Optimized Anti‑Solvent Process Yields Fully Covered, Stable FASnI₃ and CsSnI₃ Perovskite Films for Lead‑Free Solar Cells
Abstract
Lead‑free perovskite solar cells based on formamidinium tin triiodide (FASnI₃) and cesium tin triiodide (CsSnI₃) offer a sustainable alternative to conventional lead‑containing devices. In this work, we integrate solvent engineering with an anti‑solvent dripping technique to produce perovskite films of exceptional quality. A 10 mol % SnCl₂ additive is introduced into the FASnI₃ precursor to inhibit Sn⁴⁺ formation, while a 10 mol % SnF₂ additive is employed in the CsSnI₃ solution to suppress Sn²⁺ oxidation. By selecting toluene or chlorobenzene as the anti‑solvent, we achieve films with complete surface coverage and evaluate their oxidative stability under ambient conditions. These findings pave the way for scalable, environmentally benign tin‑based perovskite photovoltaics.
Background
Organic‑inorganic halide perovskites have rapidly advanced the field of photovoltaics, achieving power conversion efficiencies (PCEs) that have risen from 9 % in 2012 to over 22 % in recent years.1–4 The majority of high‑efficiency devices rely on lead‑based compositions such as methylammonium lead iodide (MAPbI₃) and formamidinium lead iodide (FAPbI₃).5–8 However, the intrinsic toxicity of lead restricts commercial deployment.1,9–11 Consequently, researchers have turned to tin as a non‑toxic B‑site cation, leading to the development of lead‑free perovskites like CH₃NH₃SnI₃ and FASnI₃.12–14 Initial reports demonstrated PCEs around 6 % for these tin‑based systems, but rapid Sn²⁺ oxidation and resulting instability have remained significant challenges.15–17
Recent strategies to enhance stability involve incorporating halide additives (SnF₂, SnCl₂, SnBr₂, SnI₂) that can suppress Sn²⁺ oxidation and improve film morphology.18–22 For FASnI₃, SnF₂‑pyrazine complexes and diethyl ether anti‑solvent dripping have yielded smoother, denser films and PCEs exceeding 6 %.23–25 Similar efforts with CsSnI₃ have achieved PCEs of 2–3 % and identified SnCl₂ as a stabilizing additive.26–28 These advances highlight the importance of controlled solvent and additive engineering in producing high‑quality, stable tin‑based perovskite layers.
In this study, we systematically evaluate the impact of anti‑solvent dripping (diethyl ether, toluene, chlorobenzene) and halide additives (SnCl₂ for FASnI₃, SnF₂ for CsSnI₃) on film morphology, crystallinity, and oxidative stability. The goal is to establish a reproducible, scalable process that yields fully covered, defect‑free perovskite films suitable for next‑generation lead‑free solar cells.
Methods
FASnI₃ precursor preparation. A solution was prepared by dissolving 372 mg of SnI₂ and 172 mg of formamidinium iodide (FAI) in 800 µl anhydrous dimethylformamide (DMF) and 200 µl anhydrous dimethyl sulfoxide (DMSO). Ten mol % SnCl₂ was added and the mixture stirred until clear. Spray pyrolysis at 500 °C deposited a compact TiO₂ layer on FTO glass, which was annealed at 500 °C for 15 min. A mesoporous TiO₂ scaffold was spin‑coated at 4 500 rpm for 20 s and annealed at 500 °C for 1 h. FASnI₃ films were then spin‑coated at 4 000 rpm for 60 s in a nitrogen glove box while dripping the chosen anti‑solvent (diethyl ether, toluene, or chlorobenzene). Subsequent annealing at 70 °C for 20 min produced the final perovskite layers.
CsSnI₃ precursor preparation. A 0.6 M solution containing equimolar CsI and SnI₂, with or without 10 mol % SnF₂, was stirred overnight at 70 °C in DMSO. Sixty microliters of this solution were spin‑coated onto the TiO₂ substrate at 4 000 rpm, followed by annealing at 70 °C for 10 min, yielding mirror‑like black perovskite films.
Results and Discussion
Effect of anti‑solvent dripping on FASnI₃ morphology. Figure 1 illustrates SEM images of FASnI₃ films fabricated with different anti‑solvents. Without anti‑solvent, the film shows discontinuous nucleation and numerous pinholes. Diethyl ether produces a net‑like morphology with many holes. In contrast, toluene and chlorobenzene yield highly uniform, dense films with complete coverage. Toluene‑dripped films exhibit slightly larger crystal grains compared to chlorobenzene. These observations are consistent with prior reports and underscore the critical role of anti‑solvent choice in achieving defect‑free films.

SEM images of perovskite films prepared (a) without anti‑solvent dripping, (b) with diethyl ether, (c) with toluene, and (d) with chlorobenzene dripping.
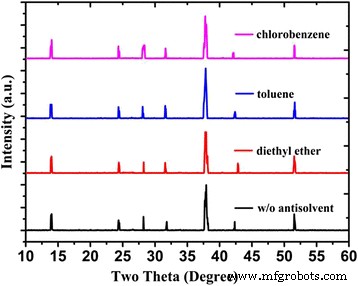
Crystallographic analysis. XRD patterns (Figure 2) confirm that all FASnI₃ films adopt an orthorhombic structure with random orientation, irrespective of the anti‑solvent used. No secondary phases were detected, indicating that the anti‑solvent does not alter the crystalline phase.
XRD patterns of FASnI₃ perovskite films under different anti‑solvent conditions.
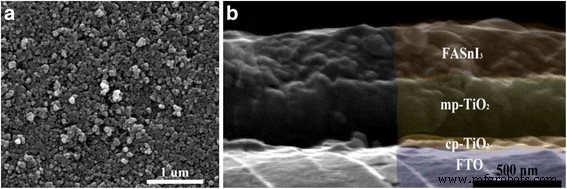
Film thickness and interface. Cross‑sectional SEM (Figure 3) reveals a layered architecture: FTO/compact TiO₂/mesoporous TiO₂/FASnI₃, with an average FASnI₃ thickness of ~250 nm. The sharp interface suggests excellent adhesion and minimal intermixing.
a SEM top view of TiO₂ surface. b Cross‑sectional SEM of the complete stack.
Optical absorption and degradation. Figure 4a shows UV‑Vis spectra of FASnI₃ films with 10 mol % SnCl₂ under various anti‑solvent conditions. All films display an absorption onset at ~900 nm, characteristic of FASnI₃. The absorption intensity varies with anti‑solvent, reflecting differences in film quality. Time‑resolved spectra (Figure 4b) demonstrate that FASnI₃ degrades rapidly in air, with the FA₂SnI₆ absorption peak emerging after ~17 h. This confirms the well‑known oxidation pathway of Sn²⁺ to Sn⁴⁺.
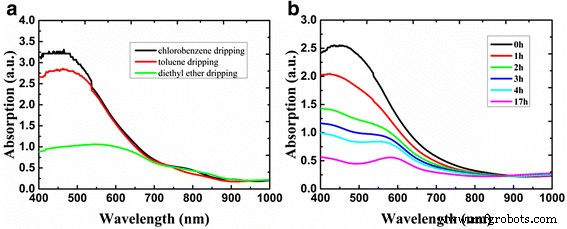
Absorption spectra of FASnI₃ + 10% SnCl₂ films (a) with different anti‑solvents and (b) over time in ambient air.
Device performance. Figure 5a presents a cross‑sectional SEM of a complete FASnI₃ PSC (FTO/cp‑TiO₂/mp‑TiO₂/FASnI₃/Spiro‑OMeTAD/Au). J‑V characteristics (Figure 5b) reveal that toluene and chlorobenzene‑dripped films deliver the highest PCEs, attributable to improved coverage and larger crystal domains that reduce grain‑boundary recombination. Although absolute efficiencies remain modest, the trends clearly illustrate the influence of anti‑solvent choice on device performance.
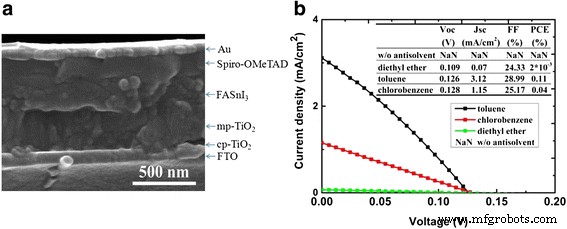
a Cross‑sectional SEM of the completed device. b J‑V curves for FASnI₃ PSCs fabricated with different anti‑solvents.
CsSnI₃ film stability. Without additives, CsSnI₃ solutions appear yellowish, indicating rapid oxidation. Adding 10 mol % SnF₂ darkens the solution, signifying reduced Sn²⁺ oxidation (Figure 6). Time‑dependent UV‑Vis measurements (Figure 7) reveal that CsSnI₃ films without additives degrade within minutes under 57 % RH and 13 °C. Films containing SnF₂ exhibit a slower degradation profile, with a noticeable delay before the onset of oxidation. These results confirm that SnF₂ effectively suppresses Sn²⁺ oxidation and extends film stability.
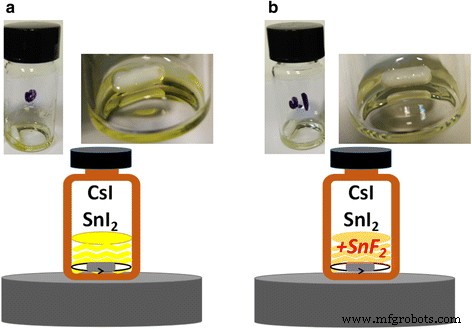
a Pure CsSnI₃ without additives. b CsSnI₃ + 10 mol % SnF₂.
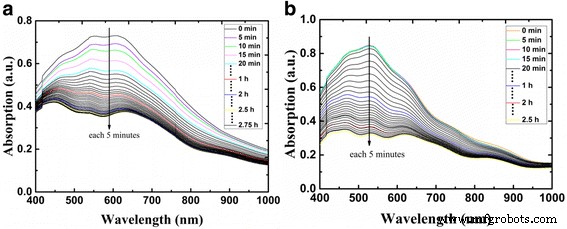
Absorption spectra of (a) pure CsSnI₃ films and (b) CsSnI₃ + 10 mol % SnF₂ films at different times in ambient air.
Conclusions
Our systematic investigation demonstrates that anti‑solvent choice and halide additives critically influence the morphology, crystallinity, and oxidative stability of tin‑based perovskite films. Toluene and chlorobenzene, combined with 10 mol % SnCl₂, produce FASnI₃ layers that are fully covered, defect‑free, and maintain stability for several hours. CsSnI₃ films benefit from 10 mol % SnF₂, which slows oxidation and improves film integrity, although their stability remains limited to minutes under ambient conditions. The most decisive factor for advancing lead‑free perovskite photovoltaics is the suppression of Sn²⁺ oxidation. Our anti‑solvent engineering approach offers a scalable route to high‑quality, stable tin‑based perovskite films, laying the groundwork for efficient, environmentally benign solar cells in the near future.
Nanomaterials
- High‑Efficiency, Low‑Cost Perovskite Solar Cells: Progress, Challenges, and Future Directions
- Cu-Catalyzed Growth of Amorphous Silicon Nanowires on SiO₂ by Annealing: A Simple, Contamination‑Free Method
- Optimizing MAPbI3 Perovskite Solar Cells with Oblique-Angle Sputtered ITO Electrodes
- High‑Efficiency Planar Perovskite Solar Cells via Sequential Vapor‑Grown Hybrid Perovskite Layers
- Optimizing CH₃NH₃PbI₃ Morphology for Enhanced Perovskite Solar Cell Performance
- Efficient Ambient‑Air Fabrication of Mesoporous Perovskite Solar Cells Using N‑Butyl‑Amine‑Enhanced PbI₂ Precursors
- NH4SCN Doping Enhances Grain Size and Lowers Trap States in Triple‑Cation Perovskite Films
- High‑Efficiency 22%+ Perovskite Solar Cells Using Ultra‑Compact FA0.1MA0.9PbI3 Films
- Enhanced Stability and Efficiency of 2D Perovskite Solar Cells Through Bromine Incorporation
- Comprehensive Guide to Sand Testing Methods in Foundries



