Indigo Dye: History, Production, and Future Trends
Background
Indigo, chemically known as indigotin, is a historic dye traditionally sourced from indigo and woad species. Archaeological evidence places its use in ancient Egypt around 1600 B.C., with subsequent discoveries across Africa, India, Indonesia, and China.
The dye delivers a vivid blue to textiles. Typical processes involve soaking and drying cotton or linen 15–20 times, while silk may require over 40 cycles. Sun‑drying post‑dyeing intensifies the hue. Indigo uniquely colors the fiber surface, leaving inner layers lighter—a trait that gives denim its distinctive worn appearance. Modern production is largely synthetic, available as a 100 % powder or a 20 % solution. In the early 1990s, market prices hovered around $44 per pound ($20 per kilogram).
History
The term "indigo" derives from the Roman "indicum", implying "from India". Although the dye originated in the region, it is cultivated worldwide—in Asia, Java, Japan, and Central America. The ancient Greek "nil" also referred to the color, giving rise to the Arabic "al‑nil" (blue) and the English "aniline".
While multiple species yield indigo, the leguminous *Indigofera tinctoria* and *I. suffruticosa* have historically dominated production. With only 2–4 % of the leaf mass as dye, vast plant volumes were needed, prompting global cultivation and the establishment of plantations to secure supply.
The Industrial Revolution spurred a surge in indigo demand, driven in part by Levi Strauss & Co.'s blue denim. Traditional extraction proved too costly for mass production, prompting chemists to devise synthetic routes. In 1883, Adolf von Baeyer elucidated indigo’s structure, revealing that alkali‑treated omega‑bromoacetanilide yields oxindole. Building on this, K. Heumann engineered a practical synthetic pathway, leading to commercial production within 14 years. Baeyer received the Nobel Prize in 1905 for his groundbreaking work.
By the late 1990s, BASF AG dominated the global indigo market, supplying about half of all dyestuffs. Increasing awareness of the hazardous reagents in conventional synthesis has driven industry toward greener alternatives.
Raw Materials
Natural indigo production relies exclusively on the leaves of *Indigofera* species, woad (*Isatis tinctoria*), and polygonum, which contain the highest dye concentrations. Synthetic routes employ a range of chemicals, detailed below.
The Manufacturing Process
Natural Extraction
- Indican, the natural precursor, is released from freshly harvested leaves in a fermentation vessel. The enzyme indimulsin hydrolyzes indican into indoxyl and glucose, producing a yellow‑tinted broth and releasing CO₂.
- After roughly 14 hours, the broth is transferred to a second tank where aeration oxidizes indoxyl into indigo, which precipitates at the bottom. The supernatant is removed, and the pigment is collected in a third tank for heat‑termination of fermentation, filtration, and drying into a thick paste.
- In Japan, a variant employs the polygonum plant mixed with wheat husk, limestone, lye ash, and sake, allowing a week‑long fermentation that yields the pigment sukumo.
Synthetic Production
- Modern synthetic routes combine multiple reactants under controlled conditions, forming indigo through a series of chemical transformations. The process generates ancillary by‑products that must be managed.
- Reactions occur in large stainless‑steel or glass vessels equipped with jacketed cooling/heating jackets to regulate temperature. Production is predominantly batch‑based, though continuous processes have been pioneered by German manufacturers.
Types of Reactions
- The initial commercial synthesis built on Heumann’s work: N‑phenylglycine reacts with alkali to produce indoxyl, which is air‑oxidized to indigo, though the yield is modest. A more efficient, industry‑favoured route employs anthranilic acid, popularized by BASF and Hoechst for over three decades. A widely used variant reacts aniline, formaldehyde, and hydrogen cyanide to form phenylglycinonitrile, which is hydrolyzed to phenylglycine and then converted to indigo. Presently, a sodamide‑alkali process lowers the reaction temperature from ~570 °F (300 °C) to 392 °F (200 °C), markedly improving efficiency.
Finishing Operations
- Once the synthesis concludes, the raw dye is thoroughly washed to eliminate impurities, then dried into a fine powder. This powder is either stored in drums or reconstituted with water to produce a 20 % solution for distribution in pails.
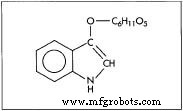 The chemical symbol for indican, the compound found in the leaves of the indigo plant that is used to make indigo dye.
The chemical symbol for indican, the compound found in the leaves of the indigo plant that is used to make indigo dye.
Quality Control
Manufacturers rigorously monitor each stage of indigo synthesis—pH, temperature, and reaction time—to maintain consistent quality. Deviations can lower yield, increasing production costs. Indigo’s hue is standardized by the Color Index as CI Natural Blue (CI 75780), ensuring uniformity across batches.
Byproducts/Waste
Indigo production generates diverse waste streams that must be managed responsibly. Beyond the primary reactants, side products—some hazardous—require disposal per local and federal regulations. Waste can enter the environment during synthesis, dye application, and washing of denim, the latter often releasing indigo into runoff during stone‑washing or wet processing.
The Future
Much of the demand for indigo is now met with alternative blue dyes, and the majority of indigo used worldwide is produced outside the United States. Researchers are exploring greener manufacturing routes, notably biocatalytic processes. Genencor International in Rochester, NY, is evaluating a biotechnology‑based method that yields chemically identical indigo, yet the technology remains costly. The company seeks a major partner to scale the process.
Fabric manufacturers are also innovating dye application. Burlington’s Denim Division launched its 1994 "Stone Free" system, accelerating indigo fade by 50 % during the stone‑wash cycle. By reducing reliance on pumice stones and bleach, the method cuts material handling costs and environmental impact while preserving the desired worn look.
Manufacturing process
- Hair Dye: From Ancient Roots to Modern Manufacturing
- Premium Pigments & Dyes: Product Range of Trusted Manufacturers
- Dyes: The Essential Colorants for Textiles, Paper, and More
- Professional Nylon Part Dyeing with Markforged: Step‑by‑Step Guide
- The Unexpected Purple: How Indirubin Reveals Natural Indigo’s Hidden Story
- Mastering Indigo Fermentation: Lessons from a Five‑Year‑Old Vat
- Choosing the Right Indigo Vat Size for Studio Dyeing: Why 50‑Liter Vats Win
- Mastering Fresh Indigo Leaf Dyeing: A Sustainable Approach
- Optimizing Yellow Flavonoid Dyes: The Crucial Role of pH and Chalk in Natural Dyeing
- Professional Guide: Dyeing 3D Printed Polyamide Parts for Vibrant Color and Durability



