Mechanism of Ordered, Densely Packed Nanoparticle Superlattices Formed by Gas‑Phase Deposition on Template‑Free Surfaces
Abstract
Nanoparticle superlattices are usually produced via solution‑based chemistry. In this work we demonstrate that a gas‑phase cluster‑beam deposition can generate self‑assembled monolayer superlattices on template‑free substrates. Fe nanoparticles form an average two‑dimensional, densely packed hexagonal lattice. By tuning the nanoparticle coverage, the monolayer spreads across the entire substrate. We propose that the ordered morphology arises from a balance between surface diffusion of the nanoparticles and their arrival rate, while inter‑particle attractive forces drive the lattice ordering. This model is validated by a series of carefully designed cluster deposition experiments.
Introduction
Nanoparticle superlattices, where the particles behave as artificial atoms arranged in crystalline order, have attracted significant interest since their first reports [1–7]. Such superstructures, assembled from tailored nanoparticle or nanocrystal building blocks, enable the design of novel materials and the tuning of their properties [8–16]. Non‑lithographic, bottom‑up approaches based on thermodynamically driven self‑assembly are especially appealing because they offer simpler technology and the potential for large‑scale production of structures beyond current lithographic limits.
Ordered arrays are most commonly produced from colloidal solutions, where monodisperse nanoparticles encapsulated in surfactant monolayers spontaneously form ordered lattices [17–20]. Biological molecules and their specific interactions have also been used to guide assembly [21–22]. The encapsulating ligands provide a soft shell that prevents disordered aggregation, tunes inter‑particle potentials, and programs lattice structures and inter‑particle distances [23–24]. However, these chemical additives can limit the intrinsic properties of the resulting assemblies [25].
In contrast, gas‑phase deposition yields nanoparticle assemblies free of organic encapsulation, offering pure inter‑particle interfaces and intrinsic properties. Nevertheless, creating spatially ordered arrays from the gas phase has traditionally required pre‑patterned templates or surface steps [26–30]. Even then, the resulting structures often contain many defects and exhibit relatively loose packing due to the large period of the templates. Post‑processing or sintering typically yields irregular aggregates [31]. To date, achieving ordered arrays on truly template‑free surfaces has remained elusive.
Gas‑phase nanocluster deposition is a mature process that allows precise control over size, density, and functional morphology [32–35]. Low‑energy clusters can diffuse across the surface and aggregate [36], while surface defects constrain their migration. Earlier studies reported either random arrays [37] or ramified aggregates [38] depending on deposition mass and kinetic energy, and these structures were considered highly disordered.
In this paper we show that well‑ordered, densely packed monolayer superlattices of Fe nanoparticles can form on a smooth amorphous carbon substrate by gas‑phase cluster deposition without any template. With moderate coverage, the ordered arrays extend over sub‑micron domains and exhibit clear superlattice periodicity. A series of deposition experiments elucidates the formation mechanism, highlighting the interplay between cluster flux, kinetic energy, and surface diffusion. This method offers a rapid, clean alternative to solution‑based synthesis, with the advantages of rapid processing, clean interfaces, and high stability.
Methods
Deposition of Nanoparticle Arrays
Fe and TiN clusters were generated using a magnetron plasma gas‑aggregation cluster source [37,39]. The source consists of a liquid‑nitrogen‑cooled aggregation tube ending in a 3 mm orifice. A magnetron discharge at 64 Pa Ar sputters atoms from the target, which then aggregate in the 75 mm long Ar stream. The resulting cluster beam exits the orifice into a high‑vacuum chamber (<1×10⁴ Pa) with a divergence of ≈3°. Substrates mounted on a rotary holder receive the beam, allowing the incident angle to be tuned from 0° to 90°. Deposition rates were monitored with a quartz crystal microbalance and precisely controlled via the cathode discharge power (MDX500, Advanced Energy). Substrates were kept at room temperature during deposition.
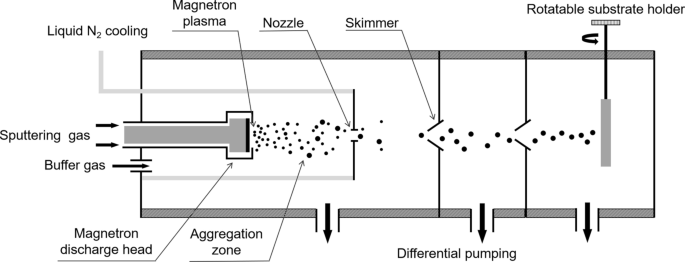
Schematic diagram of a magnetron plasma gas‑aggregation cluster source and the cluster beam deposition process
Characterization of the Nanoparticle Arrays
Nanoparticles were deposited on ultrathin amorphous carbon films on formvar‑coated 300‑mesh copper grids. Their structure and morphology were examined by transmission electron microscopy (TEM, FEI TECNAI F20s TWIN) at 200 kV. Energy‑dispersive X‑ray spectroscopy (EDX) provided compositional analysis. X‑ray photoelectron spectroscopy (XPS) with a monochromatic Mg Kα source (ESCALABMK‑II) assessed the oxidation state of Fe arrays. For magnetic studies, Fe nanoparticles were also deposited on silicon wafers and measured with a superconducting quantum interference device (SQUID, MPMS‑3).
Results and Discussion
Structure of the Nanoparticle Film
Figure 2a shows a TEM image of Fe arrays deposited vertically on amorphous carbon at a deposition rate of 0.1 Å s⁻¹ (5 min). The film consists of densely packed 2D monolayer domains that extend over 100–200 nm. A high‑magnification view (Fig. 2b) reveals a defect‑free monolayer. The arrays typically contain minor defects (lattice distortions, dislocations, vacancies, voids) and a modest size variation. The average particle diameter is 6.1 ± 1.6 nm, measured from ≥300 particles (Additional file 1: Fig. S1). The dispersion is larger than that of thiol‑passivated superlattices [40,41]. High‑resolution TEM (Fig. 2d) shows that individual Fe particles are predominantly single‑crystalline spheres, randomly oriented on the substrate. After prolonged air exposure, their surfaces oxidize, as confirmed by EDX mapping (O co‑localized with Fe) and XPS of the Fe 2p core level [Additional file 1: Fig. S3]. After Ar‑ion sputter cleaning, metallic Fe peaks dominate, indicating that oxides are confined to the particle surfaces. Thus, the arrays comprise closely packed core–shell particles with 0.85 nm amorphous oxide shells and a mean edge‑to‑edge spacing of 1.7 ± 0.6 nm.

a Low‑magnification TEM image of Fe arrays deposited on amorphous carbon. Domains of close‑packed 2D monolayers are visible. b High‑magnification TEM of an ordered densely packed 2D superlattice. c FFT of the array showing hexagonal spots. d HRTEM of individual Fe nanoparticles.
The FFT (Fig. 2c) displays a clear hexagonal pattern, confirming long‑range order. Only the first‑order hexagon is visible, indicating the domain size is limited.
Tailoring Morphology with Deposition Conditions
Deposition rate strongly influences the ordered monolayer formation. TEM images (Fig. 3a–c) of Fe arrays grown at 0.3–0.7 Å s⁻¹, with identical total coverage, show that increasing the rate reduces the domain size. Particle sizes remain consistent (≈6 nm). Quantitative analysis (Fig. 3d) reveals the average number of particles per monolayer domain decreases from 77 (0.1 Å s⁻¹) to 27 (0.7 Å s⁻¹). FFTs become progressively diffuse; at 0.7 Å s⁻¹ only a ring appears, indicating random distribution.
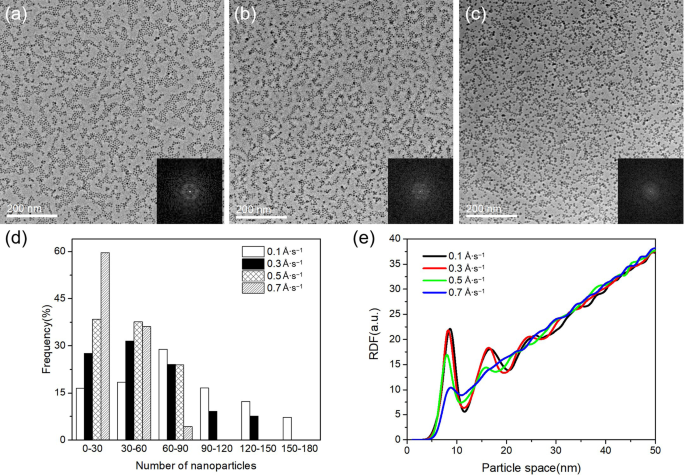
a 0.3 Å s⁻¹, b 0.5 Å s⁻¹, c 0.7 Å s⁻¹. Inset: FFT of each distribution. d Histogram of particles per domain (77, 55, 39, 27). e Radial distribution functions (RDFs) for each rate.
The RDFs (Fig. 3e) show that at 0.1 Å s⁻¹ and 0.3 Å s⁻¹ the first, second, and third peaks are sharp, corresponding to nearest‑neighbor spacings of 8 nm, 17 nm, and 24 nm, respectively. At 0.5 Å s⁻¹ the second peak diminishes and the third is indistinct; at 0.7 Å s⁻¹ only a weak first peak remains, indicating loss of short‑range order. Thus, a low deposition flux is essential for forming well‑ordered monolayers.
Substrate chemistry also governs the assembly. Fe arrays deposited on a formvar film (same conditions as Fig. 2) display a completely random distribution (Fig. 4). The lack of mobility on the polymer surface prevents diffusion and aggregation. In contrast, amorphous carbon offers a smooth, defect‑rich surface that facilitates nanoparticle migration and coalescence. The necessity of some mobility is clear: without it, the particles remain pinned and no ordered monolayer forms.

Fe nanoparticle arrays on formvar film (0.1 Å s⁻¹). No order is observed.
The formation mechanism can be summarized as follows: initially, incoming clusters are trapped by surface defects and act as nuclei. Subsequent arrivals diffuse across the surface; if the arrival flux is low, each particle can explore the surface, locating the energetically favorable lattice sites dictated by van der Waals attraction. When the flux exceeds the diffusion capacity, the surface becomes saturated and particles aggregate randomly, forming fractal islands. Thus, the balance between surface diffusion and deposition rate governs the degree of ordering.
We also increased the lateral migration energy by depositing at a 45° glance incidence, which transfers part of the kinetic energy into surface diffusion. The resulting arrays (Fig. 5a) exhibit larger domains and sharper FFT spots; the RDF (Fig. 5b) shows enhanced second‑ and third‑neighbor peaks, confirming improved ordering.
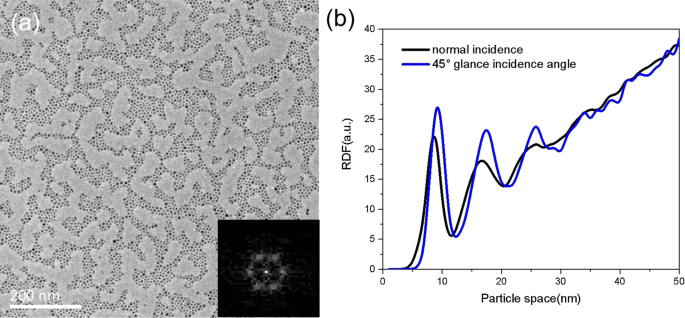
a TEM of Fe arrays deposited at 45° incidence. Inset: FFT. b RDF comparison with normal incidence.
Inter‑particle van der Waals forces, rather than ligand mediation, drive the ordering. Magnetic dipolar interactions in Fe are negligible in these superparamagnetic nanoparticles, as evidenced by the absence of hysteresis at 300 K (Fig. 6a). TiN nanoparticles, which are non‑magnetic, also form ordered densely packed monolayers under identical conditions (Fig. 6b), confirming that the mechanism is universal for metallic clusters.
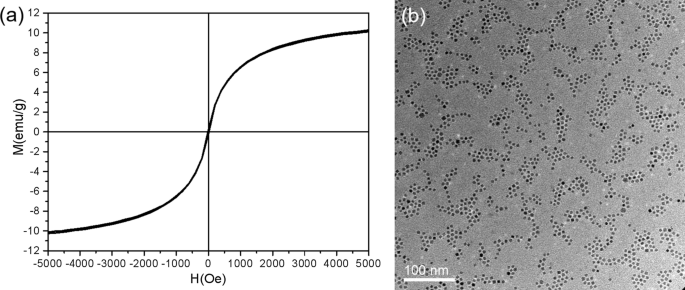
a Hysteresis loop of Fe arrays (superparamagnetic). b TiN array TEM showing ordered monolayers.
Large‑scale monolayers were achieved by controlling the total deposition mass. A nearly complete monolayer of Fe nanoparticles (≈100 % coverage) at 0.1 Å s⁻¹ with 45° incidence spans the entire deposition spot (Fig. 7). The FFT shows two hexagonal rings, confirming long‑range order over centimeter‑scale domains.

Fe nanoparticle film at ~100 % coverage (0.1 Å s⁻¹, 45°). The FFT inset confirms hexagonal ordering.
Conclusion
We have demonstrated a simple, rapid, and scalable method to fabricate ordered, densely packed 2D monolayer superlattices of Fe (and TiN) nanoparticles on template‑free amorphous carbon by gas‑phase cluster deposition. By finely tuning deposition rate and incident angle, monolayers with well‑defined hexagonal lattices extend over 100–200 nm domains and can be expanded to cover entire substrate surfaces. The ordering arises from a balance between surface diffusion and arrival flux, with inter‑particle van der Waals attraction driving the lattice. This technique offers a clean, additive‑free alternative to colloidal chemistry for producing large‑area, defect‑free nanoparticle superlattices.
Availability of Data and Materials
The datasets used and/or analyzed during this study are available from the corresponding author on reasonable request.
Abbreviations
- TEM:
Transmission electron microscope
- EDX:
Energy‑dispersive X‑ray spectroscopy
- XPS:
X‑ray photoelectron spectroscopy
- HRTEM:
High‑resolution transmission electron microscopy
- FFT:
Fast Fourier transform
- RDFs:
Radial distribution functions
Nanomaterials
- Nanotechnology: From In‑Vivo Imaging Systems to Controlled Drug Delivery
- Precise Glancing‑Angle Deposition of Gold Nanohelices on Lithography‑Free Nanoseeded Surfaces
- Water‑Soluble α‑NaGdF₄/β‑NaYF₄:Yb,Er Core–Shell Nanoparticles: Controlled Synthesis and Superior Upconversion Luminescence
- Calcium‑Mediated Exopolymeric Substance Release in Marine Phytoplankton Exposed to Engineered Nanoparticles
- Decoding the Skin‑Core Chemical Architecture of Stabilized Polyacrylonitrile Fibers
- Nanoporous Silica Tablets for Quantifying Biomolecules and Nanoparticles via Water Self‑Diffusivity in Bio‑Inspired Pores
- Gold Nanoparticle-Drug Interactions Cause Acute Kidney Damage in Mice: A Size-Dependent Toxicity Study
- Boosting ZnO UV Emission with Silver Nanoparticle Arrays via Surface Plasmon Resonance
- Influence of Accumulated Laser Energy on Nanoparticle Morphology in Pulsed Dewetting of AgCu Thin Films
- Eco‑Friendly, Cost‑Effective Synthesis of Tin Oxide Nanoparticles: A Comprehensive Review of Methods, Formation Mechanisms, and Applications



