Co‑Adsorption Effects on Interfacial Charge Transfer in CdSe Quantum Dot–N719/D131 Dye Composites
Abstract
Quantum‑dot (QD) dye composites are promising for next‑generation photovoltaics, yet the interfacial electronic environment often limits charge‑transfer efficiency. In this study we employed density‑functional theory (DFT) to probe how the co‑adsorption of the broadband‑absorbing D131 dye influences the binding geometry, electronic alignment, and charge‑transfer couplings of N719‑functionalized Cd33Se33 QDs. Our calculations show that isocyanate anchors favor electron transfer, whereas carboxylate anchors promote balanced electron–hole separation. The presence of D131 stabilises the dye’s frontier orbitals, reduces mid‑gap states, and increases the driving force for electron injection while suppressing back‑transfer. These insights clarify how dye‑binding motifs govern interfacial dynamics and provide design rules for high‑efficiency QD‑based devices.
Introduction
Dye‑sensitized photocatalysis and solar cells attract intense interest because they harness abundant solar energy without relying on fossil fuels. Transition‑metal complexes such as Ru(bpy)3Cl2 offer long‑lived metal‑to‑ligand charge‑transfer (MLCT) states that enable efficient electron injection into semiconductors. However, the narrow absorption window of most dyes limits their light‑harvesting capacity. Quantum dots (QDs) complement dyes with size‑tunable bandgaps and the potential for multiple carrier generation, which can increase the photocurrent density in sensitized systems.
Upon photoexcitation, electrons in a QD are promoted from the valence band (VB) to the conduction band (CB). Effective charge‑separation requires that these carriers be extracted across the QD–dye interface before they recombine. The interfacial electronic structure is highly sensitive to the nature of the anchoring groups, surface pH, and the presence of co‑adsorbents, which can alter both the energy alignment and the electronic coupling between donor and acceptor states. While many studies have examined single‑dye adsorption on TiO2, systematic analyses of multi‑dye systems on CdSe QDs remain scarce.
Here we focus on a model CdSe QD functionalized with the high‑performance N719 dye and the co‑adsorbent D131, a commonly used “shunt” dye that broadens the spectral response and suppresses aggregation. By exploring a range of anchoring chemistries (carboxylate versus isocyanate) and deprotonation states, we elucidate how co‑adsorption modifies the interfacial charge‑transfer landscape.
Computational Methodology
Ground‑state geometries of N719–D131–Cd33Se33 composites were optimized using the B3LYP functional with a LANL2DZ/6‑31G* mixed basis set. Relativistic corrections were applied to Cd, and the polarizable continuum model (PCM) simulated solvent effects (benzene). After optimization, total and projected density of states (TDOS/PDOS) were calculated via a Gaussian‑broadened Kohn–Sham spectrum with a 100 meV line width.
Three key charge‑transfer pathways were examined (Figure 1): (1) QD→dye electron transfer (et), (2) dye→QD electron–hole recombination (re), and (3) QD→dye hole transfer (ht). Charge‑localized reactant and product states were obtained with constrained DFT (CDFT). Electronic couplings were extracted from the secular equation using NWChem 6.8. All calculations employed the same functional and basis set for consistency.
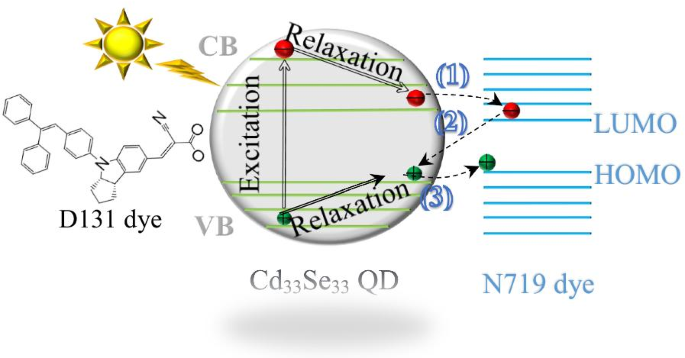
Results and Discussion
Electronic and Geometrical Structures of QD@dye Composites
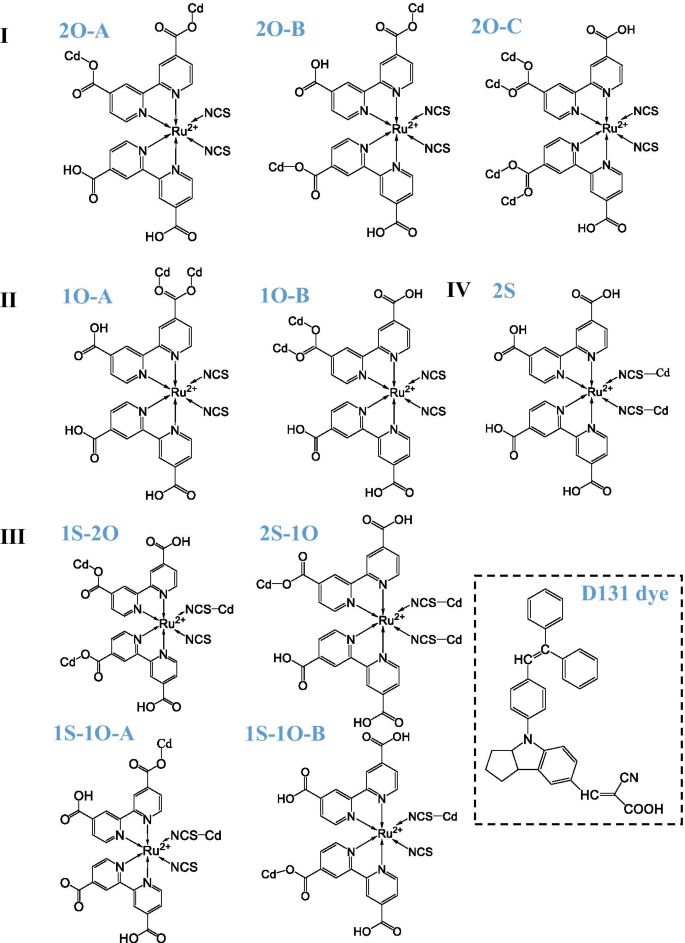
Both N719 and D131 anchor to the QD via the most reactive 2‑coordinate Cd sites. N719 can adopt four distinct anchoring motifs, defined by the number and type of carboxylate (O) or isocyanate (S) groups and the deprotonation pattern (A, B, C). The schematic of each motif is shown in Figure 2. The binding energy of a N719–D131 complex ranges from 1.42 eV to 2.83 eV, lower than that of a single‑dye system, reflecting the steric competition introduced by D131.
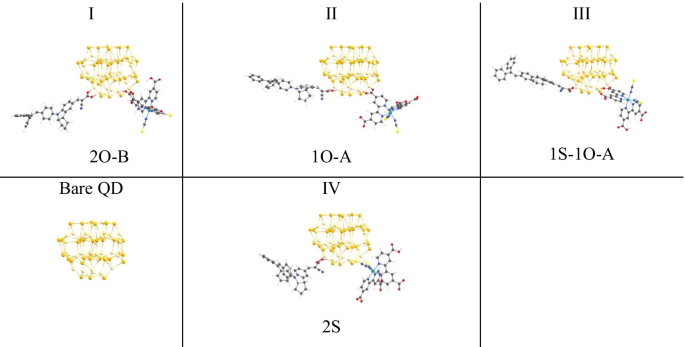
Our model omits surface ligands, a reasonable simplification because ligand passivation has negligible influence on the electronic structure of small CdSe QDs and avoids additional computational cost.
Energy Alignment of QD and Dye Orbitals
The calculated bandgap of the pristine Cd33Se33 QD is 2.78 eV, consistent with experimental magic‑size nanocrystals. Functionalization introduces mid‑gap states that can serve as recombination centers. For carboxylate‑anchored N719, the highest occupied state is dominated by dye orbitals, facilitating hole transfer to the dye. In contrast, isocyanate anchoring pushes dye states deeper into the QD VB, suppressing hole transfer. Co‑adsorption with D131 mitigates this effect, stabilising the occupied frontier orbitals and preserving favorable hole‑transfer energetics.
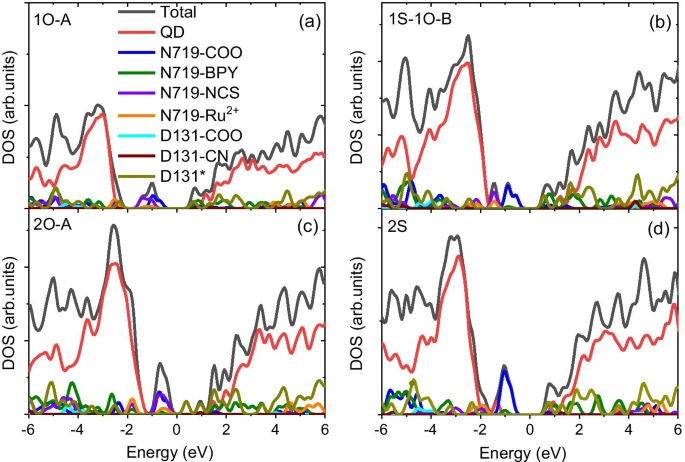
Solvent screening lowers the energy of dye states relative to the QD VB, reducing the driving force for hole transfer only in isocyanate‑anchored structures. Electron injection remains favourable across all motifs because the CB edge is dictated by the N719 LUMO, regardless of anchor type.
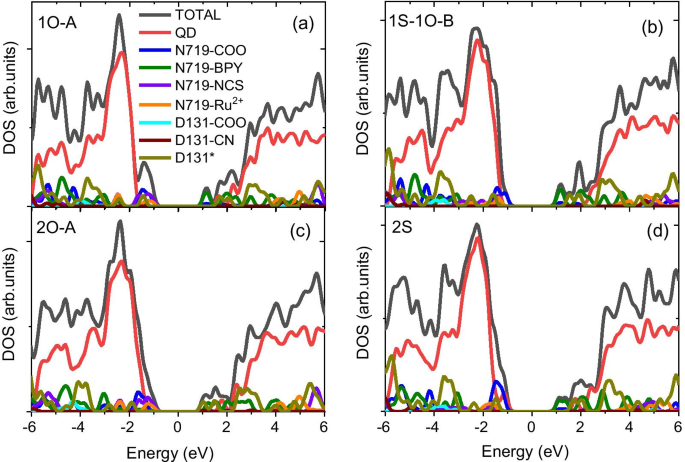
Electronic Coupling
Electronic coupling (HDA) determines the rate of charge transfer and depends strongly on the donor–acceptor distance. For carboxylate‑anchored motifs, the coupling for electron transfer (et) is comparable to that for hole transfer (ht), implying efficient charge separation. In isocyanate‑anchored motifs, Het exceeds Hht by up to two orders of magnitude, favouring electron injection over hole extraction. Importantly, Het consistently exceeds Hre in the co‑adsorbed system, indicating a faster electron injection than recombination, a desirable feature for photovoltaics. In contrast, a single‑dye system shows comparable et and re couplings, leading to higher recombination losses.
Solvent inclusion generally increases all couplings by enhancing orbital delocalisation, further improving charge‑transfer efficiency.
Conclusion
DFT calculations reveal that co‑adsorption of D131 with N719 on CdSe QDs fine‑tunes the interfacial electronic landscape. Carboxylate anchors support balanced electron–hole transfer and reduce recombination, while isocyanate anchors bias the system toward electron injection. The presence of D131 stabilises the dye’s frontier orbitals, suppresses mid‑gap states, and increases the driving force for electron injection, thereby enhancing charge‑separation efficiency. These findings provide a mechanistic basis for designing QD–dye composites with tailored anchoring chemistries to optimise photovoltaic performance.
Availability of Data and Materials
All data generated in this study are presented in the published article.
Abbreviations
- QD
- Quantum dot
- DFT
- Density functional theory
- VB
- Valence band
- CB
- Conduction band
- et
- Electron transfer
- ht
- Hole transfer
- re
- Electron–hole recombination
- CDFT
- Constrained density functional theory
- N719
- Di‑tetrabutylammonium cis‑bis(isothiocyanato)bis(2,2′‑bipyridyl‑4,4′‑dicarboxylato)ruthenium(II)
- D131
- (2E)-2‑Cyano-3-{4-[4-(2,2‑diphenylvinyl)phenyl]-1,2,3,3a,4,8b‑hexahydrocyclopenta[b]indol‑7‑yl}acrylic acid
- MLCT
- Metal‑to‑ligand charge transfer
- CdSe
- Cadmium selenide
- CdTe
- Cadmium telluride
- TDOS
- Total density of states
- PDOS
- Projected density of states
- O
- Oxygen
- S
- Sulphur
Nanomaterials
- Core/Shell CoFe₂O₄/Fe₃O₄ Nanoparticles: Interfacial Magnetism and Tunable Anisotropy
- Impact of Shell Thickness on Energy Transfer in Eu‑Doped Core–Shell ZnSe/ZnS Quantum Dots
- How Surface Charge Alters Oil Contact Angles in Water‑Decane–Silicon Dioxide Systems
- How Nanoparticle Size and Agglomeration Influence Interfacial Strength and Tensile Performance in Polymer Nanocomposites
- Magnetic ZnO/Fe3O4/g‑C3N4 Nanocomposite: High‑Efficiency Visible‑Light Photocatalyst for Monoazo Dye Degradation
- Enhancing Solid‑State CuInS₂ Quantum‑Dot Solar Cells: Optimizing Charge Recombination via Controlled SILAR Deposition and Annealing
- Efficient Charge Transfer in Au/CdSe Janus Nanoparticles Boosts Photocatalytic Hydrogen Production
- Improving WLED Color Rendering with Composite Orange‑Red Quantum Dots
- Enhancing Transfer Charge in Knit-Structured Triboelectric Nanogenerators via Surface Morphology Analysis
- Boosting Carbon-Based Perovskite Solar Cells: Tin-Doped Quantum Dot Interfacial Layer Enhances Photovoltaic Efficiency



