Curcumin‑Loaded Chitosan–Bovine Serum Albumin Nanoparticles Enhance Amyloid‑β42 Clearance and Modulate Macrophage Polarization in Alzheimer’s Disease
Abstract
Alzheimer’s disease (AD) remains the leading neurodegenerative disorder among older adults, yet effective therapies are limited by the blood–brain barrier (BBB), poor amyloid‑β (Aβ) clearance, and chronic neuroinflammation. We engineered chitosan (CS)–bovine serum albumin (BSA) nanoparticles (NPs) to deliver curcumin—a potent anti‑inflammatory and anti‑amyloidogenic polyphenol—directly to the brain. Curcumin‑loaded CS‑BSA NPs achieved a 95.4 % encapsulation efficiency, 143.5 nm mean diameter, and a negative surface charge of –10.8 mV, ensuring stability and favorable BBB permeability. In vitro BBB models showed a 60.2 % penetration of NPs after 3 h, markedly higher than free curcumin (29.8 %). Cellular uptake studies revealed enhanced intracellular curcumin levels in RAW 264.7 macrophages, which in turn increased Aβ42 phagocytosis. Mechanistically, NPs suppressed the TLR4‑MAPK/NF‑κB axis, down‑regulated pro‑inflammatory cytokines (TNF‑α, IL‑6), and shifted macrophages from a neurotoxic M1 to a protective M2 phenotype. These findings demonstrate that curcumin‑loaded CS‑BSA NPs potentiate Aβ42 clearance while modulating neuroinflammation, offering a promising nanomedicine platform for AD therapy.
Introduction
AD is characterized by progressive cognitive decline and the hallmark deposition of extracellular Aβ42 fibrils. While monomeric Aβ42 is physiologically benign, oligomeric aggregates are highly neurotoxic and drive disease progression. Targeting Aβ42 aggregation and promoting its clearance are therefore central to therapeutic strategies. Microglial cells, the brain’s resident macrophages, recognize Aβ via toll‑like receptors (TLRs) and phagocytose the peptide. However, chronic activation polarizes microglia toward a pro‑inflammatory M1 phenotype, exacerbating neuronal injury. Promoting a shift to the anti‑inflammatory M2 phenotype has emerged as a viable disease‑modifying approach.
Curcumin, derived from turmeric (Curcuma longa), exhibits anti‑amyloidogenic, anti‑oxidative, and metal‑chelating properties. It can modulate macrophage polarization by inhibiting the TLR4‑MAPK/NF‑κB signaling cascade. Unfortunately, curcumin’s poor solubility, rapid metabolism, and limited BBB permeability curtail its clinical utility.
Nanoparticle carriers can overcome these barriers. CS is a cationic, biocompatible polysaccharide capable of forming stable complexes with anionic proteins such as BSA. BSA is abundant in plasma, non‑immunogenic, and lends sustained‑release characteristics to nanoparticles. By combining CS and BSA, we fabricated curcumin‑loaded NPs optimized for BBB traversal and targeted delivery to microglia.
Materials
CS (deacetylation 80 %, ~400 kDa) was sourced from Haixin Biological Product Co., Ltd. BSA (Sigma‑Aldrich), curcumin (Dalian Meilun Biotechnology), and FITC‑labeled Aβ42 (Chinese Peptide Co.) were purchased commercially. RAW 264.7 (mouse monocyte‑macrophage) and hCMEC/D3 (human brain microvascular endothelial) cell lines were cultured in DMEM supplemented with 10 % fetal bovine serum and antibiotics. LPS (1 µg/ml) was used to polarize RAW 264.7 cells to the M1 phenotype, mimicking microglial activation in AD.
Preparation of curcumin-loaded CS‑BSA NPs
CS was dissolved in 0.1 % acetic acid (0.5 mg/ml). Curcumin (0.05 mg/ml in 100 µl DMSO) was mixed into the CS solution under magnetic stirring at room temperature. A BSA solution (1.0 mg/ml) was then added dropwise, producing an opalescent suspension that condensed into solid nanoparticles via electrostatic interaction. Particle size, zeta potential, and morphology were characterized by dynamic light scattering and TEM. In vitro release was evaluated in phosphate‑buffered saline (pH 7.4) at 37 °C, and encapsulation efficiency (EE) was calculated as EE % = (W_total – W_free)/W_total × 100 % where W_total is the initial curcumin amount and W_free the supernatant.
Cell apoptosis evaluation by MTT
Blank CS‑BSA NPs (0–2.0 mg/ml) were incubated with RAW 264.7 (M1) and hCMEC/D3 cells for 24 h to assess cytotoxicity via MTT assay.
Penetration studies using an in vitro BBB model
A monolayer of hCMEC/D3 cells (TEER > 300 Ω) was cultured on Transwell inserts. Free curcumin or curcumin‑loaded CS‑BSA NPs (equivalent curcumin 100 µg/ml) were added to the upper chamber. After 3 h incubation at 37 °C, fluorescence intensity of curcumin (excitation 425 nm, emission 530 nm) in the lower chamber was measured. Relative fluorescence ratio (RFR %) quantified penetration efficiency. Endocytic inhibitors (chlorpromazine, genistein, cytochalasin D, sodium azide) were applied to elucidate transport mechanisms.
The cellular uptake of curcumin-loaded CS‑BSA NPs
Confocal laser scanning microscopy (CLSM) assessed the intracellular distribution of curcumin in RAW 264.7 (M1) cells following exposure to free curcumin or curcumin‑loaded NPs via the Transwell system. Green fluorescence from curcumin was monitored at 6, 12, and 24 h intervals.
Detection of the phagocytosis of Aβ 42 induced by free curcumin and curcumin-loaded CS‑BSA NPs
RAW 264.7 (M1) cells pretreated with free curcumin or curcumin‑loaded NPs were incubated with FITC‑labeled Aβ42 for 3 h. Phagocytosis was quantified by CLSM fluorescence and flow cytometry.
Western blot assay
Expression levels of TNF‑α, IL‑6, TLR4, and phosphorylation states of ERK, JNK, p38, IκBα, and NF‑κB were examined by Western blot to elucidate the impact of curcumin‑loaded NPs on the TLR4‑MAPK/NF‑κB axis.
Results
Characterization of curcumin-loaded CS‑BSA NPs
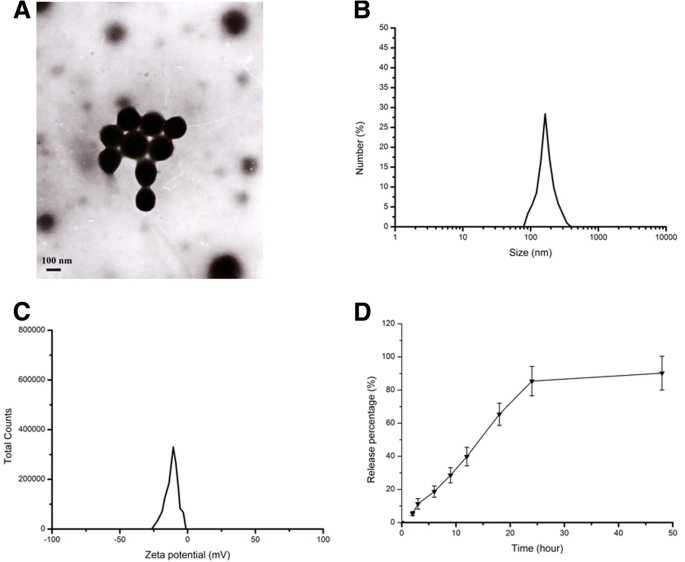
Dynamic light scattering revealed a mean diameter of 143.5 nm, zeta potential of –10.8 mV, and polydispersity index of 0.021. TEM images confirmed spherical, monodispersed particles (Figure 1). Encapsulation efficiency reached 95.4 %. Release profiling showed an initial burst of 11.3 % within 3 h, followed by sustained release over 48 h, indicating effective protection of curcumin during circulation.
Penetration through an in vitro BBB model
Free curcumin penetration was 12.3 % (1 h), 20.3 % (2 h), and 29.8 % (3 h). Curcumin-loaded NPs achieved 37.7 %, 45.6 %, and 60.2 % penetration at the same time points, respectively (Figure 2). Endocytosis inhibition experiments demonstrated that NPs relied on energy‑dependent, caveolae‑ and macropinocytosis-mediated pathways, whereas free curcumin entered cells primarily by passive diffusion.
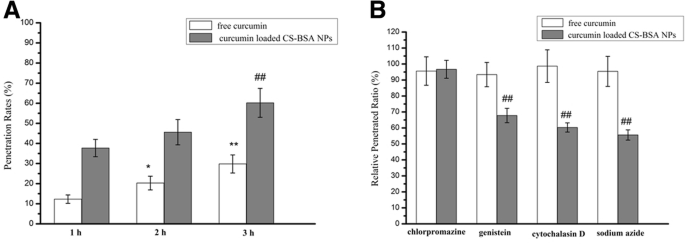
Cytotoxicity assessment
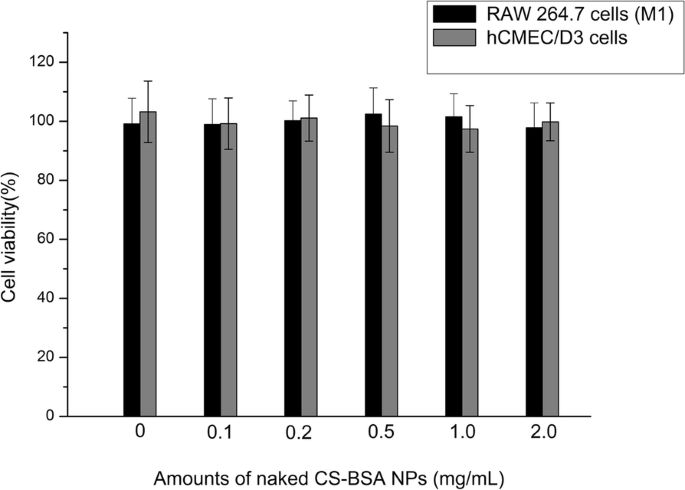
MTT assays confirmed that blank CS‑BSA NPs were non‑cytotoxic to RAW 264.7 (M1) and hCMEC/D3 cells at concentrations up to 2.0 mg/ml (Figure 3).
Cellular uptake
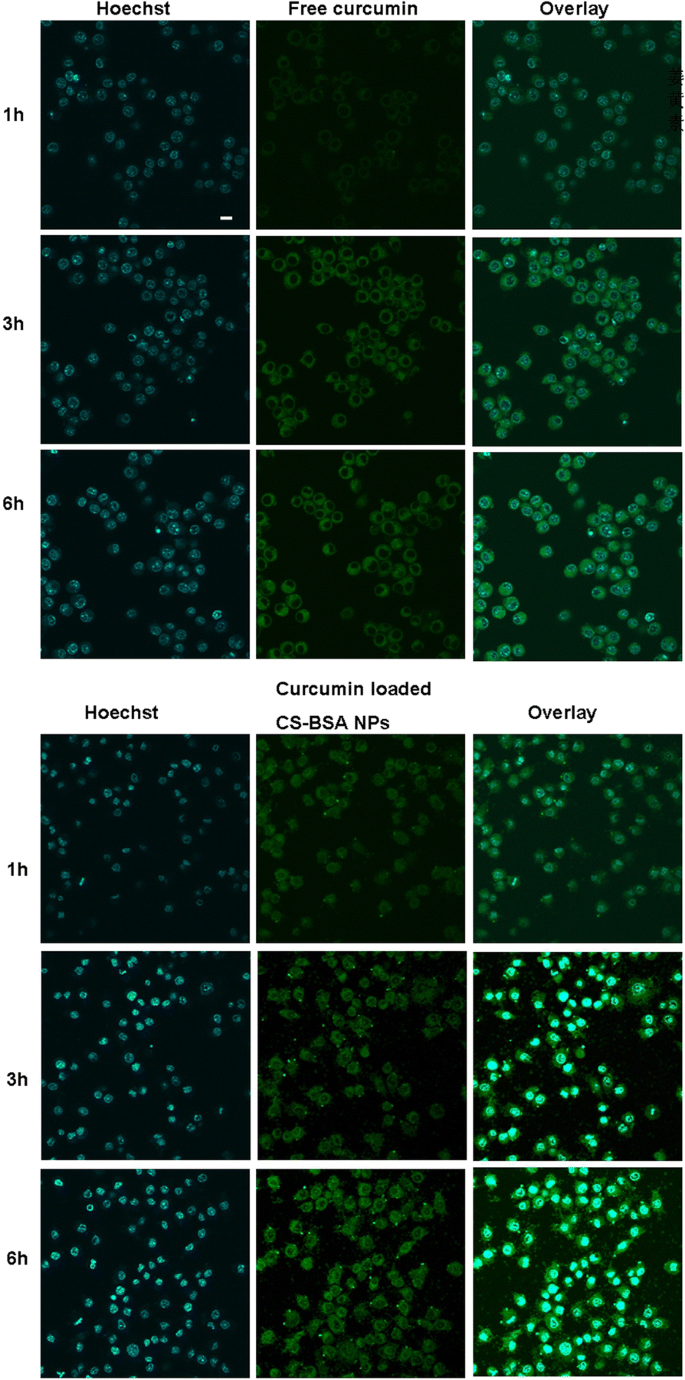
CLSM imaging showed a time‑dependent accumulation of curcumin within RAW 264.7 (M1) cells. Curcumin-loaded NPs produced markedly higher intracellular fluorescence than free curcumin, indicating enhanced delivery (Figure 4).
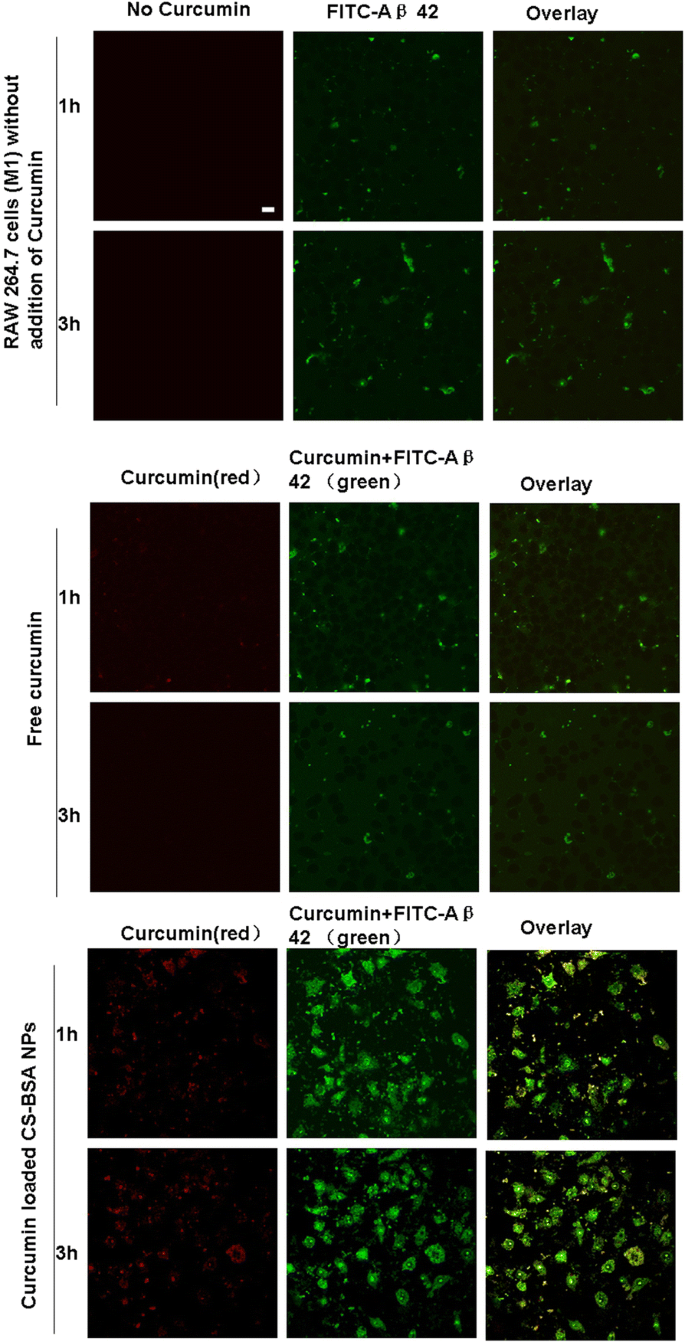
Enhanced Aβ 42 phagocytosis
Co‑localization of curcumin (red) and FITC‑Aβ42 (green) demonstrated that curcumin-loaded NPs increased the intracellular presence of Aβ42 in RAW 264.7 (M1) cells, as evidenced by a higher density of green fluorescence (Figure 5). This suggests that elevated intracellular curcumin augments microglial phagocytic capacity.
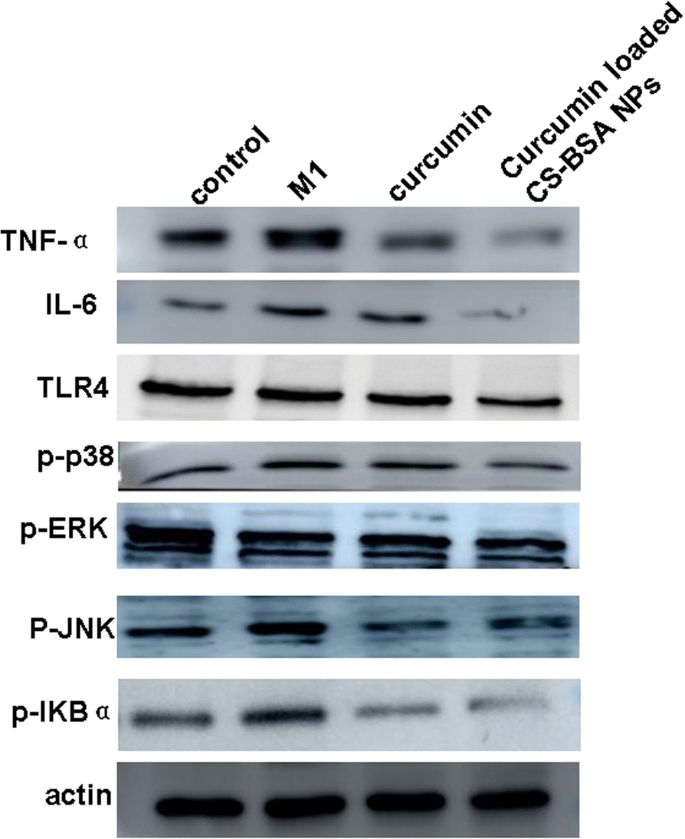
Western blot analysis
Compared to untreated controls, LPS‑polarized RAW 264.7 (M1) cells exhibited elevated TNF‑α, IL‑6, and TLR4 expression, and increased phosphorylation of ERK, JNK, p38, and NF‑κB. Curcumin-loaded NPs markedly reduced these pro‑inflammatory markers and signaling events, underscoring their capacity to inhibit M1 polarization (Figure 6).
Discussion
Our results confirm that CS‑BSA nanoparticles significantly enhance curcumin delivery across the BBB, primarily via caveolae‑ and macropinocytosis-mediated pathways. The improved brain penetration translates into higher intracellular curcumin concentrations, which, in turn, boosts microglial Aβ42 phagocytosis and reduces the pathological burden of amyloid plaques.
The anti‑inflammatory effect of curcumin-loaded NPs is evident through suppression of the TLR4‑MAPK/NF‑κB cascade and down‑regulation of TNF‑α, IL‑6, and TLR4. This cytokine modulation re‑polarizes macrophages from a neurotoxic M1 phenotype toward an anti‑inflammatory M2 state, a therapeutic strategy increasingly recognized for disease modification in AD.
Collectively, these findings position curcumin-loaded CS‑BSA NPs as a multifaceted nanomedicine platform that simultaneously addresses drug delivery, amyloid clearance, and neuroinflammation—key hurdles in AD treatment.
Conclusion
Curcumin-loaded CS‑BSA nanoparticles effectively penetrate the BBB, enhance intracellular curcumin accumulation, and promote Aβ42 phagocytosis by microglia. They also mitigate neuroinflammation by inhibiting the TLR4‑MAPK/NF‑κB pathway and favoring M2 macrophage polarization. These attributes collectively underscore the therapeutic promise of this nanoplatform for Alzheimer’s disease.
Abbreviations
- AD
Alzheimer’s disease
- BBB
Blood–brain barrier
- BSA
Bovine serum albumin
- CS
Chitosan
- LPS
Lipopolysaccharide
- NPs
Nanoparticles
- TLRs
Toll-like receptors
Nanomaterials
- Optimizing Sb-Loaded Palygorskite Nanoparticles for Superior Catalytic Hydrogenation of p-Nitrophenol
- Enhanced Photocatalytic Performance of ZnO/In₂O₃ Hybrid Nanostructures via Hydrothermal Synthesis
- gH625 Peptide vs. Folic Acid: Enhancing Fe3O4 Magnetic Nanoparticle Uptake Across the Blood–Brain Barrier
- Al₂O₃ and SiO₂ Nanoparticles with Ultrasound Significantly Reduce Water Supercooling
- Magnesium‑Doped Zinc Oxide Nanoparticles: Optimizing Photocatalytic Degradation and Antibacterial Efficacy
- High‑Efficiency Tumor‑Targeted Photo‑Chemo Theranostic Nanoparticles: Artesunate‑Loaded, ICG‑Conjugated Human Serum Albumin
- Dialysis‑Derived Tadpole and Sphere Hemin Nanoparticles: A 308‑Fold Solubility Boost for Iron Bioavailability
- Innovative Magnetic Nanoparticle Contrast Agent for Detecting Cholesterol Deposits in Alzheimer’s Disease
- Polydopamine Core–Shell Nanoparticles with Redox‑Responsive Polymer Shells for Targeted Drug Delivery and Synergistic Chemo‑Photothermal Therapy
- Ultrasmall Superparamagnetic Iron Oxide Nanoparticles for Stem Cell Tracking in Parkinson’s Therapy



