Nanomaterials in Neurological Disease Management: Current Status and Future Directions
Abstract
Nanomaterials (NMs) possess unique physicochemical traits that enable precise drug delivery across the blood–brain barrier (BBB), offering enhanced efficacy and safety for neurological therapies. Engineered NMs—including metal chelators, gold and silver nanoparticles, micelles, quantum dots, polymeric and lipid NPs, microparticles, carbon nanotubes, and fullerenes—have been applied to improve drug delivery, monitor therapeutic responses, enable early diagnosis, and advance neuro‑engineering strategies. BBB regulation limits many drugs from reaching the central nervous system (CNS), hindering treatment of Alzheimer’s disease (AD), Parkinson’s disease (PD), multiple sclerosis, amyotrophic lateral sclerosis (ALS), and primary brain tumours. Environmental metal exposure, aging, metabolic disorders, and genetic mutations further exacerbate neurodegeneration. This review examines the most widely used metal chelators, NMs/NPs, and discusses current progress and future challenges in their application for neurological disease management.
Review
Background
Nanomaterials (NMs), defined by sizes of 1–100 nm, span composite, dendrimer, carbon‑based, and metal‑based categories (e.g., quantum dots, nanosilver, nanogold, cerium, titanium, iron, and zinc oxides). Precision synthesis of these nanoparticles has expanded their use across drug‑gene delivery, disease management, pharmaceuticals, cosmetics, food, photonic crystals, coatings, catalysis, bioremediation, plant growth, and materials science [1–12].
Commercial production has surged, with ~3,000 tons of TiO₂ NPs annually, over 50% of which are incorporated into sunscreens [13,14]. Silver and gold NPs are widely adopted in medicine, diagnostics, sensors, and pharmaceuticals [2,11,15–18]. Iron and iron‑oxide NPs serve cancer therapy, drug delivery, MRI, and water purification [11]. Platinum NPs function as antioxidants and catalysts [10,19], while palladium NPs are used in catalysis and cancer therapy [10].
In recent years, NMs have emerged as nanomedicines for diagnosing and treating neurological disorders. Engineered NMs enhance drug bioavailability, evade immune defenses, and prolong systemic circulation [20–22]. Their ability to interact at the molecular level allows tailored responses to cellular microenvironments, reducing adverse effects and enabling precise therapeutic action [21].
Neurological disorders affect the CNS and PNS, causing speech, swallowing, breathing, and learning deficits. The BBB restricts most drugs from reaching the CNS, necessitating novel delivery strategies. Nanotechnology offers solutions—nanotubes, nanospheres, nanogels, nanoemulsions, nanocarriers, polymeric and lipid NPs, micelles, solid lipid carriers, liquid crystals, liposomes, microemulsions, and hydrogels—to achieve targeted, efficient delivery and diagnosis [22].
Current research focuses on developing nanomedicines that cross the BBB to treat AD, PD, ALS, MS, neurological tumours, and ischemic stroke [23]. For example, AD is characterized by memory loss, lexical impairment, and neuronal death; prevalence rises sharply with age (60–85 years), obesity, diabetes, and cardiovascular disease, with >90% of cases being sporadic and driven by APOE ε4 allele [24–28]. Environmental metal exposure (e.g., CeO₂, TiO₂) exacerbates AD and PD via ROS generation and neurotoxicity [33–35]. Gold NPs, combined with electromagnetic fields, can induce dopamine neuron generation for PD therapy [36].
BBB and blood–cerebrospinal fluid barrier (BCSFB) limit drug entry; only lipophilic molecules <600 Da or transporter‑mediated substrates cross efficiently [20,37–40]. Nanoparticles can be engineered to exploit receptor‑mediated transcytosis, endocytosis, or passive diffusion, enhancing CNS delivery without toxicity [42,50,51,102].
Stem cell therapies—neural stem cells (NSCs) and mesenchymal stem cells (MSCs)—hold promise for neuroregeneration, but efficient CNS targeting remains a hurdle. Nanoparticles, such as dextran‑coated magnetic NPs, improve MSC homing and functional recovery in ischemic stroke models [44–46].
This review emphasizes frequently used metal chelators, NMs/NPs, and their status as drug‑delivery vehicles for neurological disease management.
Neurological Disorders and Management
Key CNS challenges include lack of smart diagnostics and poor drug permeability across the BBB. Various NMs/NPs demonstrate promising applications for AD, PD, ALS, MS, tumours, and stroke, as illustrated in Fig. 1.
Different types of NPs and their application in neurological disorder treatment and management
Alzheimer’s Disease
AD currently affects >35 million people, projected to exceed 50 million by 2050. Symptomatic therapy relies on cholinesterase inhibitors and NMDA antagonists, but nanotechnology offers superior diagnostic and therapeutic avenues. Nanoparticles (e.g., polymeric, micellar, liposomal, lipid, and solid lipid NPs) can bind amyloid‑β (Aβ) plaques, creating a “sink effect” and facilitating clearance. Ultra‑sensitive NP‑based biosensors, immunosensors, and scanning microscopy enable early Aβ detection [46].
Aβ aggregation and neurofibrillary tangles are central to AD pathology (Fig. 2). Therapeutic NPs designed to cross the BBB employ ligand conjugation (e.g., thiamine, transferrin) or surfactant coating (polysorbate 80) to exploit receptor‑mediated transcytosis [49–51].
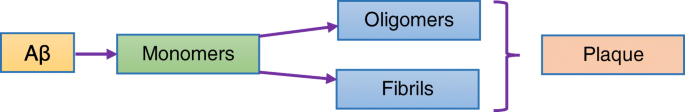
Plaque formation from amyloid‑β protein
Nanoparticle strategies include: 1) radio‑labeled ¹²⁵I‑clioquinol‑loaded polymeric NPs for PET imaging; 2) magnetic iron‑oxide NPs for targeted delivery; 3) gold‑NPs functionalized with lipoic acid‑PEG to inhibit Aβ aggregation; 4) solid lipid NPs (SLNs) to solubilize lipophilic drugs and enhance BBB penetration; 5) rivastigmine‑loaded liposomes and cell‑penetrating peptide‑modified liposomes to improve brain distribution [57–60].
Polysorbate 80‑coated poly(n‑butyl cyanoacrylate) and PEGylated PLA NPs conjugated with BBB‑targeting peptides (TGN, QSH) demonstrate dual‑functional delivery of therapeutic agents directly to Aβ plaques in AD mouse models [61–62].
Metal chelation is another therapeutic axis; excess Cu²⁺, Zn²⁺, and Fe³⁺ accumulate in plaques, promoting oxidative stress. Chelators such as deferiprone, D‑penicillamine, and 5‑chloro‑7‑iodo‑8‑hydroxyquinoline disrupt metal‑Aβ interactions, reducing aggregation and toxicity [63–68,128,129]. Encapsulation of these chelators in PEG‑coated or polysorbate 80‑coated NPs improves bioavailability and BBB crossing [129].
Natural antioxidants (curcumin, EGCG, quercetin) exhibit anti‑oxidative, anti‑inflammatory, and metal‑chelating properties, but suffer from poor solubility and rapid clearance. Liposome‑encapsulation or nano‑formulation (e.g., curcumin–phospholipid conjugates, quercetin‑loaded liposomes) enhances BBB penetration and neuronal protection in AD models [69–70,130–133].
Gold NPs, especially under microwave or laser irradiation, can photothermally destabilize Aβ fibrils, reducing plaque burden without adverse effects [176–178].
Parkinson’s Disease
PD affects ~1 in 100 individuals >65 years, causing motor deficits through dopaminergic neuron loss and α‑synuclein aggregation. Nanomaterials enable neuroprotection, regeneration, and targeted drug delivery across the BBB. Gold‑ and TiO₂‑incorporated nanotube arrays function as photoelectrochemical sensors for α‑syn detection [134]. Catalase‑loaded PEI NPs mitigate neuroinflammation, while α‑syn‑conjugated polybutylcyanoacrylate NPs facilitate protein clearance [23,135–136].
Amyotrophic Lateral Sclerosis
ALS involves loss of upper and lower motor neurons, often associated with mutant SOD1 aggregates. Gold NPs functionalized with SOD or fullerene nanotubes provide antioxidant protection and serve as colorimetric detection platforms for ALS biomarkers [138–139]. Riluzole, a glutamate antagonist, can be efficiently delivered via carbon NPs, enhancing therapeutic concentration at disease sites [140–141].
Multiple Sclerosis
MS disrupts CNS signaling through demyelination and neuroinflammation. Fullerenes combined with NMDA antagonists, PEGylated PLA or PCL‑PEG NPs, and catalase‑loaded poly(ethyleneimine) NPs improve drug efficacy and reduce lesion severity in animal models [142–146].
Neurological Tumours
Brain tumours benefit from passive targeting of <100‑nm NPs exploiting the enhanced permeability and retention effect. Folate‑decorated polymeric NPs enhance tumour accumulation, while PEG‑PLGA NPs loaded with paclitaxel extend survival in glioma models [143–145].
Ischemic Stroke
Ischemic stroke remains a leading cause of death worldwide. Nanomaterials—especially single‑walled carbon nanotubes (SWCNTs) functionalized with amine groups—enable imaging, targeted drug delivery, and neuroprotection, improving motor recovery in preclinical studies [147–150].
Metal Chelators and NMs/NPs Used in Neurological Disease Management
Metal Chelators
Multidentate chelators (e.g., desferrioxamine, penicillamine) form stable, water‑soluble complexes that can be cleared from the CNS, mitigating metal‑induced neurotoxicity. Elevated Cu²⁺, Zn²⁺, and Fe³⁺ concentrations are reported in AD brains versus normal tissue (Cu: 390 vs. 70 µM; Zn: 1055 vs. 350 µM; Fe: 940 vs. 340 µM) [63,152–153].
Nanomaterials
Engineered NMs overcome BBB limitations through receptor‑mediated endocytosis, surfactant coating, and PEGylation. Polylactic acid, poly(lactic‑co‑glycolic acid), polycaprolactone, chitosan, gelatin, and polybutyl cyanoacrylate are common biodegradable polymers that can be surface‑modified to enhance CNS delivery and reduce immune clearance [39,154,157].
Polymeric Nanoparticles
Polymeric NPs (10–100 nm) exist as nanocapsules (core‑shell) or nanospheres (homogeneous matrix). Their degradable polymer matrices (e.g., PBCA, PLGA) protect payloads, allow tunable release, and facilitate BBB crossing via polysorbate 80 coating that adsorbs apolipoprotein E [154,161–163]. Growth factor delivery (IGF, bFGF, NGF) is challenging due to enzymatic degradation; polymeric NPs provide protection and targeted release, improving memory and learning in AD and MS models [165–167].
Solid Lipid Nanoparticles
SLNs are solid at room temperature, enabling stable encapsulation of lipophilic drugs. Polysorbate 80 coating promotes apolipoprotein binding and BBB penetration, while PEGylation extends circulation time. SLNs have shown promise in delivering acetylcholinesterase inhibitors (donepezil, galantamine) and enhancing bioavailability in AD models [40,171].
Liposomes
Biocompatible liposomes can encapsulate hydrophilic or hydrophobic agents, evade reticuloendothelial clearance, and fuse with neuronal membranes. PEGylated liposomes increase half‑life, while lipid composition (e.g., DHA, phosphatidylcholine) modulates membrane fluidity and promotes non‑amyloidogenic APP processing, activating PI3K/Akt pathways and reducing apoptosis [172–175].
Gold Nanoparticles
Gold NPs offer excellent biocompatibility and surface functionalization flexibility. They can disrupt Aβ fibrils via photothermal or photochemical mechanisms, enhancing plaque clearance without neurotoxicity. Gold NPs conjugated with metal‑chelating ligands or anti‑α‑syn antibodies further improve therapeutic outcomes in AD and PD models [115,176–178].
Microparticles
Microparticles (0.1–1 µm) serve as controlled‑release vehicles for FDA‑approved drugs like donepezil. PLGA microparticles implanted subcutaneously achieve steady plasma levels for weeks, reducing gastrointestinal side effects and improving cognition in AD models [181,182].
Carbon Nanotubes and Fullerenes
Carbon nanotubes (CNTs) exhibit high strength, conductivity, and low immunogenicity, making them suitable for drug delivery, imaging, and neuroprotection in AD, PD, and stroke. Fullerene derivatives (e.g., C₆₀HyFn) provide antioxidant and neuroprotective effects, mitigating chronic alcohol‑induced CNS damage and potentially modulating neurotransmitter metabolism [188–190].
Conclusions
Neurological disorders uniformly involve CNS dysfunction, with AD exemplifying neuronal loss and plaque deposition. The BBB remains the paramount obstacle for effective therapy. Nanomaterials—through engineered size, surface chemistry, and ligand conjugation—offer a promising platform to cross the BBB, deliver drugs, and modulate disease pathways with minimal toxicity. While preclinical evidence supports the utility of NMs in diagnostics, drug delivery, and stem‑cell therapies, comprehensive toxicity profiling and scalable manufacturing are essential before clinical translation.
Abbreviations
- AD:
Alzheimer’s disease
- ALS:
Amyotrophic lateral sclerosis
- APP:
Amyloid precursor protein
- Aβ:
Amyloid‑β
- BBB:
Blood‑brain barrier
- CNS:
Central nervous system
- CNT:
Carbon nanotube
- DHA:
Docosahexaenoic acid
- FDA:
Food and Drug Administration
- LCs:
Liquid crystals
- MEs:
Microemulsions
- MS:
Multiple sclerosis
- MPs:
Microparticles
- MSCs:
Mesenchymal stem cells
- NFT:
Neurofibrillary tangles
- NMs:
Nanomaterials
- NPs:
Nanoparticles
- PBCA:
Poly (butyl cyanoacrylate)
- PD:
Parkinson’s disease
- PLGA:
Poly(d,l‑lactic‑co‑glycolic acid)
- ROS:
Reactive oxygen species
- SLN:
Solid lipid nanoparticle
- SOD:
Superoxide dismutase
- SWCNTs:
Single‑walled carbon nanotubes
Nanomaterials
- Tantalum Products Explained: Powder, Wire, Capacitors & Their High-Tech Applications
- 13 Types of Refractory Materials and Their Key Industrial Applications
- Gold Nanoparticles: Advancing Diagnostic and Therapeutic Applications in Medicine – A Comprehensive Review
- Advances in Synthesis and Applications of Silver Nanostructures
- How Nanoparticle Properties Drive Their Toxicity: A Comprehensive Review
- Biomedical Applications of Gold Nanoclusters: Recent Advances and Future Directions
- Large-Scale Dendritic Silver Nanostructures: Controlled Morphology for Enhanced Catalysis and SERS Performance
- Eco‑Friendly, Cost‑Effective Synthesis of Tin Oxide Nanoparticles: A Comprehensive Review of Methods, Formation Mechanisms, and Applications
- Essential PCB Components & Their Applications: How They Drive Modern Electronics
- Exploring Hot and Cold Chamber Die Casting: Types, Applications, and Advantages



