Solvent‑Free Benzyl Alcohol Oxidation on Au–Pd/TiO₂: Comparative Activity of Rutile, Brookite, and Anatase Supports
Abstract
Supported Au–Pd bimetal nanoparticles on TiO₂ (P25) have demonstrated remarkable performance in solvent‑free benzyl alcohol oxidation. Yet, the influence of the TiO₂ crystal form on catalytic activity remains largely unexplored. Here, we successfully synthesized rutile, brookite, and anatase TiO₂ and loaded them with Au–Pd (1:1 molar ratio) nanoparticles via deposition‑precipitation. Experiments reveal that the Au–Pd/rutile catalyst delivers the highest benzyl alcohol conversion, while exhibiting the lowest benzaldehyde selectivity and the highest toluene yield among the three supports. ICP‑AES, XRD, XPS, and TEM characterizations attribute this superior performance to a narrower Au–Pd size distribution and increased surface concentrations of O_α and Pd^2+ species. Recycling studies show that Au–Pd/rutile suffers greater deactivation than its anatase and brookite counterparts, likely due to surface coverage by aldehyde intermediates. These findings highlight the critical role of TiO₂ crystal structure in shaping Au–Pd catalyst behavior for solvent‑free alcohol oxidation.
Introduction
Gold was long considered chemically inert until the pioneering work of Hutchings and Haruta revealed its catalytic prowess in acetylene hydrochlorination and low‑temperature CO oxidation, respectively [1, 2]. Since then, Au has been applied to a broad range of reactions—water‑gas shift, H₂O₂ synthesis, selective hydrogenation of cinnamaldehyde, and more—often in synergy with Pd, which enhances both reactivity and stability [3–5]. Hutchings et al. demonstrated that Au–Pd bimetal nanoparticles supported on TiO₂ (P25) outperform monometallic counterparts for solvent‑free benzyl alcohol oxidation [6].
For supported Au–Pd catalysts, the support’s physicochemical properties, nanoparticle size, and synthesis route critically determine performance. Reducible metal oxides such as TiO₂, CeO₂, and Fe₂O₃ are favored due to strong metal–support interactions and efficient O₂ activation. TiO₂, in particular, has been extensively studied for Au–Pd loading, consistently yielding high catalytic activity for benzyl alcohol oxidation. Various preparation strategies—sol‑immobilization, colloidal nanotube synthesis, photo‑deposition, and bio‑reduction—have all produced highly active, durable Au–Pd/TiO₂ catalysts [6–9].
While P25 is the most common TiO₂ form, it contains three polymorphs: rutile, brookite, and anatase. Most literature focuses on the photocatalytic or gas‑phase oxidation behavior of these phases, with anatase generally deemed superior for catalytic activity. However, recent studies have challenged this view, showing that brookite can support higher activity in CO oxidation and that CeO₂/rutile offers superior SCR performance [10–12]. To date, the effect of TiO₂ polymorph on solvent‑free benzyl alcohol oxidation over Au–Pd remains uninvestigated.
Addressing this gap, we systematically compare Au–Pd catalysts supported on rutile, brookite, and anatase TiO₂ under identical reaction conditions. Comprehensive XRD, ICP‑AES, XPS, and TEM analyses elucidate how TiO₂ form influences metal dispersion, oxidation states, and surface oxygen species, thereby shaping catalytic outcomes.
Methods
All reagents were analytical grade and used as received (Aladdin Company, Shanghai, China). Key chemicals included urea (99.9%), titanium bis(ammonium lactate) dihydroxide (TBD, 50 % aqueous), TiCl₄ (99.99 %), PdCl₂ (99.99 %), HAuCl₄·3H₂O (≥ 99.9 %), benzyl alcohol (99.8 %), and O₂ (99.999 %).
Synthesis of Brookite and Anatase TiO₂
Brookite TiO₂ was prepared by mixing 8 mL 50 % TBD with 17 g urea, diluting to 80 mL with deionized water, and hydrothermal treatment at 160 °C for 24 h in a 200 mL Teflon‑lined autoclave. The precipitate was filtered, washed, dried, and calcined at 500 °C for 5 h. Anatase TiO₂ was synthesized similarly, adjusting the urea amount to 0.48 g.
Synthesis of Rutile TiO₂
TiCl₄ (dissolved in ethanol) was slowly mixed with water under stirring (TiCl₄:ethanol:water = 2:20:280). The resulting sol was stirred 3 h, aged 24 h at 50 °C in a sealed autoclave, then centrifuged, washed, dried, and calcined at 500 °C for 5 h.
Preparation of Au‑Pd/TiO₂
Au:Pd = 1:1 (nominal loadings 1.00 wt % Au, 0.54 wt % Pd). For Au–Pd/brookite, 2 mL 5 mg mL⁻¹ HAuCl₄, 1.08 mL 5 mg mL⁻¹ PdCl₂, 0.985 g brookite TiO₂, and 3.48 g urea were stirred in 100 mL deionized water, heated to 80 °C for 6 h, aged 12 h, then centrifuged, washed, dried, and calcined at 300 °C for 2 h (2 °C min⁻¹). Analogous procedures were applied for anatase and rutile supports. Samples were labeled TiO₂‑B (brookite), TiO₂‑R (rutile), TiO₂‑A (anatase), ATB, ATR, and ATA respectively.
Benzyl Alcohol Oxidation
Reactions were conducted in a 50 mL glass‑lined reactor (Anhui Kemi Machinery). 15 mL benzyl alcohol and 0.05 g catalyst were sealed, purged with O₂ five times, then pressurized to 0.3 MPa O₂ at room temperature. The mixture was heated to the target temperature (120 °C) at 1000 rpm. O₂ reservoir ensured constant oxygen supply. Products were analyzed by GC (FuLi GC9790) with FID and a DM‑5 column (30 m × 0.25 mm × 0.25 µm). Each experiment was performed in triplicate and repeated at least twice. Catalyst stability was tested over three consecutive runs; after each run the catalyst was recovered, washed with acetone, and dried at 80 °C for 16 h.
Characterization
Powder XRD: Rigaku D/max‑RC (Cu Kα, 40 kV, 25 mA, 10–90° 2θ, 8 ° min⁻¹). ICP‑AES: Agilent 735‑ES (catalyst dissolved in aqua regia for 24 h). XPS: PHI‑1600ESCA (Mg‑Kα, 15 kV, 10⁻⁷ Pa, reference C 1s = 284.6 eV). TEM: JEM‑2100 (200 kV, sample dispersed in ethanol, sonicated, deposited on carbon‑film grids).
Results and Discussion
XRD confirmed the successful synthesis of anatase, brookite, and rutile TiO₂ (see Fig. 1). Scherrer analysis yielded crystallite sizes of 11.2 nm (anatase), 18.9 nm (brookite), and 27.6 nm (rutile). Loading Au–Pd nanoparticles did not produce new diffraction peaks, indicating high dispersion (≤ 5 nm). ICP‑AES revealed slightly lower bulk metal loadings than nominal values, likely due to leaching during washing.
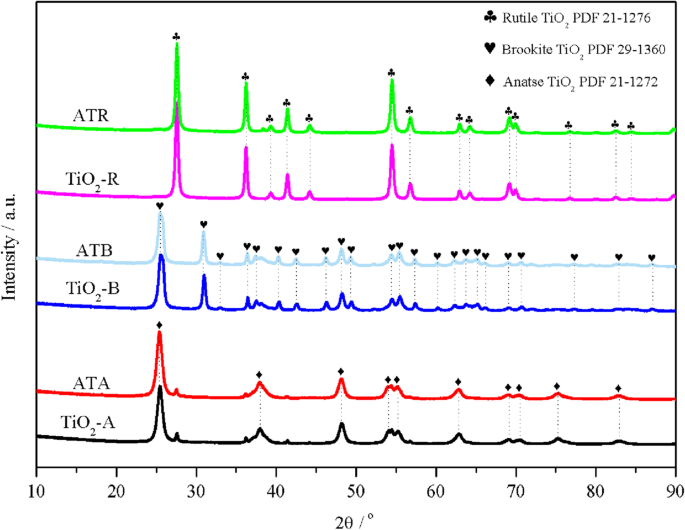
XRD patterns of TiO₂‑A, ATA, TiO₂‑B, ATB, TiO₂‑R, and ATR samples
XPS analysis showed metallic Au (Au 4f₇/₂ = 84.0 eV) with slight negative shifts, confirming electronic interaction with Pd. Pd existed as both Pd⁰ (Pd 3d₃/₂ ≈ 335 eV) and Pd²⁺ (≈ 337 eV). The Pd²⁺ fraction followed ATR > ATB > ATA (55.4 %, 48.2 %, 34.8 %). O1s spectra revealed lattice O_β (529.1 eV) and surface O_α (531.0 eV). The O_α/O_β ratio was highest for ATR (43.8 %) and lowest for ATA (20.2 %). These surface oxygen species are key to O₂ activation and metal stabilization.
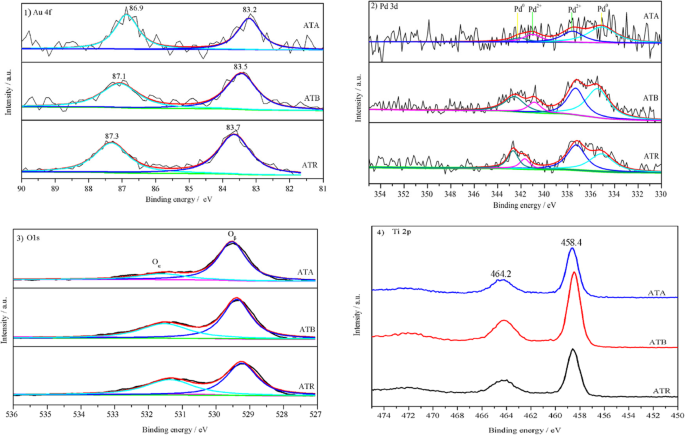
XPS spectra of (1) Au 4f, (2) Pd 3d, (3) O1s, and (4) Ti 2p for ATA, ATB, and ATR samples
TEM images (Fig. 3) indicated mean Au–Pd particle sizes of 4.6 nm (ATA), 4.3 nm (ATB), and 4.1 nm (ATR). The smallest particles on ATR correlate with its higher O_α and Pd²⁺ content, explaining its superior activity.
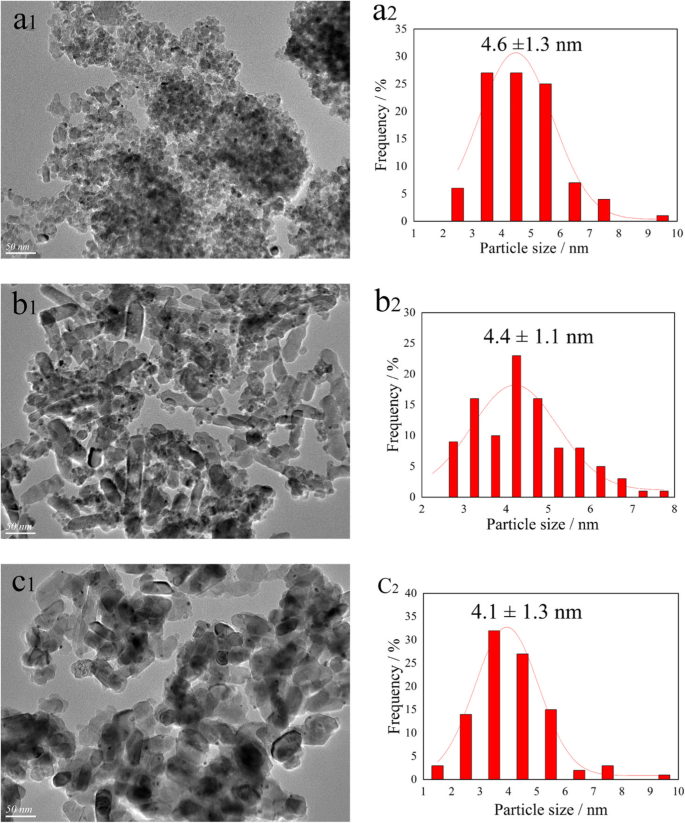
TEM images and Au–Pd particle size distributions for ATA (a‑1, a‑2), ATB (b‑1, b‑2), and ATR (c‑1, c‑2) catalysts
Under solvent‑free conditions (120 °C, 0.3 MPa O₂), benzyl alcohol conversion after 3 h was 65.1 % (ATR), 60.0 % (ATB), and 51.8 % (ATA). Conversion followed the order ATR > ATB > ATA, mirroring the O_α and Pd²⁺ trends. Product distribution (Fig. 5) showed that ATR favored toluene (higher selectivity) at the expense of benzaldehyde, whereas ATA and ATB maintained higher benzaldehyde yields.
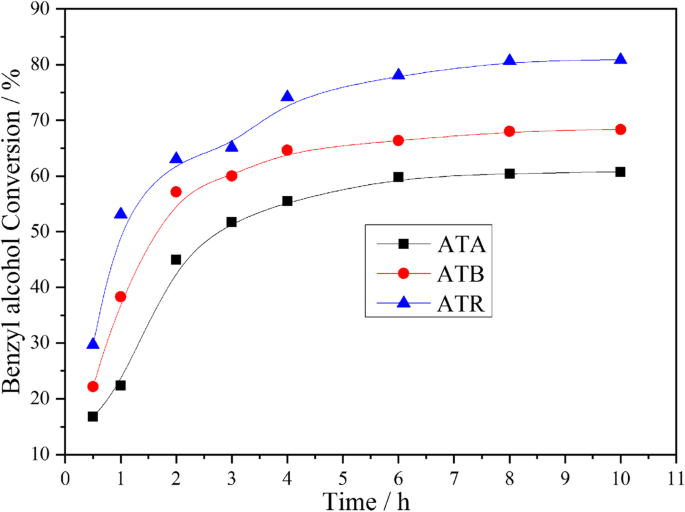
Benzyl alcohol conversion vs. time for ATA, ATB, and ATR
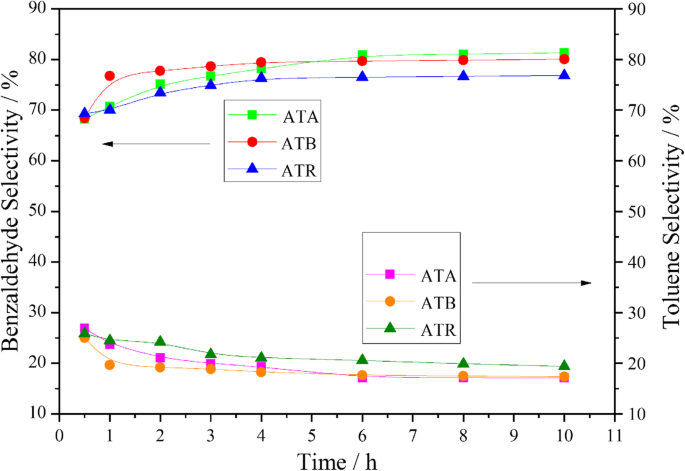
Benzaldehyde and toluene selectivities over time for ATA, ATB, and ATR
Recycling tests (Fig. 6) revealed that ATA and ATB retained > 59 % conversion over three cycles, whereas ATR dropped from 65.1 % to 59.2 %. The decline is attributed to Pd poisoning by accumulated aldehyde intermediates; ATR’s higher surface Pd content (0.65 at. %) likely exacerbates this effect.
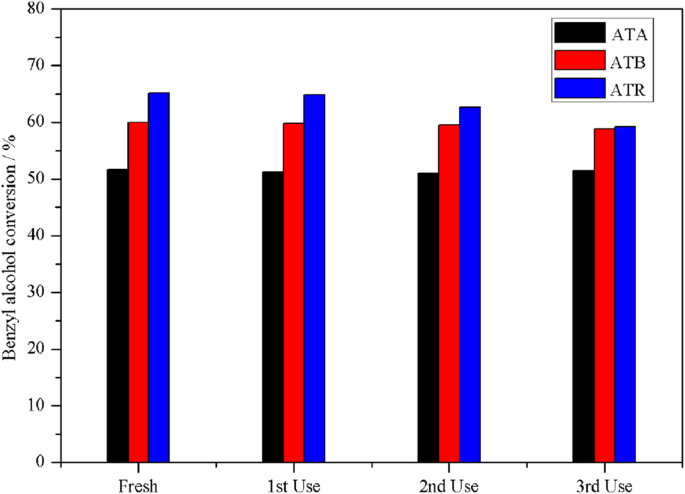
Reuse performance of ATA, ATB, and ATR in solvent‑free benzyl alcohol oxidation
Conclusion
Au–Pd (1:1) nanoparticles deposited on TiO₂ polymorphs via deposition‑precipitation exhibit activity that is strongly dependent on the support phase. ATR (rutile) delivers the highest benzyl alcohol conversion due to its high surface O_α, Pd²⁺ content, and smallest metal particle size. However, its catalytic stability is limited by aldehyde adsorption on the Pd sites. In contrast, ATA and ATB maintain superior durability while offering higher benzaldehyde selectivity. These insights emphasize the importance of TiO₂ crystal structure in tailoring Au–Pd catalysts for solvent‑free alcohol oxidation.
Nanomaterials
- Rapid Electrospun SrTiO₃‑Modified Rutile TiO₂ Nanofibers: Enhanced UV‑Driven Photocatalytic Degradation of Methyl Orange
- TiO₂ Nanofluids: Applications, Challenges, and Future Directions – Part 2
- Optimizing Gold Nanoparticle Placement in TiO₂ Enhances Dye‑Sensitized Solar Cell Efficiency
- Chromium‑Doped Titanium Dioxide: A Novel Colored Cool Pigment with High Near‑Infrared Reflectance
- Optimizing TiO₂ Nanotube Arrays via Soft–Hard Template for Superior Field Emission Performance
- Transition Metal‑Doped TiO₂ Nanoparticles: Surface‑Spectroscopic Insight into Catalytic Activity
- Influence of Peptizing Acid on TiO₂ Phase Composition and Photocatalytic Efficiency: A Comparative Study of Sulfuric, Nitric, and Acetic Acids
- Enhanced Up‑Conversion TiO₂ Nanomaterial Boosts Perovskite Solar Cell Efficiency to 16.3 %
- Enhanced Visible‑Light Photocatalysis and Self‑Cleaning via CuS‑Decorated TiO₂/PVDF Nanofibers
- Enhanced TiO₂ Catalysis and Antimicrobial Efficacy via Nitrogen and Carbon Nitride Co‑Doping



