Reversible Electrochemical Control of Photoluminescence in CdSe/ZnS Core/Shell Quantum Dot Films
Abstract
Semiconductor quantum dots (QDs) underpin a broad range of optoelectronic devices, from LEDs to photovoltaic cells. Electrochemical tuning offers a precise probe of their electrical and optical behavior. In this study, we examined how applying a negative electrochemical potential modulates the photoluminescence (PL) spectra of core/shell CdSe/ZnS QD films. We observed that the core emission shifts to longer wavelengths while the surface emission moves to higher energies. These spectral changes arise from (i) electrostatic expansion of the excitonic wave function due to nonuniform adsorption of cations on the QD surface, and (ii) occupation of lower surface trap states by injected electrons, which forces photoexcited carriers to higher surface states and yields a blueshift. Importantly, both the spectral shifts and the accompanying PL quenching are fully reversible upon resetting the potential.
Background
Colloidal semiconductor QDs attract attention for their size‑tunable optical properties, a direct consequence of quantum confinement that allows the bandgap to be engineered with nanometer‑scale dimensional control[7]. Their high surface‑to‑volume ratio, however, introduces surface‑trap states that can capture excitons, thereby diminishing PL efficiency[8‑10]. In addition, charged QDs host trions—negative or positive—whose fast nonradiative Auger recombination further quenches emission[10‑13]. Trion photophysics are marked by spectral shifts, shortened lifetimes, and stochastic blinking[14‑16].
Understanding and manipulating these quenching pathways is crucial for both fundamental insights and practical device optimization. Electrochemical electron injection provides a controllable means to modulate QD charge states, enabling reversible control over PL “on” and “off” states[18‑22] and accurate in‑situ determination of trap densities[23‑24] and heterojunction offsets[25]. Parallel strategies involve ionic adsorption: anionic adsorption can irreversibly blueshift absorption due to size or structural changes[26], while reversible blueshifts result from anion‑enhanced quantum confinement[27]. Yet, the effect of cationic adsorption on core confinement and the evidence for electron‑induced surface‑state shifts have remained unexplored.
Here we report reversible core redshifts driven by cation adsorption and surface blueshifts caused by electron injection into surface traps, both controllable electrochemically.
Methods/Experimental
Core/shell CdSe/ZnS QDs (center emission 600 nm) were sourced from Sigma‑Aldrich and stabilized with octadecylamine ligands. A ~300 nm thick film was spin‑coated onto cleaned ITO substrates, treated in 10 mM 1,7‑diaminoheptane methanol for 20 s, and cross‑linked by baking at 70 °C for 30 min. The electrochemical cell comprised a Pt counter electrode, an Ag wire quasi‑reference electrode, and the ITO working electrode, with 0.1 M TBAP in DMF as electrolyte. The Ag reference was calibrated against the Fc/Fc⁺ couple (offset ≈ 65 mV vs SHE). The applied potential between ITO and Ag set the QD film’s Fermi level. Steady‑state and time‑resolved PL were recorded simultaneously using an Ocean Optics 4400 fiber‑optic spectrometer and a TCSPC system (180 ps resolution) under 375 nm, 60 ps pulse, 20 MHz excitation.
Results and Discussion
Cyclic Voltammograms and Absorption Spectra
Figure 1 displays the cyclic voltammogram of the CdSe/ZnS QD film (solid) and bare ITO (dashed) at 100 mV s⁻¹. A pronounced reduction peak at −1.7 V corresponds to electron injection into the 1Sₑ ground state, leading to bleaching of the 1S₃/₂→1Sₑ and 2S₃/₂→1Sₑ transitions (Fig. 2). Partial bleaching at −1.6 V indicates single‑electron occupation of 1Sₑ, while the full bleaching at −1.7 V confirms complete filling. The charged QDs also exhibit a Stark‑shifted redshift in absorption relative to the neutral film. This bleaching recovers instantly when the potential is reset to 0 V.
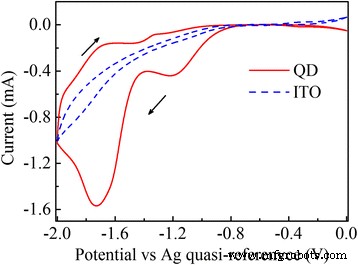
Cyclic voltammogram of the CdSe/ZnS QD film (solid) versus ITO (dashed) at 100 mV s⁻¹. The −1.7 V peak marks charging of the 1Sₑ state, while the −1.2 V peak indicates charging of surface trap states. Scan direction arrows are shown. The Ag quasi‑reference is offset by 65 mV versus SHE.
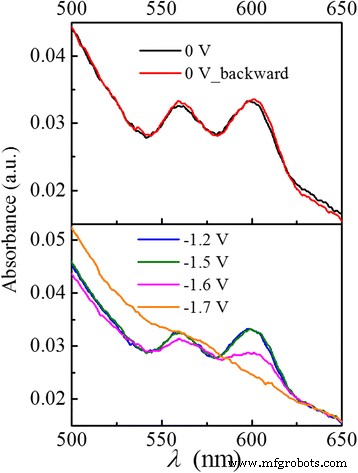
Absorption spectra under representative applied potentials: 0 V (black), −1.2 V (blue), −1.5 V (green), −1.6 V (pink), −1.7 V (orange), and 0 V during reverse scan (red). The two peaks correspond to 1S₃/₂→1Sₑ and 2S₃/₂→1Sₑ transitions. Bleaching appears above −1.6 V and recovers immediately when the potential returns to 0 V.
Reduction peaks around −0.9 V and −1.2 V do not produce bleaching, indicating that electrons populate surface trap states rather than excitonic levels.
Time‑Resolved/Steady‑State PL Under Electrochemical Control
Simultaneous steady‑state and time‑resolved PL were recorded at 0, −0.9, −1.2, and −1.6 V (Fig. 3a,b). As the potential becomes more negative, PL intensity drops dramatically, accompanied by a progressive redshift of the core emission. The spectra recover fully within ~1800 s upon returning to 0 V. The line shape also evolves, reflecting distinct emission components (Fig. 3c).
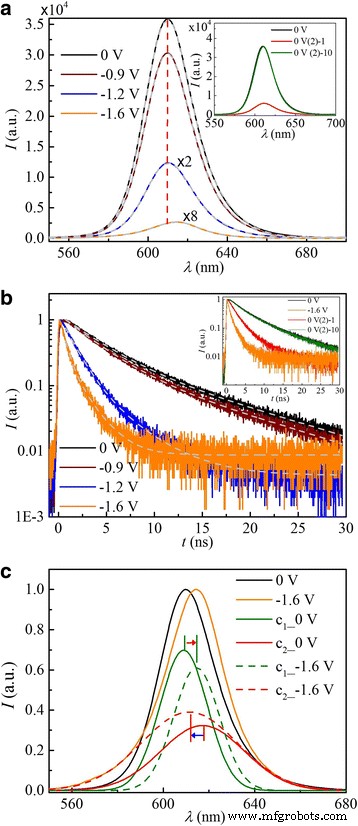
Representative spectra at selected potentials. a Steady‑state PL; b PL decay dynamics. The QDs exhibit high PL quantum yield and slow decay at 0 V (black). At −0.9 V (wine) and −1.6 V (orange), PL is quenched and redshifted. Immediate partial recovery is seen at 0 V (2) in the inset, with full recovery after ~1800 s (inset, 10). c Normalized PL spectra at 0 V (black) and −1.6 V (orange), each fitted to two Gaussian components (c₁, c₂). The black curves represent the 0 V components, while the dashed curves show the −1.6 V components.
Excitonic Emission and Quenching Mechanisms
Each steady‑state PL spectrum fits well to a double Gaussian (Fig. 3a), confirming two distinct emitting states. At 0 V, the core emission peaks at 609 nm (FWHM 14 nm), while the surface emission centers at 617 nm (FWHM 27 nm). The broader surface peak reflects a distribution of trap energies[31‑33]. Comparing 0 V and −1.6 V spectra (Fig. 3c), the core peak shifts to longer wavelengths, whereas the surface peak moves to higher energies.
Time‑resolved data, fitted to bi‑exponential decay (Fig. 3b), reveal lifetimes of 4.2 ns (core) and 15.2 ns (surface) at 0 V. These values are consistent with literature[32‑34] and reflect charge transfer to surface or interface traps[35‑36].
Plotting potential‑dependent parameters (Fig. 4) shows that at −0.9 V, the core emission decays faster and redshifts, indicative of cation adsorption on the QD surface. Electrons injected at this potential occupy surface traps; the accompanying cations act as counter‑ions and charge acceptors, driving exciton dissociation and PL quenching. At −1.6 V, the 1Sₑ state holds a single electron, forming a negative trion; rapid Auger recombination and enhanced charge transfer to adsorbed cations almost completely extinguish PL.
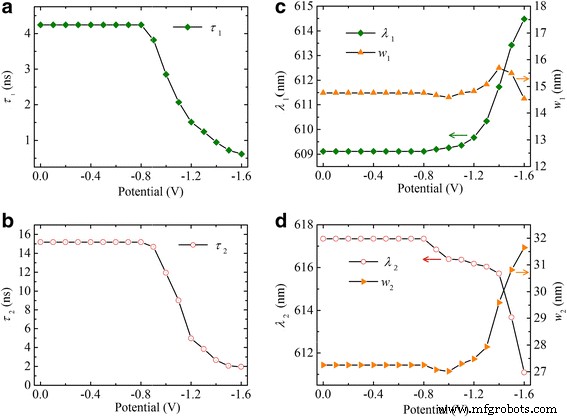
Electrochemical potential‑dependent fitting parameters for core (a,c) and surface (b,d) emission. Decay times of both states decrease at −0.9 V. The core emission redshifts (c, green squares), while the surface emission blueshifts (d, red circles). Orange triangles indicate the FWHM of each component.
Previous work showed anionic adsorption compresses the electron wavefunction, yielding a blueshift[27]. In contrast, cationic adsorption expands the wavefunction, reducing quantum confinement and shifting core emission to longer wavelengths (Fig. 5a). This nonuniform cation distribution electrostatically enlarges the effective QD size.
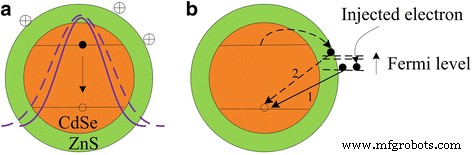
Schematic of (a) cation‑induced expansion of the excitonic wavefunction (solid violet curve = no adsorption, dashed violet curve = with adsorption) and (b) modulation of surface emission by injected surface electrons. As negative potential increases, lower surface states become populated, forcing photoexcited carriers to higher traps and producing a blueshift.
The spatial distribution of surface charges governs the electronic response: a uniform spherical shell (radius > Bohr radius) leaves the wavefunction unchanged, whereas an inhomogeneous distribution alters it. In this study, the nonuniform cation adsorption is assumed, as a uniform distribution has not been observed experimentally.
Surface emission lifetimes and peak positions (Fig. 4b,d) further confirm that increasing negative potential fills lower surface traps, shifting emission to higher energies and shortening decay times—consistent with Auger processes in multi‑electron traps.
Conclusions
Electrochemical control provides a powerful platform to dissect and reverse the photophysical behavior of QD films. We have demonstrated that applying a threshold potential of −0.9 V initiates PL quenching, accompanied by a core redshift and surface blueshift, all of which recover within ~1800 s when the potential returns to zero. The core redshift originates from electrostatic expansion of the exciton due to cation adsorption, whereas the surface blueshift reflects electron occupation of lower surface states, driving photoexcited carriers into higher traps. These reversible shifts offer a route to dynamic tuning of QD emissive properties for future optoelectronic applications.
Nanomaterials
- Phosphine‑Free Synthesis of Reabsorption‑Suppressed ZnSe/CdS/ZnS Core–Shell Quantum Dots and Their Application in Sensitive CRP Detection
- Enhancing Photovoltaic Efficiency by Detecting Spatially Localized Excitons in Self‑Organized InAs/InGaAs Quantum Dot Superlattices
- Eco-Friendly Synthesis of InP/ZnS Core/Shell Quantum Dots for High-Performance Heavy-Metal-Free LEDs
- Significantly Boosted Solar Cell Efficiency Using GaAs/InAs Nanowire–Quantum Dot Hybrid Arrays
- Restoring Luminescence of Aged CdSe/ZnS‑Cys Quantum Dots via TPPS4‑Induced Disaggregation
- Highly Efficient, Cd‑Free Cu‑Doped ZnInS/ZnS Core–Shell Nanocrystals: Tunable Synthesis and Superior Photophysical Performance
- High‑Performance Dual‑Emissive Mn‑Doped InP/ZnS Quantum Dots with 78 % Photoluminescence Quantum Yield: A Growth‑Doping Approach
- Thermal Annealing Enhances Emission in InP/ZnS Quantum Dot Films: Photoluminescence Insights
- High‑Performance 1550‑nm InAs/GaAs Quantum‑Dot SESAM with Short‑Period Superlattice Cap for Femtosecond Fiber Lasers
- Enhanced Photon Absorption in Quantum Dot Infrared Photodetectors via Metal Nanohole Array Surface Plasmons



