Copper: History, Production, and Sustainable Future
Background
Copper (Cu) is a naturally reddish‑orange metal prized for its exceptional thermal and electrical conductivity. It forms the backbone of countless everyday items—from electrical wiring and cookware to automotive radiators and protective coatings. Historically, copper’s versatility extended to pigments, paper preservatives, and textile dyes. Alloying copper with zinc produces brass, while tin yields bronze.
Archaeological evidence shows copper use dates back ~10,000 years. A copper pendant dated to 8700 B.C. was unearthed in northern Iraq, and by 6400 B.C., the Anatolian region was melting and casting copper. Egyptian practices followed by 4500 B.C., and systematic mining is documented in the Sinai Peninsula around 3800 B.C. In 3000 B.C., Cyprus revealed rich copper deposits; the Romans named the metal aes cyprium, a term that evolved into “cuprum” and the modern symbol Cu.
In the Americas, Peruvian coastal communities produced copper artifacts by 500 B.C., and Inca metallurgy flourished until the Spanish conquest in the 16th century.
In the United States, the first commercial mine opened in Branby, Connecticut (1705), followed by Lancaster, Pennsylvania (1732). Despite early mining, U.S. copper largely came from Chile until 1844, when the Superior Copper District near Lake Superior began domestic production. Late‑19th‑century advances enabled extraction from lower‑grade ores via expansive open‑pit operations across the West.
Today, the United States and Chile dominate global output, followed by Russia, Canada, and China.
Raw Materials
Pure copper is rarely found in nature; it is extracted from a variety of ores. Around 40 countries mine roughly 15 commercial copper ores, most of which are sulfide minerals bonded with sulfur. Oxide, carbonate, and mixed ores also occur, and many contain valuable by‑products such as gold, silver, and nickel. Typical U.S. copper ores contain 1.2–1.6 % copper by weight.
The principal sulfide ore is chalcopyrite (CuFeS₂), known as copper pyrite or yellow copper ore. Chalcocite (Cu₂S) is another common sulfide. Oxide ores include cuprite (Cu₂O, red copper) and carbonate ores such as malachite (Cu(OH)₂·CuCO₃) and azurite (Cu(OH)₂·2CuCO₃). Other minerals—tennantite, boronite, chrysocolla, and atacamite—also supply copper.
Processing and refining typically employ sulfuric acid, oxygen, iron, silica, and organic reagents, depending on the chosen method.
The Manufacturing Process
Copper extraction varies by ore type and desired purity. The process systematically removes non‑copper materials and concentrates copper through physical and chemical steps, conducted both on‑site and at dedicated facilities.
Mining
- Large open‑pit mines use drilling and blasting. Overburden removal exposes ore deposits, creating pits that can exceed a mile across. Roads cut into the pit allow access for heavy machinery.
- Power shovels, each capable of 500–900 cubic feet (15–25 m³) per scoop, load ore into haul trucks that transport it out of the pit.
Concentrating
Raw ore contains dirt, clay, and non‑copper minerals. The first step is to reduce this waste via flotation.
- The ore is crushed through a series of cone crushers, which compress it between rotating inner and outer cones.
- Crushed material is further ground in rod mills and two ball mills, producing a slurry with particles around 0.01 in (0.25 mm).
- Reagents coat copper particles; a frother (pine oil or long‑chain alcohol) creates bubbles. Air is injected from the bottom of flotation cells, causing copper to attach to bubbles that rise to the surface. The froth is skimmed, water is drained, and the resulting concentrate contains 25–35 % copper, with traces of gold, silver, and other metals. Tailings—gangue—are deposited in settling ponds.
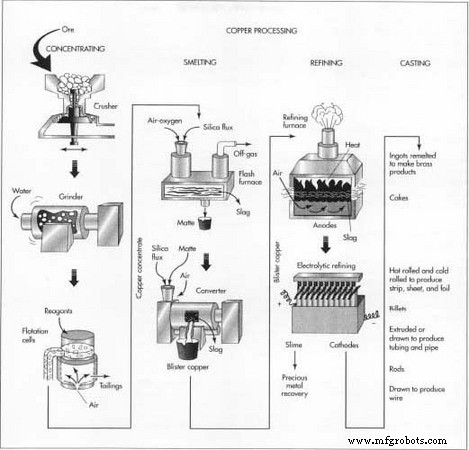
Smelting
After physical cleaning, the concentrate undergoes smelting to eliminate iron and sulfur. Traditional smelters use two furnaces; modern plants may combine stages into a single unit.
- In the flash furnace, the concentrate and silica flux are heated with oxygen‑enriched air. Iron combines with flux to form slag, which is skimmed off. Sulfur forms sulfur dioxide, vented and later converted to sulfuric acid. The remaining molten matte—about 60 % copper—flows to the converter.
- The converter adds more flux and blows oxygen. Reactions remove residual iron and sulfur, producing blister copper at ~99 % purity. Slag may be recycled back to the flash furnace; sulfur dioxide is processed in an acid plant.
Refining
Blister copper still contains impurities that hinder final use. Fire refining first oxidizes residual sulfur and oxygen. Sodium carbonate flux removes trace arsenic and antimony. The molten copper, now ~99.5 % pure, is cast into large anodes for electrorefining.
- Each anode is placed in a polymer‑concrete cell with a copper cathode opposite. The cell holds an acidic copper sulfate solution, and when current passes, copper dissolves from the anode and plates onto the cathode.
- After 9–15 days, the cathode—now 99.95–99.99 % pure—measures ~300 lb (136 kg).
- The resulting slime, rich in gold, silver, selenium, and tellurium, is collected for precious‑metal recovery.
Casting
Refined copper is melted and cast into ingots, cakes, billets, or rods, tailored to end‑use:
- Ingots: rectangular or trapezoidal blocks used for alloying.
- Cakes: 8 in (20 cm) thick slabs up to 28 ft (8.5 m) long, rolled into plates, strips, sheets, or foil.
- Billets: 8 in (20 cm) diameter cylinders, extruded into tubing and pipe.
- Rods: ~0.5 in (1.3 cm) diameter, cast long and coiled, then drawn into wire.
Quality Control
Electrical applications demand ultra‑low impurity levels. Copper is refined to near‑absolute purity, and analytical sampling at each stage ensures that any necessary process adjustments are made promptly.
Byproducts & Waste
Smelting yields sulfuric acid, reducing furnace emissions. Recovering gold, silver, and other precious metals from the slag and slime adds economic value.
Mining waste—including overburden, tailings, and slag—can contain arsenic, lead, and other hazardous substances. In the U.S., the Environmental Protection Agency (EPA) regulates storage and remediation of such waste. While managing billions of tons of material poses challenges, it also offers opportunities to recover usable resources.
The Future
Demand for copper—especially in electrical and electronics sectors—remains robust. Current industry trends favor energy‑efficient, low‑emission processes and greater waste reduction. However, stringent environmental regulations and low‑grade ore constraints can raise production costs.
Recycling drives growth: over half of U.S. copper production originates from recycled sources. Copper machining waste accounts for 55 % of recycled copper, while 45 % comes from end‑use products like wiring and radiators. As primary mining costs rise, the recycled share is expected to climb.
Manufacturing process
- Tungsten‑Copper vs. Molybdenum‑Copper Alloys: Key Differences & Applications
- Copper Electroplating Project: Build a Smart System with Arduino UNO
- Optimizing Iron Ore Beneficiation for Efficient Steel Production
- Mining Iron Ore: Process, Types, and Extraction Techniques
- Your Comprehensive Guide to Copper Alloys: Types, Uses, and Benefits
- Oxygen‑Free Copper: Superior Conductivity and Purity for Electronics
- Precise Copper Machining: Key Precautions for Superior Surface Quality
- Copper vs. Brass: Key Material Properties for Electrical Conductors
- Master Copper Brazing: A Complete Guide to Joining Tubes & Fittings
- Heat Treatment of Copper and Copper Alloys: Enhancing Strength, Durability, and Performance



