External Defibrillator: History, Design, and Manufacturing
External defibrillators deliver a precisely timed electric shock through the chest wall to restore a normal cardiac rhythm in patients experiencing life‑threatening arrhythmias such as ventricular fibrillation.
Background
A defibrillator kit typically includes a power control unit, paddle electrodes, and accessories assembled through an integrated production process. Over the decades, both external and internal models have markedly increased patient survival.
The human heart contains four chambers that function as two synchronized pumps. The right pump moves oxygen‑depleted blood to the lungs, while the left pump distributes oxygenated blood to the body. Each pump comprises an atrium and a ventricle; coordinated contraction and relaxation maintain effective circulation.
The sinoatrial node, located in the right atrium, initiates spontaneous electrical impulses via ion diffusion. These impulses travel to the atria, causing them to contract, then advance to the ventricles approximately 150 ms later, triggering ventricular contraction and subsequent relaxation. Any disruption in this sequence can lead to arrhythmias such as ventricular fibrillation.
Ventricular fibrillation, often triggered by coronary artery blockage, anesthetic reactions, or electrical shock, results in a chaotic, ineffective heartbeat. A defibrillator delivers a high‑energy pulse—typically 3 to 9 ms—over the chest. The shock resets the ventricular rhythm, allowing the heart to resume normal activity.
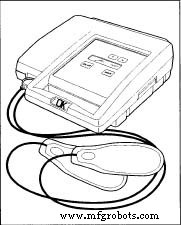
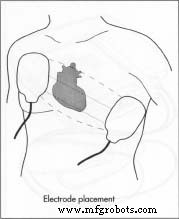
In practice, an external defibrillator can be operated in emergency sites or hospitals. The operator powers the unit, applies conductive gel to the paddle electrodes, selects the energy level, charges the device, and places the paddles on the unclothed chest with about 25 lb (11 kg) of pressure. Simultaneous activation of the charge and discharge buttons delivers the shock, after which the patient is monitored and the process repeated if necessary.
History
The concept that an electrical charge could restart a misfiring heart dates back to 1888 when Mac William proposed that ventricular fibrillation might underlie sudden death. In 1899, Prevost and Batelli demonstrated that large voltages could terminate fibrillation in animals.
Throughout the 1920s and 1930s, research—often funded by power companies—investigated electrical shock as a treatment for line‑worker fatalities. Hooker, William B. Kouwenhoven, and Orthello Langworthy published early experiments in 1933 showing that an internally applied alternating current could reverse ventricular fibrillation in dogs.
In 1947, Dr. Claude Beck performed the first successful human defibrillation during surgery, applying a 60 Hz alternating current to stabilize a patient’s heart. The following decade saw further breakthroughs: in 1954, Kouwenhoven and William Milnor achieved closed‑chest defibrillation in dogs, and in 1956, Paul Zoll executed the first successful external defibrillation of a human.
William B. Kouwenhoven (1886–1975) was a pivotal figure in this field. Trained as an electrical engineer, he invented three defibrillators—including the open‑chest, Hopkins AC, and Mine Safety Portable models—and pioneered CPR techniques. His innovations earned him multiple awards, including the American Medical Association’s Ludwig Hektoen Gold Medal (1961, 1972), the IEEE Edison Medal (1962), an honorary M.D. from Johns Hopkins (1969), and the Lasker Clinical Research Award (1973).
The 1960s ushered in direct‑current defibrillators, which offered fewer side effects than alternating‑current devices. In 1967, Pantridge and Geddes demonstrated the life‑saving potential of mobile, battery‑powered DC defibrillators. Dr. Michael Mirowski introduced the first implantable defibrillator in the late 1960s, and the 1970s saw automated detection of ventricular fibrillation in both internal and external models.
Today, defibrillation is a cornerstone of emergency response. The American Heart Association designates it as a basic life‑support skill for paramedics and rescue workers.
Raw Materials
All components of a defibrillator must be biocompatible, pharmacologically inert, sterilizable, and durable across varying environmental conditions. The control box casing is typically made of high‑grade polystyrene or lightweight metal alloy. Electrodes are fabricated from titanium and silicone rubber, while microelectronics employ modified silicon semiconductors. Battery chemistries include lead‑acid, nickel‑cadmium, zinc, lithium, sulfur dioxide, and manganese dioxide.
Design
The core architecture of an external defibrillator comprises a control box, power source, electrodes, cables, and connectors. This overview focuses on portable units used in clinical and field settings.
Controls
The control box houses high‑voltage generation circuits and a capacitor bank capable of storing up to 7 kV, delivering 30–400 J of energy. Key controls include power, energy selection, charge, and discharge buttons. Some models offer dedicated controls for internal paddles or disposable electrodes.
Electrodes
Electrodes transmit the shock to the heart. Options include hand‑held paddles, internal paddles, and self‑adhesive, pre‑gelled disposable electrodes. Disposable pads are preferred in emergencies for speed and technique reliability. Paddle size influences current density: larger paddles reduce resistance and enhance energy delivery. Adult paddles range from 3.1–5.1 in (8–13 cm); pediatric paddles are proportionally smaller.
Because skin is a poor conductor, conductive gel—typically containing lanolin, petrolatum, and chloride ions—is applied to bridge the electrode and skin, improving charge transfer and minimizing burn risk.
Battery
Defibrillators employ various battery chemistries, notably lead‑acid, lithium, and nickel‑cadmium. Batteries are usually rechargeable and stored plugged in when not in use. Extreme temperatures degrade battery performance, so units are maintained in controlled environments. Over time, battery wear necessitates replacement due to corrosive and potentially toxic chemistries.
Automated External Defibrillators
Since 1978, AEDs have incorporated sensors that detect ventricular fibrillation and provide voice prompts for shock delivery. These devices dramatically reduce the training required and have saved thousands of lives worldwide.
The Manufacturing Process
Defibrillators are complex electronic systems assembled from externally sourced components. The process is highly integrated, involving several specialized stages.
Battery Production
- Lithium batteries used in defibrillators combine lithium metal with sulfur dioxide gas in an oxygen‑free environment. The lithium is formed into a solid case, sealed hermetically, and the sulfur dioxide is sealed inside.
- Typically, four lithium cells are wired in series, each fitted with an 8‑amp fuse and vent for pressure relief.
Casing Fabrication
- Injection molding creates the outer housing and electrode casings. Plastic pellets are melted, heated by a hydraulically‑controlled screw, and injected into a two‑half metal mold. After cooling, the part is ejected onto a conveyor for inspection.
- Image:
Electronics Assembly
- The motherboard integrates semiconductor chips, resistors, capacitors, and other components. Using hybridization, a printed circuit board is populated by a computer‑controlled placement head.
- Components are soldered, cooled, tested, and then mounted into the control box.
Assembly
- Electronic boards are attached to the casing in clean conditions. Screws and fasteners secure the boards; electrode adapters are mounted. The assembly is then sealed and forwarded for testing and packaging.
- Image:
Paddle Electrode Fabrication
- The metal plate, typically tin, is produced by continuous casting and cut to size. A stannous chloride layer is applied for surface finish.
- The cable is drawn from heated metal to produce a fine wire, then bundled, insulated, and connected to the plate.
- Finally, the plate is fitted into the outer shell, the cable routed through a rear aperture, and an adapter is attached for connection to the control box.
Final Assembly
- All components—control box, electrodes, battery, cables, and manuals—are placed in a cushioned container for shipment.
- Before shipping, the device undergoes functional testing: battery charging, discharge, and output measurement under varying environmental conditions.
Quality Control
Quality is ensured through visual and electrical inspections at every production stage. Electronics are fabricated in clean rooms with lint‑free garments to mitigate contamination. Batteries undergo rigorous performance, safety, and stability tests. Post‑purchase, routine maintenance checks (every 3–6 months) include charge‑discharge cycles to confirm continued reliability.
All manufacturers must register with the U.S. Food and Drug Administration and comply with Good Manufacturing Practices, involving extensive record‑keeping and facility inspections.
The Future
Future defibrillators aim to enhance safety, efficiency, and usability. Innovations include Y‑shaped cable electrodes to reduce operator shock risk, advanced integrated circuits for lightweight design, and smarter sensors that provide real‑time patient data.
Battery technology is also advancing; researchers at Brookhaven National Laboratory have patented a metal alloy that can significantly increase nickel/metal‑hydride battery capacity, promising longer life and higher charge retention.
Where to Learn More
Books
Carr, J. J. Introduction to Biomedical Equipment Technology. 2nd ed. Prentice Hall Career and Technology, 1993.
Fox, Stuart. Human Physiology. W. C. B. Publishers, 1990.
Oever, R. V. D. Cardiac Pacing and Electrophysiology: A Bridge to the 21st Century. Kluwer Academic Publishers, 1994.
Periodicals
Shakespeare, C. F., and A. J. Camin. “Electrophysiology, Pacing, and Arrhythmia.” Clinical Cardiology 15 (1992): 601‑606.
Other
Worthington, Janet Farrar. “The Engineer Who Could.” Hopkins Medical News. 18 March 1998. 2 October 2001. https://www.hopkinsmedicine.org/hmn.
Perry Romanowski
Manufacturing process
- Artificial Heart: Technology, History, and Future Prospects
- Artificial Heart Valves: Types, History, and Future Innovations
- Comprehensive Overview of Modern ECG Machines: Technology, Manufacturing, and Future Innovations
- Pacemaker: Modern Life‑Saving Technology for Heart Rhythm Disorders
- Remote Heart Rate Monitoring System with AWS IoT Alerts
- ASMD Shock Rifle – DIY Arduino Nerf Launcher
- Industrial Shock Absorbers: Types, Applications, and Selection Guide
- Understanding Spring Shock Absorbers: Function, Design, and Benefits
- Understanding Shock Absorber Struts: Function, Placement, and Maintenance
- How Shock Absorbers Work: Enhancing Vehicle Balance and Comfort



