DC, but the magnitude of change did not differ significantly between formulations, suggesting a plateau in adipose tissue exposure.
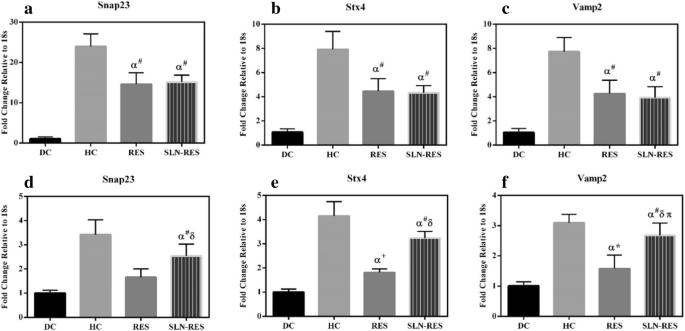
The effect of RES and SLN‑RES on Snap23, Stx4, and Vamp2 mRNA in adipose (a–c) and muscle (d–f) tissues. *p < 0.05, +p < 0.01, #p < 0.001.
SNARE Gene Expression in Muscle Tissue
STZ reduced Snap23, Stx4, and Vamp2 expression in skeletal muscle. SLN‑RES restored these genes more effectively than free RES, particularly Vamp2, indicating superior delivery to muscle tissue.
Conclusion
We successfully formulated a stable, bioavailable RES‑loaded SLN suitable for oral administration. SLN‑RES enhanced glycemic control, mitigated oxidative stress, and selectively up‑regulated SNARE proteins in muscle and adipose tissues of diabetic rats, thereby improving insulin sensitivity. Future studies should explore pharmacokinetics and tissue distribution to further validate this therapeutic strategy.
Availability of Data and Materials
All data generated or analyzed during this study are presented in this article.
Abbreviations
- RES:
Resveratrol
- SLN:
Solid lipid nanoparticle
- SLN‑RES:
Resveratrol‑loaded solid lipid nanoparticle
- SNAP‑23:
Synaptosomal‑associated protein 23
- STX4:
Syntaxin‑4
- VAMP‑2:
Vesicle‑associated membrane protein 2
- TEM:
Transmission electron microscopy
- FTIR:
Fourier‑transform infrared spectroscopy
- -SH:
Total thiol group
- TAC:
Total antioxidant capacity
- MDA:
Malondialdehyde
- TOS:
Total oxidant status
Nanomaterials
- C# Expressions, Statements & Blocks: A Comprehensive Guide with Practical Examples
- Understanding type() and isinstance() in Python: Practical Examples
- Resveratrol-Loaded Human Serum Albumin Nanoparticles with PEG–RGD Targeting: Extended Circulation, Enhanced Biocompatibility, and Potent Pancreatic Tumor Suppression
- In Vivo Toxicity Assessment of Biosynthesized Copper and Zinc Oxide Nanoparticles Administered via Intraperitoneal and Intravenous Routes in Rats
- Lowering Metal–Ge Contact Resistance with ZnO Interlayers and Argon Plasma: Achieving Ohmic Al Contacts on n‑Ge
- PdO‑CeO₂ Rod‑Like Nanoporous Catalysts with Superior CO Oxidation and Methane Combustion Performance
- Resveratrol-Loaded Solid Lipid Nanoparticles Enhance Insulin Sensitivity by Modulating SNARE Protein Expression in Adipose and Muscle Tissues of Type 2 Diabetic Rats
- Targeted Silica Nanoparticles Co‑Deliver miR‑495 and Doxorubicin to Overcome Multidrug Resistance in Lung Cancer
- SnxPy Monolayers: Promising 2D Materials with High Stability, Exceptional Carrier Mobility, and Ferromagnetic Behavior
- HDAC1 Regulates miR‑124‑5p and NPY to Modulate Learning and Memory in Depressive Rat Models



