Comparative Study of Inkjet‑Printed and Spin‑Coated ZrO₂ Dielectrics for High‑Performance IGZO Thin‑Film Transistors
Abstract
In this study, we engineered a low‑leakage ZrO₂ dielectric using a direct inkjet‑printing process for sputtered indium gallium zinc oxide (IGZO) thin‑film transistors (TFTs). Both spin‑coated and inkjet‑printed ZrO₂ films were fabricated to evaluate their formation dynamics and electrical characteristics. High‑resolution TEM confirmed the homogeneity of the ZrO₂ layers, while XPS revealed a reduced density of oxygen vacancies in the inkjet‑printed films. The inkjet‑printed TFT exhibited a superior saturation mobility of 12.4 cm²/V·s, an I_on/I_off ratio of 10⁶, a turn‑on voltage of 0 V, and only a 1.4 V V_th shift after one hour of positive bias stress (PBS). The low off‑state current stems from the dense, vacancy‑free film. However, the device suffers from a pronounced V_th shift under PBS, attributed to an In‑rich back‑channel that readily adsorbs H₂O and O₂, acting as acceptor traps. These findings highlight the potential of inkjet‑printed oxide dielectrics for high‑performance, scalable oxide TFTs, while emphasizing the need for interface engineering to mitigate bias‑induced degradation.
Background
Metal‑oxide dielectrics have emerged as promising replacements for conventional SiO₂ and SiNx in TFTs, offering high capacitance, minimal defect states, and wide bandgaps that translate into high mobility and ultra‑low off‑current [1–3]. Consequently, vacuum‑deposited oxide dielectrics are widely employed in displays, sensor arrays, and driving circuits [4]. The solution‑processed route—encompassing spin coating, inkjet printing, spray coating, and slit coating—has attracted significant interest for its cost‑effectiveness and scalability [5,6]. Among these, direct inkjet printing is particularly attractive because it enables patterning without photolithography. Nonetheless, inkjet‑printed TFTs typically lag behind their vacuum‑processed counterparts in electrical performance due to challenges such as uncontrolled precursor spreading and interfacial compatibility with the semiconductor layer [7].
The fabrication route profoundly influences the dielectric’s electrical properties. Spin coating is a mature technique that produces films with leakage densities below 10⁻⁶ A/cm² at 1 MV/cm and breakdown fields exceeding 2 MV/cm, yielding TFT mobilities around 10 cm²/V·s. In contrast, inkjet‑printed dielectrics often exhibit leakage densities two orders of magnitude higher (>10⁻⁴ A/cm² at 1 MV/cm) and mobilities below 5 cm²/V·s. Few studies have systematically compared inkjet‑printed and spin‑coated dielectrics, particularly regarding film density, surface roughness, and interfacial homogeneity—key determinants of TFT performance [8,9]. This work addresses that gap by systematically contrasting the film formation processes and electrical outcomes of spin‑coated versus inkjet‑printed ZrO₂ dielectrics on sputtered IGZO TFTs.
We synthesized high‑quality ZrO₂ films via both spin coating and inkjet printing, then evaluated their impact on IGZO TFTs. Spin coating relies on centrifugal force to yield uniform, though potentially dispersive, film coverage, whereas inkjet printing depends on fluid dynamics—gravity, surface tension, and viscoelasticity—to deposit discrete droplets. By optimizing drop spacing, jet velocity, ink composition, and substrate temperature, we controlled the inkjet process. XPS and IV measurements indicated that the double‑layer inkjet‑printed ZrO₂ film exhibited fewer oxygen vacancies compared to the spin‑coated counterpart, leading to superior uniformity and reduced defect density. The inkjet‑printed dielectric achieved lower leakage, higher mobility, a larger on/off ratio, and a higher V_th shift under PBS, albeit with reduced bias stability due to the formation of an In‑rich back‑channel that absorbs atmospheric H₂O and O₂. Our findings demonstrate that direct inkjet printing can produce dense oxide dielectrics, but interface defects must be managed to ensure electrical stability.
Methods
Materials
The ZrO₂ precursor solution was prepared by dissolving 0.6 M ZrOCl₂·8H₂O in a 10 mL mixture of 2‑methoxyethanol (2MOE) and ethylene glycol (2:3 ratio) to achieve the desired surface tension. The solution was stirred at 500 rpm at room temperature for 2 h and then aged for at least 24 h. ITO substrates were cleaned with isopropyl alcohol and deionized water, followed by 100‑W UV ozone treatment (250 nm) to enhance surface wettability. Spin‑coated films were deposited at 5000 rpm for 45 s, while inkjet‑printed films were deposited with a drop spacing of 30 µm and nozzle temperature of 30 °C. Both films were annealed at 350 °C in air for 1 h. A 10‑nm IGZO layer was sputtered using a DC pulsed system (1 mTorr, O₂:Ar = 5%) and patterned with a shadow mask. The IGZO was annealed at 300 °C for 1 h. The channel dimensions were 550 µm × 450 µm (W/L = 1.22). Finally, 150‑nm Al source/drain electrodes were deposited by DC sputtering at room temperature.
Instruments
XPS (ESCALAB250Xi, Thermo‑Fisher) measured the chemical states at a base pressure of 7.5 × 10⁻⁵ mTorr. Cross‑sectional TEM (JEM‑2100F, JEOL) and EDS mapping (Bruker) provided elemental distribution. Capacitance–voltage (C‑V) characteristics were recorded with an Agilent 4284A LCR meter under dark and ambient conditions. Transfer characteristics and leakage currents were measured with an Agilent 4156C semiconductor parameter analyzer, sweeping V_GS from –5 V to +5 V at V_DS = 5 V. Field‑effect mobility was extracted from the saturation region using I_DS = (W µ C_i/2L)(V_GS–V_th)². The relative dielectric constant was calculated from ε_r = (C·d)/(ε_0·S).
Result and Discussion
Figure 1 illustrates the contrasting film formation mechanisms of spin coating and inkjet printing. Spin coating forces droplets to spread uniformly via centrifugal force, producing a homogeneous distribution but leaving voids due to droplet ejection. Inkjet printing, however, relies on the balance between spreading and retraction governed by gravity, surface tension, and ink viscosity. By fine‑tuning drop spacing, jet velocity, ink composition, and substrate temperature, we controlled the inkjet process. UV ozone treatment significantly improved precursor wetting; the 40‑s ozone exposure yielded the best morphology (see Supplementary Fig. S1). Multiple‑layer printing effectively sealed vacancies and enhanced film density, as evidenced by the thickness increase from 45 nm (single layer) to 60 nm (double layer) without a simple additive relationship (Supplementary Fig. S2).
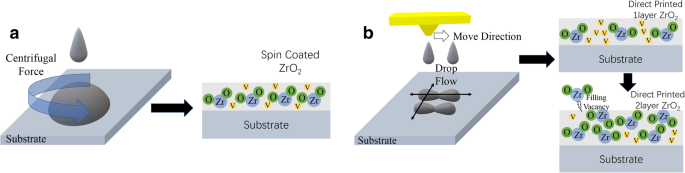
Film formation process of a spin coating and b direct inkjet printing method
The O 1s XPS spectra (Fig. 2) were deconvoluted into three components: metal‑oxygen bonds (V_M‑O, ~529.8 eV), oxygen vacancies (V_O, ~531.7 eV), and weakly bound species (V_M‑OR, ~532.1 eV) [17,18]. The double‑layer inkjet‑printed film (DP2) displayed 81.57 % V_M‑O and the lowest V_O fraction, confirming a denser, vacancy‑free dielectric. AFM (Supplementary Fig. S3) revealed that spin‑coated films had the smoothest surface (R_q = 0.29 nm), while DP1 and DP2 exhibited R_q of 1.05 nm and 0.67 nm, respectively—illustrating the roughness reduction achieved with additional layers.
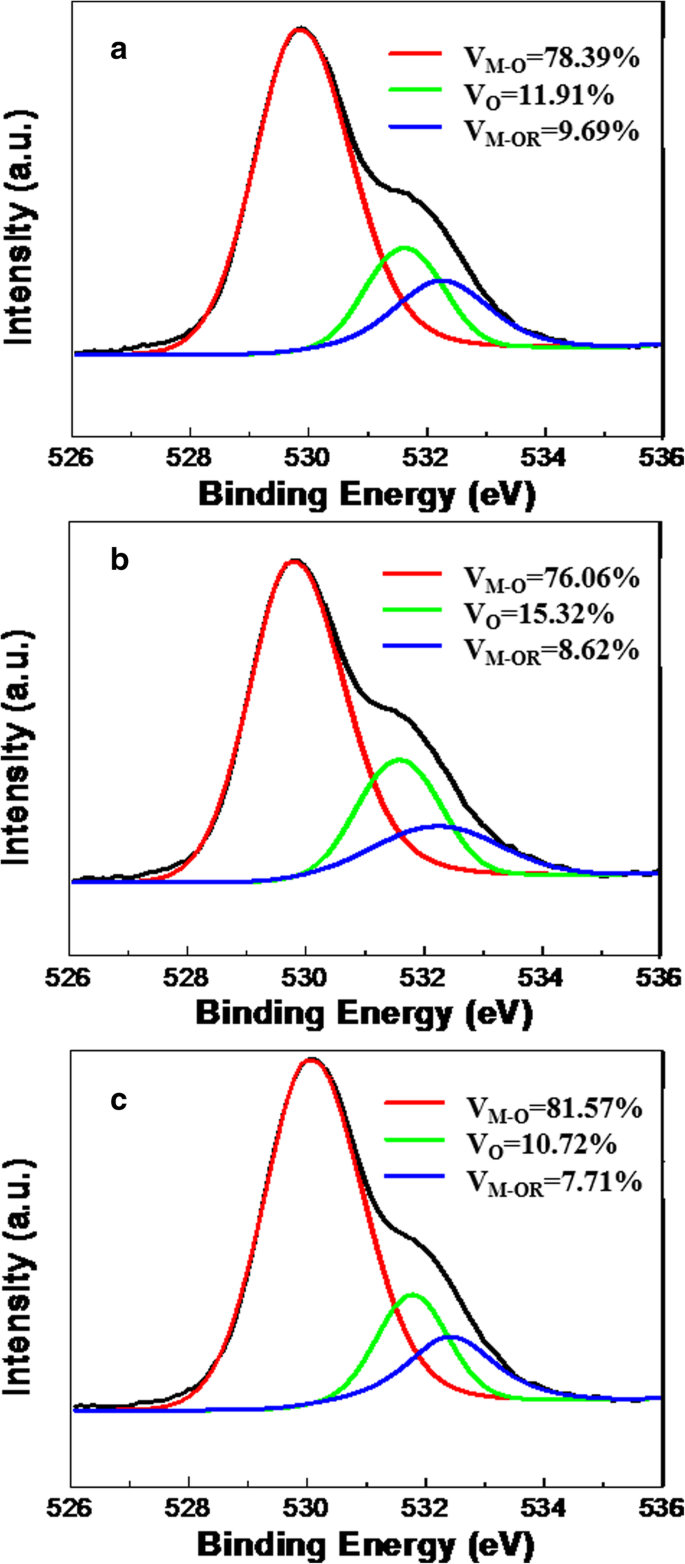
O 1s spectrum of a SC, b DP1‑layer, and c DP2‑layer ZrO₂ film
Electrical testing of Al/ZrO₂/ITO capacitors (Fig. 3) showed that DP1 suffered from high leakage due to abundant vacancies, whereas DP2 achieved a leakage density of 2.4 × 10⁻⁵ A/cm² at 1 MV/cm and a breakdown field > 2.5 MV/cm. Capacitance‑voltage curves (Fig. 4) yielded relative dielectric constants of 19.2 (SC), 20.1 (DP1), and 18.8 (DP2), consistent with the literature value (~18). Hysteresis was minimal for SC and increased for DP1, reflecting the role of film uniformity on trap density.
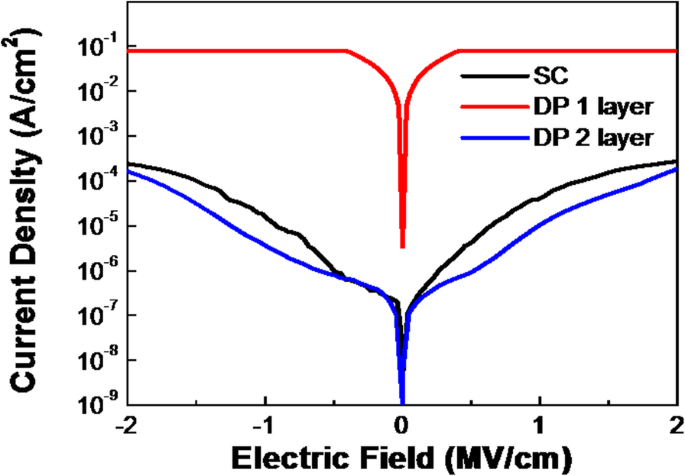
Leakage current density of SC, DP1‑layer, and DP2‑layer ZrO₂ film
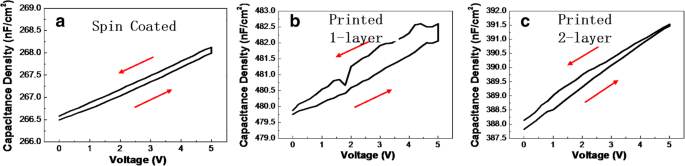
Capacitance density of a SC, b DP1‑layer, and (c) DP2‑layer ZrO₂ film
Transfer characteristics under negative (NBS) and positive (PBS) bias stress (Fig. 5) revealed that DP2‑IGZO TFTs delivered a higher saturation mobility (12.5 cm²/V·s) and I_on/I_off ratio (10⁶) compared to SC‑IGZO (10.2 cm²/V·s, 2 × 10⁵). V_th shifts were negligible after NBS for both devices, indicating good hole‑trap resistance. However, under PBS, SC‑IGZO exhibited a 0.4 V shift, whereas DP2‑IGZO suffered a pronounced 1.2 V shift—attributable to the In‑rich back‑channel absorbing H₂O and O₂, which act as acceptor traps. TEM and EDS (Fig. 6) confirmed the formation of an In‑rich region at the Al/IGZO interface in DP2 devices, leading to increased V_th drift.
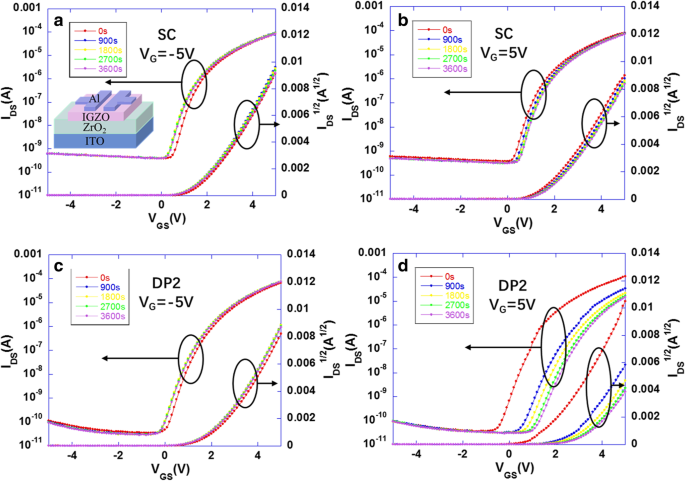
a NBS and b PBS results of SC‑ZrO₂ IGZO TFT. c NBS and d PBS results of DP2‑ZrO₂ IGZO TFT
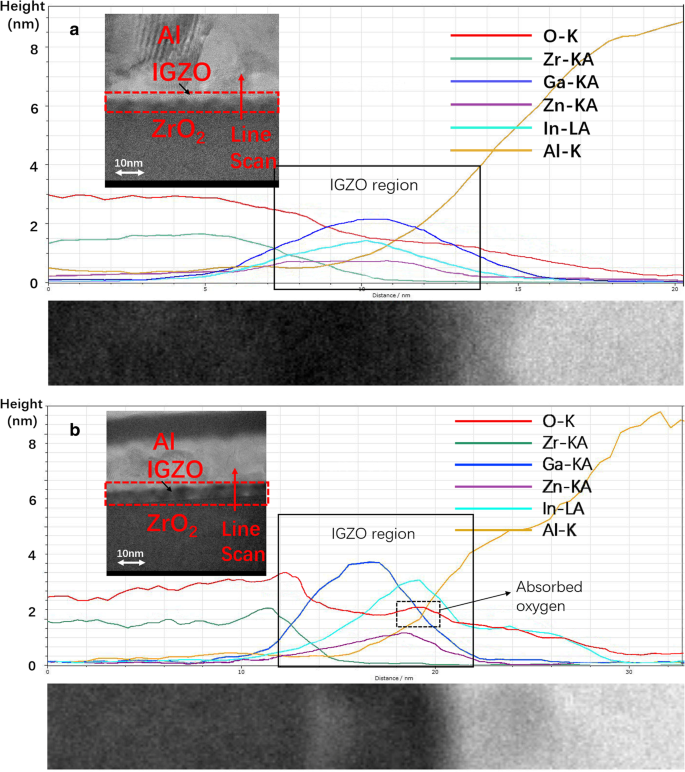
a TEM image and EDS line scanning of SC‑ZrO₂ IGZO TFT. b TEM image and EDS line scanning of DP2‑ZrO₂ IGZO TFT
Band‑diagram analysis (Fig. 7) suggests that while DP2‑ZrO₂ TFTs accumulate more carriers at the static state due to superior insulation, they are more susceptible to acceptor‑like traps under PBS. Adsorbed H₂O and O₂ molecules form hydroxide species that consume electrons, shifting V_th positively. The differential oxygen bond dissociation energies—Zr–O (756 kJ/mol) vs. Ga–O (364 kJ/mol) and In–O (336 kJ/mol)—favor oxygen incorporation into ZrO₂, leaving In and Zn to migrate toward the back‑channel where they react with ambient species. Strategies such as top‑gate architectures, interfacial passivation layers, or post‑deposition treatments can mitigate PBS‑induced degradation and are promising avenues for future research.
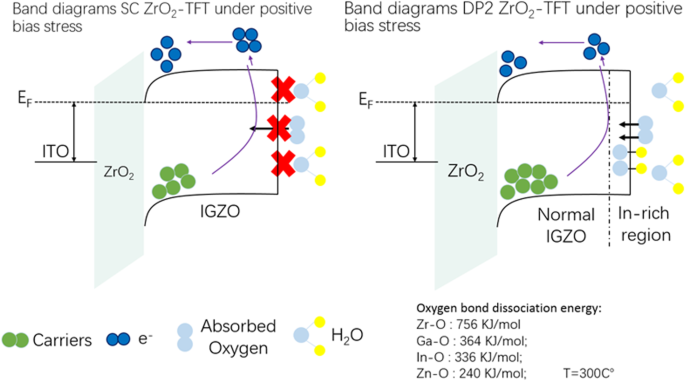
Band diagrams of SC‑ZrO₂ TFT and DP2‑ZrO₂ TFT under positive bias stress
Conclusion
We successfully fabricated a dense, low‑leakage, inkjet‑printed ZrO₂ gate insulator using a multi‑layer printing strategy that obviates the need for photolithographic patterning, making it attractive for large‑area, cost‑effective production. The inkjet‑printed film exhibited a higher density and lower oxygen vacancy concentration than its spin‑coated counterpart, as confirmed by XPS and TEM. Its leakage density (2.4 × 10⁻⁵ A/cm² at 1 MV/cm) and breakdown field (> 2 MV/cm) surpassed spin‑coated films. The resulting IGZO TFT achieved a saturation mobility of 12.4 cm²/V·s, an I_on/I_off ratio of 10⁶, a turn‑on voltage of 0 V, and a 1.2‑V V_th shift after one hour of PBS. TEM and EDS revealed an In‑rich back‑channel that absorbs atmospheric H₂O and O₂, acting as acceptor traps and causing the observed V_th drift. Despite this stability challenge, the performance metrics demonstrate that inkjet printing can deliver high‑quality oxide dielectrics. With further interface optimization—such as passivation layers or alternative gate architectures—inkjet‑printed dielectrics hold great promise for scalable, high‑performance oxide TFTs.
Abbreviations
- 2MOE:
Methoxyethanol (solvent)
- AFM:
Atomic force microscope
- Al:
Aluminum
- DP1/2:
Direct‑printed 1/2 layer
- EDS:
Electronic differential system
- H₂O:
Water molecule
- IGZO:
Indium gallium zinc oxide (oxide semiconductor)
- ITO:
Indium tin oxide (electrode)
- O 1s:
Oxide 1s atomic orbital
- O₂:
Oxygen molecule
- PBS/NBS:
Positive/negative bias stress (test mode)
- SC:
Spin coated
- SiNₓ:
Silicon nitride (dielectric)
- SiO₂:
Silicon dioxide (dielectric)
- TEM:
Transmission electron microscope
- TFT:
Thin‑film transistor
- UV:
Ultraviolet
- V_M‑O:
Percentage of metal‑oxygen bond
- V_M‑OR:
Percentage of metal‑organic bond
- V_O:
Percentage of oxide vacancy bond
- V_th:
Threshold voltage
- XPS:
X‑ray photoelectron spectroscopy
- ZrO₂:
Zirconia (oxide dielectric)
- ZrOCl₂·8H₂O:
Zirconium oxychloride octahydrate (material)
Nanomaterials
- Plastic Packaging Film Production: A Comprehensive Overview of Processes and Materials
- Cu-Catalyzed Growth of Amorphous Silicon Nanowires on SiO₂ by Annealing: A Simple, Contamination‑Free Method
- Highly Sensitive Non‑Enzymatic Glucose Sensor Based on Mesoporous NiO Nanopetals Grown on FTO
- Low‑Temperature ALD of In₂O₃ Nanofilms for High‑Performance Thin‑Film Transistors
- Monoclinic V1−x−yTixRuyO₂ Thin Films: Superior Thermal Sensitivity for Microbolometers
- Flame‑Assisted Printing Enables Large‑Area Nanostructured Oxide Thin Films for Electrochromic Devices
- Energiron HYL Process: Advanced Direct Reduction of Iron Ore to High-Quality DRI
- Midrex: Leading Gas‑Based Direct Reduction of Iron Ore
- Direct Iron Ore Smelting (DIOS): Efficient, Coke‑Free Ironmaking
- PERED Direct Reduction Technology – Accelerated, Energy‑Efficient Iron Production



