Hair Dye: From Ancient Roots to Modern Manufacturing
Historical Overview
Hair dye is one of the oldest beauty practices known to humanity. Ancient Egyptians, Greeks, Hebrews, Persians, Chinese, and early Hindus all documented the use of natural colorants. Early formulations combined plant extracts, metallic compounds, and sometimes both.
Roman bleaching methods relied on rock alum, quicklime, and wood ash, while herbal preparations such as mullein, birch bark, saffron, myrrh, and turmeric were also employed. Henna, a reddish dye, was widely recognized across many cultures.
Prior to modern synthetic dyes, Europeans and Asians used a variety of plant extracts. Indigo, traditionally a fabric dye, was mixed with henna to produce shades ranging from light brown to black. Chamomile flowers were long used for lightening hair, a practice that persists in many contemporary products. Bark, leaves, and nuts from trees like brazilwood and mulberry-derived fustic provided brown tones, while walnut leaves, nut husks, and oak galls were also used. These natural dyes were sometimes combined with copper or iron to deepen or enrich the resulting color.
The golden red hue that graces many Renaissance portraits was achieved with a simple Italian recipe: comb a solution of rock alum, black sulfur, and honey through the hair, then expose it to sunlight. Sixteenth‑century formulas also included lead, quicklime, salt, or silver nitrate in rose water. Powdered colorants—white from wheat or potato starch mixed with plaster of Paris, chalk, or burnt alabaster—were popular among aristocrats in the seventeenth and eighteenth centuries. Colored powders, created by adding natural pigments like burnt sienna or umber to white starch, or using India ink for black, were also used. In Biblical times, powdered gold was applied to hair; a brief 19th‑century fad saw gold and silver powders return among the European elite.
Prior to the late 19th century, these primitive preparations were the only options. Hydrogen peroxide, discovered in 1818, was showcased at the 1867 Paris Exposition as an effective hair lightener. A London chemist and a Parisian hairdresser marketed a 3% hydrogen peroxide solution—dubbed 'eau de fontaine de jouvence golden'—as the first modern chemical dye. Subsequent advances introduced synthetic organic dyes: pyrogallol, derived from walnut shells, began to color hair brown in 1845 and was often paired with henna. The 1880s saw the introduction of amino dyes, notably p‑phenylenediamine, patented in Germany in 1888. These dyes required a weak alkaline base and hydrogen peroxide to develop, producing natural‑looking blacks and shades of red and brown.
In 1910, French hairdresser Gaston Boudou launched a standardized range of 18 colors, from black to light blond. Prior to this, colors were mixed on the spot, resulting in inconsistent hues. Boudou’s line offered predictable results and quickly gained popularity across Europe and the United States. However, amino dyes caused allergic reactions in a significant portion of users. U.S. researchers responded by creating a less toxic amino‑based dye, standardizing application methods, and establishing strict purity specifications. The mid‑20th century marked another milestone when Clairol introduced the first one‑step dye in 1950, eliminating the need for pre‑shampooing and lightening. Intensive marketing drove the percentage of women dyeing their hair in the U.S. from about 8% to nearly 50% by 1973.
Raw Materials
Commercial hair dye formulas are intricate, comprising dozens of ingredients that vary by manufacturer. Generally, a formula includes dyes, modifiers, antioxidants, alkalizers, surfactants, fragrances, and other specialty chemicals that confer specific properties such as softness or permanence. Dye molecules are typically amino compounds—names such as 4‑amino‑2‑hydroxytoluene and m‑aminophenol appear on ingredient lists. Pigments like titanium dioxide and iron oxide are also common.
Modifiers stabilize pigments or adjust tonal nuances. Resorcinol is a widely used modifier, but many others exist. Antioxidants, chiefly sodium sulfite, protect dyes from oxidation. Alkalizers—often ammonium hydroxide—raise the pH, optimizing dye performance. Additional ingredients may be added to create a creamy texture, foam, or to enhance the overall effect of the product.
Most formulations are sold with a separate developer, usually based on hydrogen peroxide, supplemented by other chemicals as dictated by the manufacturer.
The Manufacturing Process
Ingredient Verification
- Before production, each chemical is verified for identity and potency. Manufacturers conduct in‑house testing or rely on Certificates of Analysis from reputable suppliers.
Weighing
- Ingredients are weighed according to batch specifications. Small‑quantity components are measured precisely, while larger amounts may be piped from storage tanks for large batches.
Pre‑Mixing
- Some dyes are pre‑mixed in heated water (158°F/70°C). The mixture is agitated for about 20 minutes. Additional solvents or additives may be incorporated during this step.
Mixing
- Pre‑mixed dye is transferred to a larger mixing tank containing the remaining ingredients. Small batches use portable tanks (~1,600 lb/725 kg), while large production lines employ interconnected vessels up to ten times that size.
- If the pre‑mix is hot, the second mix must cool before adding solvents, surfactants, or alkalizers. Alcohols are introduced only after the mix reaches 104°F/40°C to prevent evaporation, and fragrances are typically added at the end.
Filling
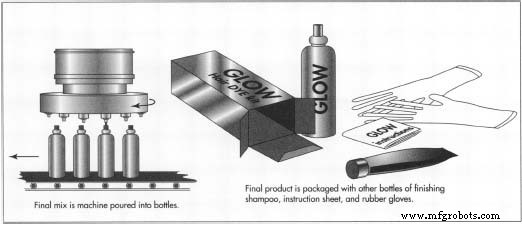
- Finished dye is pumped into a filling station. A nozzle dispenses measured amounts onto a conveyor belt, where bottles are filled, labeled, and capped.
Packaging
- Bottles are boxed with the developer, instructions, and optional accessories (e.g., gloves, finishing shampoo). The completed cartons are stored in warehouses awaiting distribution.
Quality Control
Regulatory agencies, such as the U.S. FDA, set strict limits on cosmetic ingredients to ensure safety. Before manufacturing, laboratory tests confirm non‑irritation, consistent performance, and correct viscosity and pH. During production, technicians sample batches for viscosity, pH, and color development on hair swatches. New or modified formulas undergo additional post‑filling testing to guarantee reliability.
The Future
Automation is increasingly integral to hair dye production. Computer systems now manage ingredient weighing, reaction control, and pump regulation, enhancing precision and efficiency. Continued digitization promises fully automated plants and even faster, more sustainable manufacturing workflows.
Manufacturing process
- Amber: From Ancient Resin to Scientific Treasure – History, Properties, and Modern Uses
- The Guillotine: History, Design, and Legacy
- The Modern Hair Dryer: Design, Safety, and Future Innovations
- Indigo Dye: History, Production, and Future Trends
- Silicon: Properties, Production, and Market Outlook
- The Ukulele: From Portuguese Roots to Modern Craftsmanship
- The Complete Guide to the U.S. Chicken Industry: History, Production, and Quality Control
- Compost: The Ultimate Soil Enrichment Solution
- Gelatin: From Ancient Protein to Modern Applications – Production, Uses, and Safety
- The Evolution and Manufacturing of Modern Pin‑Tumbler Locks



