Sliding Speed Effects on Tribochemical Wear of Oxide‑Free Silicon
Understanding how sliding speed governs the tribochemical wear of oxide‑free single‑crystalline silicon is essential for optimizing ultra‑precision surface finishing. We report the speed‑dependent nanowear of freshly etched silicon against SiO2 microspheres in humid air (60 % RH) and deionized water. When the contact pressure is insufficient to trigger silicon yield, the presence of water molecules facilitates a tribochemical reaction that progressively reduces wear volume logarithmically with increasing speed, eventually plateauing. Transmission electron microscopy and Raman spectroscopy reveal that the dynamic rupture and reformation of interfacial Sisubstrate–O–Sitip bonds underpin this behavior. Material wear can be mechanical—fracture, plastic deformation, or viscous flow—or tribochemical, driven by stress‑assisted bond breaking and sometimes chemical corrosion. Single‑crystal silicon is a cornerstone of semiconductor manufacturing, and chemical mechanical polishing (CMP) remains the most effective method for achieving atomically smooth substrates. In CMP, material removal before silicon yields is predominantly tribochemical. Because CMP involves a complex mix of pad, slurry, load, and speed, many studies simplify the system by examining silicon against a single SiO2 microsphere. Earlier work has identified a tribochemical pathway in which interfacial bonding bridges transfer mechanical energy to the silicon substrate, promoting atom removal. However, these studies largely used silicon with a native oxide layer, which markedly alters wear behavior. Few investigations have addressed truly oxide‑free silicon, the state relevant to CMP after oxide removal. Our goal was to clarify how sliding speed influences tribochemical wear of oxide‑free silicon in humid air and deionized water, providing insights that could enhance CMP efficiency. We employed p‑type Si(100) wafers, removing the native oxide with 40 % HF for 2–3 min followed by ultrasonic cleaning in methanol, ethanol, and DI water. The resulting surface, terminated with Si–H, had an RMS roughness of 0.12 ± 0.02 nm over a 500 × 500 nm area and a static water contact angle of 82 ± 2°, indicating moderate hydrophobicity. Using an AFM (SPI3800N, Seiko), we slid a 1.25 µm radius SiO2 microsphere mounted on a cantilever (k = 10.5–13.8 N/m) against the silicon surface at room temperature, 2 µN load, 200 nm scratch amplitude, and 100 cycles per test. Sliding speeds ranged from 0.08 to 50 µm/s, under 60 % RH or in DI water. Post‑test, wear scars were imaged with a sharp Si3N4 tip (R ≈ 10 nm) in vacuum (<10−3 torr). Cross‑sectional TEM (Tecnai G2, FEI) and Raman (RM2000, Renishaw) analyses quantified subsurface damage and chemical changes, respectively. AFM images (Fig. 1a,b) show clear material removal after 100 cycles in both humid air and DI water. Wear volume decreased logarithmically with speed and plateaued at ~2 × 104 nm3 in air and ~5 × 104 nm3 in water beyond ~8 µm/s. AFM images and cross‑sectional profiles of wear scars at 0.08–50 µm/s in humid air (a) and in water (b). Volume versus speed (c). Load = 2 µN, amplitude = 200 nm, cycles = 100. In air, the Si surface, terminated with Si–H, can adsorb water molecules, facilitating the formation of interfacial Sisubstrate–O–Sitip bonds that enable tribochemical removal. In DI water, the higher water concentration lowers the energy barrier for hydrolysis, yielding more Si–OH groups and consequently higher wear volumes. Under the same 2 µN load, the contact pressure (~1 GPa) is far below silicon’s yield stress (7 GPa). Instead of material removal, hillocks formed (Fig. 2a), originating from stress‑induced amorphization. Hillock volume decreased with speed, indicating incomplete crystalline‑to‑amorphous transformation at high speeds. This behavior differs from the tribochemical wear observed in humid air and water, where removal dominates. Hillock formation in dry air (a). Hillock volume versus speed (b). Load = 2 µN, amplitude = 200 nm. Inset shows a typical hillock cross‑section. High‑resolution TEM of wear scars at 0.08 µm/s and 50 µm/s (Fig. 3) revealed well‑ordered silicon lattices beneath the worn surface, with no amorphization or dislocations. Scar depths were ~11 nm and ~2.3 nm, respectively, supporting a tribochemical mechanism where Sisubstrate–O–Sitip bonds mediate atom removal. High‑resolution TEM of wear scars at 0.08 µm/s (a) and 50 µm/s (b) in humid air. Insets show scar depths of ~11 nm and ~2.3 nm. Raman spectra (Fig. 5) of wear debris displayed pronounced O–Si–O and Si–OH peaks relative to the pristine surface, indicating that dehydration and hydrolysis reactions contribute to tribochemical wear. The relative intensities of these bonds increased with wear, confirming their role in bond formation and breakage during sliding. Raman spectra of (a) original Si and (b) wear debris. Normal load = 1 N, cycles = 2000. We propose that interfacial Sisubstrate–O–Sitip bridges form under mechanical stress and water presence. At low speeds, sufficient contact time allows bridge reformation via dehydration, strengthening the bond and enabling material removal. As speed increases, reduced contact time limits bridge reformation, diminishing the tribochemical reaction. Beyond ~8 µm/s, a dynamic equilibrium between rupture and reformation yields a constant wear volume. Schematic of interfacial states at increasing sliding speed. We demonstrated that tribochemical wear of oxide‑free silicon against SiO2 microspheres decreases logarithmically with sliding speed and stabilizes in both humid air and deionized water. TEM confirms subsurface integrity, while Raman shows concurrent dehydration and hydrolysis. The observed behavior can be modeled by the kinetics of interfacial Sisubstrate–O–Sitip bond formation and rupture. These insights advance the fundamental understanding of silicon CMP and suggest that controlling hydrolysis (e.g., via slurry pH) can optimize polishing efficiency. Atomic force microscope Chemical mechanical polishing Deionized water Root‑mean‑square Transmission electron microscopy Time‑of‑flight secondary ion mass spectrometryAbstract
Background
Methods
Results and Discussion
Sliding Speed‑Dependent Nanowear in Aqueous Environments
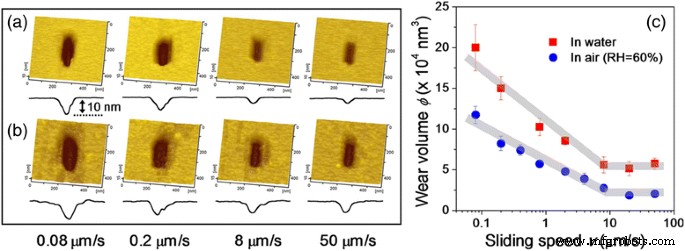
Sliding Speed‑Dependent Nanowear in Dry Air
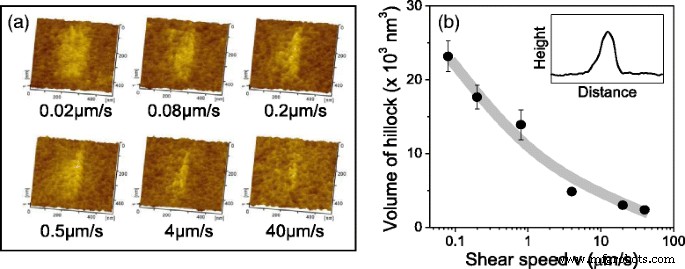
TEM Observation of Wear Tracks

Raman Analysis of Dehydration and Hydrolysis
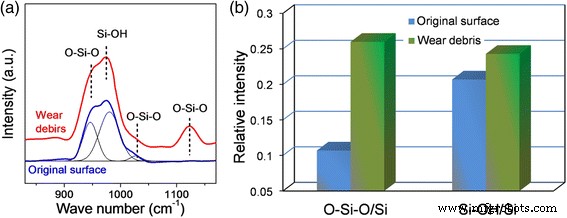
Mechanism of Sliding Speed‑Dependent Tribochemical Wear
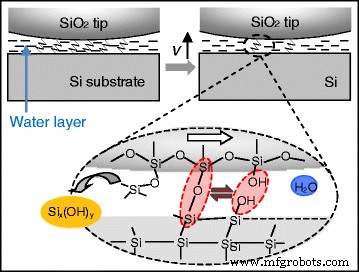
Conclusions
Abbreviations
Nanomaterials
- Silicon: Properties, Production, and Market Outlook
- Nanosilicon: Rapid, Carbon‑Free Hydrogen Generation from Water
- Transparent, Flexible SiOx–Graphene Memory: A Leap Beyond Conventional Flash
- Cu-Catalyzed Growth of Amorphous Silicon Nanowires on SiO₂ by Annealing: A Simple, Contamination‑Free Method
- Raman Mapping of Monolayer Graphene on Silicon Micro‑Ring Resonators: Unveiling Doping and Strain Effects
- Review of Nanostructured Black Silicon Applications
- Unveiling the True Lifetime Distribution of Silicon Nanocrystals: A Combined Time‑Resolved and Frequency‑Resolved Spectroscopic Study
- Ultra‑High Efficiency Silicon Cells with Inverted‑Pyramid Passivation: A Photovoltaic Breakthrough
- Silicon Diodes Explained: Key to Modern Electronics
- Hydraulic Pump Lifespan: How Long Do They Last and How to Extend Their Life



