Understanding Toy Slime: Chemistry, History, and Production
Background
Toy slime is a fascinating example of a cross‑linked polymer that behaves as a non‑Newtonian fluid. It is typically created by mixing a polyvinyl alcohol (PVA) solution with a borate ion source, most often sodium borate (Borax), in a large mixing vessel. The resulting mass is usually greenish, cold to the touch, and has an unmistakable slimy feel.
In physics, a non‑Newtonian fluid has a viscosity that changes with the applied shear rate. Slime appears liquid when stirred gently but resists motion when struck or pressed, demonstrating shear‑thickening behavior. Other everyday examples include ketchup, gelatin, glue, and quicksand.
At the molecular level, the behavior stems from long, tangled polymer chains that can slide past each other when little force is applied but become locked together by transient ionic cross‑links when a higher force is imposed.
Cross‑linking agents are critical for giving slime its unique feel. The borate ions form weak, reversible bonds with the hydroxyl groups of PVA, holding the polymer network together without turning the mixture into a solid.
History
The modern understanding of slime’s chemistry traces back to the early 20th century, when Nobel laureate Hermann Staudinger introduced the chain‑like polymer model that would underpin synthetic polymer science. By the 1930s, the field had matured, and manufacturers began producing polymer‑based play materials.
Early toys such as modeling clay were soon followed by the 1950s’ “silly putty,” and in the 1980s a wave of slime‑type toys appeared, made from polyvinyl alcohol, guar gum, or even fortified milk.
Design & Safety
Most commercial slimes come in small tubs or sachets and are sold either as standalone products or as part of themed sets. Common colors are green, blue, and red; manufacturers sometimes add fragrances to mask the natural odor.
Safety is paramount. All ingredients must be non‑irritating and non‑toxic, and the finished product must not damage clothing or upholstery. Regulatory guidelines and independent lab testing are used to confirm compliance.
Raw Materials
Production begins in a laboratory where chemists define the desired aesthetic attributes—consistency, color, and scent—often guided by consumer testing. Small test batches are then made using the primary raw materials.
- Water: typically de‑ionized and constituting >90% of the formulation.
- Polymeric binder: usually 2% PVA by weight; alternatives include PVAC, guar gum, methylcellulose, or cornstarch.
- Cross‑linker: ~2% sodium borate, which forms reversible ionic bonds with the polymer chains.
- Colorants: food‑grade dyes such as FD&C Blue or FD&C Yellow.
- Fragrance & preservatives: volatile oils for scent and agents like formaldehyde or methylparaben to inhibit microbial growth.
- pH adjusters: acids or bases to maintain a stable pH.
Manufacturing Process
Production proceeds in two main stages: compounding and packaging. In many cases the cross‑linker is added just before filling to simplify the filling line.
Compounding
- Large stainless‑steel tanks equipped with mixers and temperature controls hold the base water and gradually introduce the polymer, colorants, preservatives, and, finally, the borate solution.
- Computer‑controlled mixers maintain the optimal shear rate and temperature, ensuring a homogeneous product.
- After mixing, the batch is sampled for quality control.
Slime is made by combining a 5:1 mixture of polyvinyl alcohol (PVA) solution and a Borax solution.
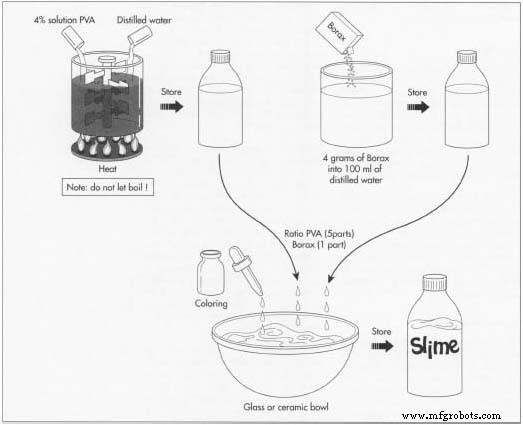
Quality Control
- Each sample undergoes pH, viscosity, appearance, and odor tests. Adjustments can be made before the batch is approved.
- Approved batches are stored in a holding tank until filling.
Filling & Packaging
- For tub products, a hopper aligns empty tubs and feeds them onto a conveyor that brings them beneath a carousel of filling heads.
- Product is injected into each tub, then capped by an automated capping machine.
- Optional labeling and shrink‑wrapping steps precede palletisation and shipping.
Quality Assurance
Quality assurance spans the entire production cycle. Incoming raw materials are inspected for particle size, pH, viscosity, appearance, and odor. Batches that fail to meet specifications are rejected or corrected. Stability studies expose finished products to varied environmental conditions to ensure long‑term safety and performance.
The Future
While current slime formulations are popular, there is ongoing research into new textures, eco‑friendly polymers, and customizable kits that allow consumers to create their own slime at home. Future developments will likely focus on enhancing safety, expanding sensory features, and creating sustainable materials.
Manufacturing process
- Titanium in Building Materials: Innovative Applications & Global Use
- Titanium’s Role in Energy Materials: From Batteries to Geothermal Power
- Magnets: Types, Materials, Manufacturing, and Future Applications
- Blast Furnace Stock House: Managing Raw Materials for Hot Metal Production
- Choosing the Right Cutting Tool Materials for Optimal Machining Performance
- Mastering Polyurethane Bonding: Techniques for Strong, Reliable Adhesion
- Your Comprehensive Guide to Copper Alloys: Types, Uses, and Benefits
- Eco-Friendly Biodegradable Materials for 3D Printing: Sustainable Solutions
- Exploring Material Flexibility: How Elasticity Determines Performance
- Understanding Brittleness in Materials: Causes, Consequences, and Testing



