Silicon: Properties, Production, and Market Outlook
Silicon
Background
Silicon ranks as the second most abundant element in Earth’s crust, surpassed only by oxygen. It appears naturally in rocks, sand, clays, and soils, usually bound as silicon dioxide or as silicates that also contain oxygen and other metals. These compounds are ubiquitous in water, the atmosphere, plants, and even some animals.
On the periodic table, silicon is the 14th element and a member of Group IVA, alongside carbon, germanium, tin, and lead. Pure silicon is a dark‑gray crystalline solid, sharing a diamond‑like lattice structure. Key physical data: melting point 2,570 °F (1,410 °C), boiling point 4,271 °F (2,355 °C), density 2.33 g/cm³.
Thermal treatment of silicon leads to halide formation with fluorine, chlorine, bromine, or iodine, and to silicides with certain metals. Heating in an electric furnace with carbon yields silicon carbide, a wear‑resistant ceramic. Hydrofluoric acid is the only acid that can etch silicon. At elevated temperatures, silicon reacts with water vapor or oxygen to form a protective silicon dioxide layer.
When refined to high purity and doped with boron, phosphorus, or arsenic, silicon becomes a semiconductor. The purification typically involves reducing silicon tetrachloride or trichlorosilane to elemental silicon, followed by crystal growth via the Czochralski method. Lower‑purity silicon serves as a reducing agent and alloying element in metallurgy, enhancing casting ease, strength, and hardness of alloys such as aluminum, brass, and bronze. Silicon oxides and silicates are key raw materials for concrete, bricks, glass, ceramics, and soap, while silicon metal is the foundation for silicone polymers used in synthetic oils, caulks, sealers, and anti‑foaming agents.
History
Silicon was first isolated and identified as a distinct element in 1824 by Swedish chemist Jöns Jacob Berzelius, following an impure extraction in 1811. Crystalline silicon was produced by electrolysis in 1854.
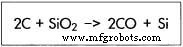 The reaction of silica with carbon inside an electric arc furnace generates silicon metal.
The reaction of silica with carbon inside an electric arc furnace generates silicon metal.
Electric arc furnaces—originally invented by Paul Louis Toussaint Heroult in 1899 for steelmaking—are now the standard for silicon production. The first U.S. furnace operated in Syracuse, New York, in 1905, and modern designs feature advanced electrodes and heat‑management systems.
Raw Materials
Silicon metal is produced by reducing silica (SiO₂) with carbon sources such as coke, coal, or hardwood chips. Typical feedstock characteristics include:
- Metallurgical‑grade gravel: 99.5 % silica, particle size 3 × 1 or 6 × 1 in (8 × 3 cm or 15 × 3 cm).
- Low‑ash coal: 1–3 % ash, ~60 % carbon, particle size matched to gravel.
- Hardwood chips: ½ × ⅛ in (1 × 3 cm).
Manufacturing Process
The Reduction Process
- Weigh and load raw materials into the furnace via a fume hood or conveyor. A typical batch contains 1,000 lb (453 kg) of gravel and chips, and 550 lb (250 kg) of coal.
- Ignite the electric arc between electrodes; temperatures reach 4,000 °F (2,350 °C), melting the charge and driving the reaction SiO₂ + C → Si + CO.
- Continually charge the furnace while the molten silicon forms. The process lasts 6–8 hours.
- After melting, introduce controlled air or oxygen to reduce calcium and aluminum impurities. Final silicon metal grades range from 98.5 % to 99.99 % purity.
Cooling and Crushing
- Separate slag (oxidized material) into pots and allow it to cool.
- Cast molten silicon in large iron trays (≈8 ft × 8 in). Once cooled, the metal is poured into trucks, weighed, and placed in storage.
- Prior to shipping, silicon is sized to customer specifications, often using jaw or cone crushers.
Packaging
Silicon metal is typically shipped in 3,000‑lb (1,361 kg) sacks or wooden boxes. Powdered silicon is packaged in 50‑lb (23 kg) pails, 500‑lb (227 kg) drums, or 3,000‑lb (1,361 kg) sacks.
Quality Control
Statistical Process Control (SPC) and computer‑controlled monitoring maintain product consistency. Key controllable variables are raw material ratios and furnace temperature. Laboratory analyses verify chemical composition, and regular supplier audits trace quality from extraction to delivery.
Byproducts & Waste
Silica fume, a byproduct of the reduction reaction, is sold to the refractory and cement sectors to enhance strength. It also finds use as a heat‑insulation filler and in rubber, polymers, and grouts. The cooled slag is crushed into sandblasting material or sold to other manufacturers. Electric arc furnaces emit particulates, so facilities must comply with EPA regulations.
Future Outlook
While Western analysts projected a ~7 % annual growth in chemical‑grade silicon demand through 2003, recent economic slowdowns in Asia and Japan have moderated that trajectory. If supply remains ahead of demand, prices could continue to decline. Conversely, the automotive sector shows positive momentum, with more manufacturers adopting aluminum‑silicon alloys for vehicle components. Emerging production methods—such as bulk amorphous silicon via supercooling and hydrothermal synthesis of porous silicon powders—are under investigation for advanced optical applications.
Manufacturing process
- Amber: From Ancient Resin to Scientific Treasure – History, Properties, and Modern Uses
- The Guillotine: History, Design, and Legacy
- The Ukulele: From Portuguese Roots to Modern Craftsmanship
- The Complete Guide to the U.S. Chicken Industry: History, Production, and Quality Control
- Compost: The Ultimate Soil Enrichment Solution
- Gelatin: From Ancient Protein to Modern Applications – Production, Uses, and Safety
- The Evolution and Manufacturing of Modern Pin‑Tumbler Locks
- Vodka: From Ancient Distillation to Modern Production
- Milk: Production, Processing, and Nutrition in the United States
- The History and Modern Production of Mascara: From Ancient Kohl to Contemporary Formulations



