Aspirin: From Willow Bark to Modern Tablets – Production & Quality Assurance
Background
Aspirin remains one of the safest, most affordable pain relievers worldwide. While earlier analgesics existed, it was aspirin’s mass‑market success at the turn of the twentieth century that cemented its position as the standard over‑the‑counter medication.
In the United States alone, consumers use 16,000 tons of aspirin tablets annually—roughly 80 million pills—contributing to an estimated $2 billion spend on non‑prescription pain relief each year.
Aspirin is available in multiple dosage forms: tablets (60–650 mg), capsules, caplets, suppositories, and liquid elixir. Tablets are the most common, but capsules and liquids cater to patients who need faster absorption or easier swallowing.
The drug treats a wide spectrum of conditions: single‑tablet daily use for cerebral thrombosis prevention; 2–6 tablets per day for pain or fever; and adjunctive therapy for rheumatic fever, gout, and rheumatoid arthritis. Aspirin also lowers heart‑attack risk and is a staple in research for its anti‑inflammatory and gene‑activating properties.
Until the 1970s, the mechanism of action was unclear. Scientists later discovered that aspirin and related NSAIDs inhibit cyclooxygenase enzymes, blocking the synthesis of prostaglandins that mediate pain and inflammation. Subsequent research has confirmed that aspirin’s therapeutic effect derives from its capacity to suppress inflammatory cell proliferation.
History
Salicylic acid, the active component of aspirin, was first isolated from willow bark (Salix alba) in 1763 by Reverend Edmund Stone of Chipping Norton, England. Hippocrates had earlier noted willow’s fever‑reducing properties.
Throughout the 1800s, chemists refined extraction techniques and synthesized salicylic acid. In 1853, French chemist Charles F. Gerhardt produced a rudimentary aspirin analogue. German chemist Felix Hoffmann, working for Bayer in 1897, developed a safer synthesis that eliminated the gastrointestinal irritation associated with salicylic acid. Hoffmann’s work led to Bayer’s 1899 launch of “Aspirin,” which dominated the market until World War I, when Sterling Drug acquired Bayer’s U.S. assets.
Bayer’s “Aspirin” trademark remains in many countries, though in the U.S. and U.K. the name is generic. Modern production has evolved from manual blending to fully automated, computer‑controlled processes.
Raw Materials
Hard aspirin tablets require only four core ingredients: acetylsalicylic acid (the active ingredient), corn starch (binder and filler), water, and a lubricant. The binder holds the tablet together, while the filler provides the necessary bulk. Lubricants—such as hydrogenated vegetable oil, stearic acid, talc, or aluminum stearate—prevent sticking to equipment.
Chewable formulations use different fillers (mannitol, lactose, sorbitol, sucrose, inositol) for rapid dissolution and palatability, supplemented by saccharin for sweetness and FD&C colorants (Yellow No. 5/6, Red No. 3/40, Blue No. 1/2, Green No. 3, select D&C dyes, iron oxides) approved by the FDA.
The Manufacturing Process
Aspirin tablets come in various shapes and sizes, often scored for easy splitting. Production follows a standardized sequence:
Weighing
- Accurate measurement of corn starch, acetylsalicylic acid, and lubricant in sterile canisters ensures batch consistency.
Mixing
- Corn starch is dispersed in cold purified water, heated, and stirred until a translucent paste forms.
- The paste, acetylsalicylic acid, and part of the lubricant are combined in a Glen Mixer, which blends and de‑aerates the mixture.
- The blend is mechanically divided into 7/8–1 inch “slugs.”
Dry Screening
- Small batches are screened manually through a stainless‑steel spatula; large batches use a Fitzpatrick mill.
- Remaining lubricant is added, and the mixture is gently blended in a rotary granulator.
Compression
- Single‑punch machines produce tablets for small batches; rotary tablet presses handle large‑scale runs.
- In a single‑punch system, a feed shoe deposits the mixture into a dye cavity, a steel punch compresses it, and a lower punch ejects the finished tablet.
- Rotary presses rotate the dye plate, sequentially filling cavities and ejecting tablets via synchronized upper and lower punches.
Testing
- Tablets undergo hardness, friability, and disintegration tests (e.g., Schleuniger/Heberlein hardness tester, Roche Friabilator, Vanderkamp disintegration tester) to confirm mechanical integrity and dissolution profile.
Bottling and Packaging
- Automated lines fill clear or color‑coated plastic or glass bottles, seal with cotton packing, aluminum caps, and child‑proof plastic/rubber lids.
- Post‑sealing, bottles are labeled, date‑coded, and boxed—first in individual cardboard cartons, then in larger shipping crates.
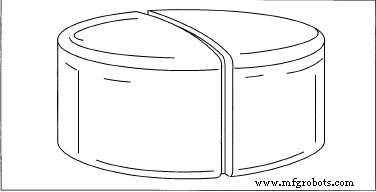
Finished tablets often feature a central “score” line to facilitate splitting.
Quality Control
Strict quality assurance aligns with FDA regulations. All equipment is sterilized before use, and operators perform routine checks to maintain dosage accuracy. Batch records document every step.
Post‑manufacturing, tablets are subjected to:
- Hardness and Friability: Ensures tablets can withstand packaging and handling.
- Disintegration: Confirms tablets dissolve at the intended rate in physiological conditions.
These tests use industry‑standard instruments—Schleuniger or Heberlein hardness testers, Roche Friabilator, and Vanderkamp disintegration apparatus—to verify each batch meets rigorous quality benchmarks.
Manufacturing process
- Aspirin: From Willow Bark to Modern Tablets – Production & Quality Assurance
- Maximizing Chain Longevity: Expert Tips for Maintenance and Replacement
- U.S. Agricultural Exports Face Ongoing Challenges in 2019
- How Real-Time Data Helps Brands Navigate Inflationary Pressures
- Innovative Electronic Skin Mimics Human Pain Sensing in Real Time
- Product Data Exchange: The Key Challenge for Automotive Manufacturers
- Three Key Challenges Facing the Utility Industry — How Asset Tags Offer Solutions
- Solving the 3 Most Common CNC Machining Challenges: Expert Solutions
- Top Procurement Pain Points in 2022: Insights from 250 Manufacturing Clients
- Infographic: Engineering Challenges in 2022 – Key Pain Points & Solutions



