Zirconia Nanoparticles Trigger ROS‑Mediated Cytotoxicity and Suppress Osteogenic Differentiation in 3T3‑E1 Osteoblasts
Zirconia (ZrO2) nanoparticles are increasingly employed in biosensors, cancer therapy, implants, and dentistry because of their high mechanical strength and low reported toxicity. However, as usage expands, so does potential human exposure, making a clear toxicological profile essential. Titanium dioxide (TiO2) nanoparticles, known to be weakly toxic, served as a benchmark in this study. We examined the cytotoxicity of TiO2 and ZrO2 NPs in osteoblast‑like 3T3‑E1 cells, focusing on reactive oxygen species (ROS) generation, apoptosis, morphological changes, and osteogenic differentiation. Both NPs induced ROS in a concentration‑dependent manner, leading to apoptosis and altered cell morphology at high concentrations. High doses also impaired osteogenic differentiation, with ZrO2 NPs displaying more pronounced toxicity than TiO2 NPs. These findings highlight the need for careful dose management when considering ZrO2 nanoparticles in biomedical applications. Engineered nanoparticles (NPs) have permeated diverse sectors, from electronics to medicine. Zirconia (ZrO2) NPs, prized for their mechanical robustness, are now common in biosensors, cancer therapeutics, implants, and dental materials [1, 2]. Despite their advantages, the expanding use of ZrO2 NPs raises legitimate concerns about potential health and environmental risks. Existing toxicological data are sparse and sometimes contradictory: some studies report excellent biocompatibility compared to other oxides such as ferric oxide, TiO2, or ZnO [3–6], while others have detected mild cytotoxicity or cellular damage [7–12]. Previous work has also demonstrated that nanoparticles can enhance osteogenic differentiation in osteoblasts [14–17]. Nevertheless, the safety profile of ZrO2 NPs, particularly regarding bone cell function, remains unclear. This study evaluates the cytotoxic effects of ZrO2 NPs on 3T3‑E1 cells, using TiO2 NPs as a well‑characterized control. Outcomes assessed include cell viability, ROS production, apoptosis, morphology, and osteogenic markers. TiO2 (CAS 637262) and ZrO2 (CAS 544760) NPs were purchased from Sigma‑Aldrich and characterized by transmission electron microscopy (TEM), zeta potential, and dynamic light scattering (DLS). TEM provided morphology and primary particle size, while DLS assessed agglomeration in complete culture medium. Prior to cell exposure, NPs were sonicated for 30 min and diluted to the desired concentrations in α‑MEM. 3T3‑E1 cells were maintained in α‑MEM supplemented with 10% FBS and 1% antibiotic/antimycotic, at 37 °C, 5% CO2. Medium was refreshed every other day. Cell viability was quantified using the CCK‑8 assay. Cells (5 × 103 per well) were exposed to TiO2 or ZrO2 NPs (0–150 μg/mL) for 24 or 48 h, with or without 10 mM N‑acetyl‑l‑cysteine (NAC). After 2 h incubation with CCK‑8, reagents were transferred to a fresh plate to avoid NP interference, and absorbance was measured at 450 nm. After 48 h exposure, cells were harvested, stained with FITC‑Annexin V and propidium iodide, and analyzed by flow cytometry to quantify early and late apoptosis. Intracellular ROS were measured using DCFH‑DA after 48 h exposure to NPs (0–100 μg/mL). Fluorescence was detected by flow cytometry. At 24 h, cells on coverslips were fixed, permeabilized, and stained for α‑tubulin (green), F‑actin (red), and nuclei (blue). Imaging was performed with an Olympus FV10i confocal microscope. Cells were cultured for 7, 14, or 21 days with 10 or 100 μg/mL NPs, then fixed and stained with alizarin red S to visualize mineralized nodules. Total RNA was isolated, reverse‑transcribed, and quantified by SYBR‑green RT‑PCR. Osteogenic genes (RUNX2, COL1α1, ALP, OPN, OCN, BSP) were examined at days 3, 7, 14, and 21. Data are expressed as mean ± SEM. ANOVA with Bonferroni or Dunnett’s T3 post‑hoc tests evaluated significance (p < 0.05). TEM revealed TiO2 and ZrO2 NPs as rod‑shaped spheres with primary diameters of 25.4 ± 2.8 nm and 31.9 ± 1.9 nm, respectively. DLS showed hydrodynamic sizes of 81.2 nm (TiO2) and 93.1 nm (ZrO2), indicating moderate agglomeration in medium. Zeta potentials were +32.9 mV (TiO2) and +42.4 mV (ZrO2), suggesting good colloidal stability (Fig. 1). Figure 1. (a) TEM images of TiO2 and ZrO2 NPs. (b) DLS size distribution. (c) Distribution of NPs on 3T3‑E1 cells after 1 h exposure. CCK‑8 assays revealed no significant toxicity at 10 μg/mL for either NP. Viability decreased with concentrations above 20 μg/mL, reaching <50% at 150 μg/mL after 48 h (Fig. 2). NAC partially restored viability, confirming ROS involvement. Figure 2. Cell viability after exposure to TiO2 (a) and ZrO2 (b) NPs, with and without NAC. Both NPs induced ROS in a concentration‑dependent manner, peaking at 100 μg/mL after 48 h. ZrO2 NPs produced higher ROS levels at lower concentrations than TiO2. NAC effectively suppressed ROS (Fig. 3). Figure 3. ROS levels after NP exposure. Flow cytometry indicated dose‑dependent apoptosis. At 50 μg/mL, late apoptotic/necrotic cells rose to 43% (ZrO2) and 29% (TiO2). At 100 μg/mL, early apoptosis was more pronounced with TiO2 (34%) compared to ZrO2 (17%) (Fig. 4). Figure 4. Apoptosis and necrosis profiles after NP exposure. Confocal imaging revealed minimal changes at 10 μg/mL. At 100 μg/mL, cells shrank, exhibited rounded morphology, and displayed disrupted actin filaments and microtubules. Quantitative analysis confirmed reduced cell area (Fig. 5). Figure 5. Cell area changes after 24 h exposure. Figure 6. Cytoskeletal organization after NP treatment. Alizarin red staining showed no significant mineralization at 10 μg/mL across 14–21 days. At 100 μg/mL, mineralized nodules were reduced and less defined, indicating impaired osteogenic capacity (Fig. 7). Figure 7. Alizarin red staining of mineralized nodules. RT‑PCR revealed upregulation of RUNX2, COL1α1, and ALP at 10 μg/mL, peaking at day 3. High‑dose (100 μg/mL) exposure downregulated these genes by day 7. Late‑phase markers (OPN, OCN, BSP) were increased at 10 μg/mL but suppressed at 100 μg/mL (Fig. 8). Figure 8. Osteogenic gene expression over time. Our data confirm that ZrO2 NPs are biocompatible at low concentrations but become cytotoxic above 20 μg/mL, primarily through ROS‑mediated apoptosis and cytoskeletal disruption. Compared to TiO2, ZrO2 exerts stronger oxidative stress and faster apoptosis at equivalent doses. The observed ROS production likely originates from nanoparticle interaction with mitochondrial membranes, consistent with prior reports of organelle damage by ZrO2 [12]. The impairment of osteogenic differentiation at high NP doses aligns with reduced mineralization and downregulation of key osteogenic genes. These findings underscore the importance of dose optimization for any ZrO2‑based biomaterial, especially in load‑bearing implants where osteointegration is critical. Zirconia nanoparticles are generally safe for bone cells at low concentrations but exhibit dose‑dependent cytotoxicity, ROS generation, apoptosis, and suppression of osteogenesis at higher levels. These insights are essential for the rational design of ZrO2‑containing biomaterials. Silver Alkaline phosphatase Collagen 1α1 Dynamic light scattering Fetal bovine serum N-acetyl‑l‑cysteine Nanoparticles Osteocalcin Optical density Osteopontin Phosphate‑buffered saline Reactive oxygen species Runt‑related transcription factor 2 Silica Transmission electron microscope Titanium dioxide Zirconia Minimum essential medium‑alphaAbstract
Introduction
Materials and Methods
Materials Preparation and Characterization
3T3‑E1 Cell Culture
Cell Proliferation Assay
Annexin V Apoptosis Analysis
ROS Generation Analysis
Confocal Microscopy
Mineralization Induction Detection
RNA Extraction and RT‑PCR
Statistical Analysis
Results
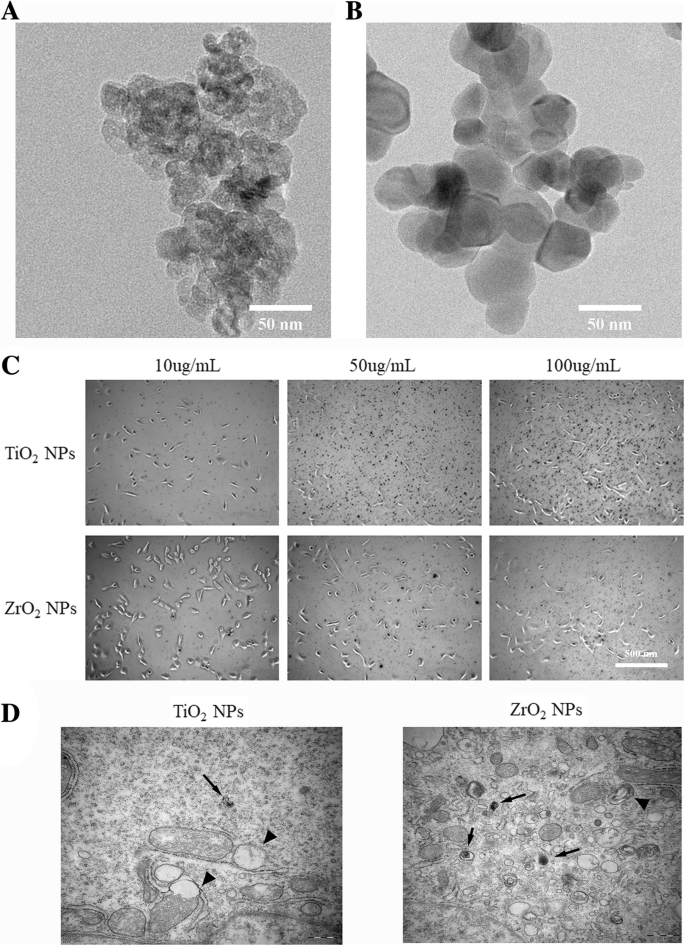
Nanoparticle Characterization
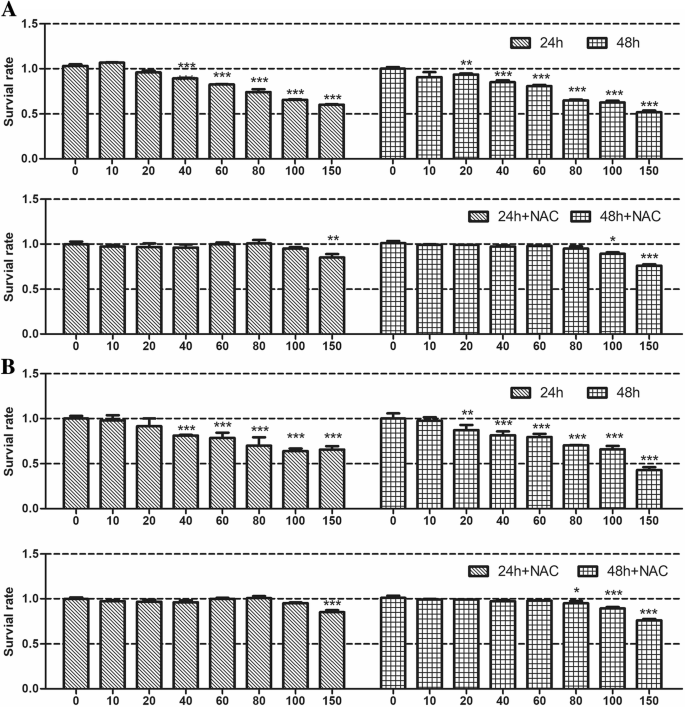
Cytotoxicity Assessment
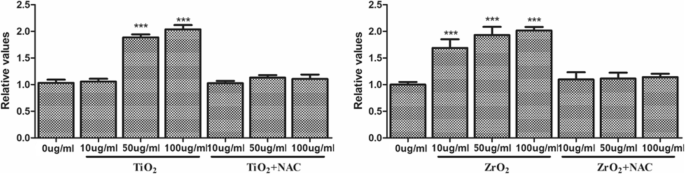
ROS Generation
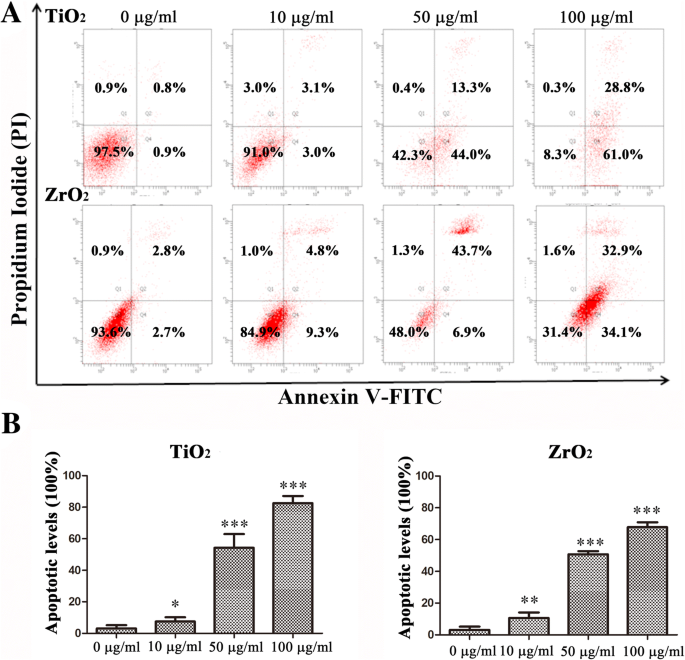
Apoptosis and Necrosis
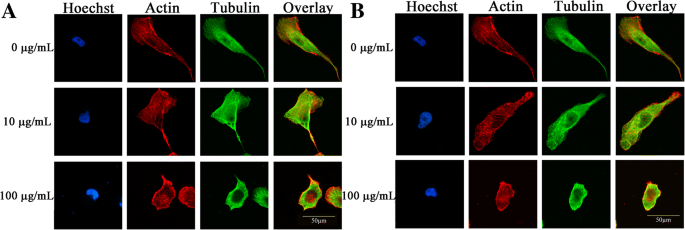
Morphological Alterations
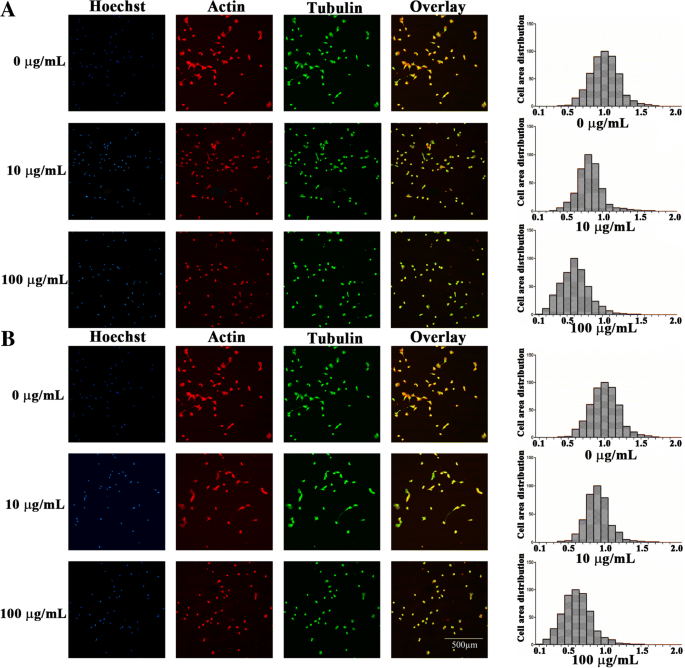
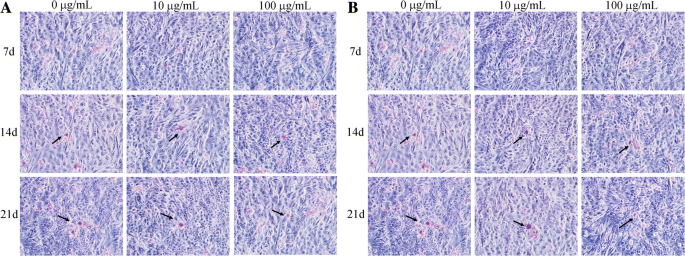
Mineralization
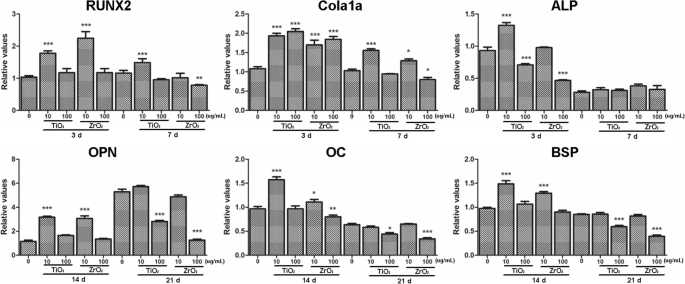
Gene Expression
Discussion
Conclusion
Abbreviations
Nanomaterials
- High‑Efficiency Graphene Solar Cells: 9% Power Conversion with TFSA Doping
- Nano‑Heterojunctions: Boosting Solar Cell Efficiency with Colloidal Quantum Dots
- Hybrid nanocomplexes of rare‑earth nanoparticles and cholesterol inhibit CD44⁺ CSCs in Ehrlich carcinoma, reducing tumor growth 75 % and extending survival
- Impact of Magneto‑Dipole Interactions on the Specific Absorption Rate of Iron Oxide Nanoparticle Assemblies
- Water-Dispersed PEG-Polymer Complexes of 4‑Thiazolidinone Chemotherapeutics Enhance Pro‑Apoptotic Activity Against Rat Glioma C6 Cells
- Enhanced Synergy of Lovastatin and Doxorubicin Delivered via Pullulan Nanoparticles Against Triple‑Negative Breast Cancer
- Optimizing Fe3O4‑Au Core‑Shell Nanoparticle Uptake for Targeted Endocytosis in Colorectal Cancer Cells
- Boosting Carbon-Based Perovskite Solar Cells: Tin-Doped Quantum Dot Interfacial Layer Enhances Photovoltaic Efficiency
- Hydroxyapatite Nanoparticles Enhance MSC Osteogenesis via Endothelial Cell Interaction
- Optimizing NIR-Triggered Hyperthermia with Cs0.33WO3 Nanoparticles for HepG2 Liver Cancer Treatment



