Ultra‑Sensitive Colorimetric TB Detection Using ESAT‑6 Antigen–Antibody Complexes and Gold Nanoparticles
Abstract
Tuberculosis (TB) remains a leading cause of mortality worldwide, driven by the virulence factor ESAT‑6, an early secretory antigenic target of Mycobacterium tuberculosis. Rapid, low‑concentration detection of ESAT‑6 is essential for early intervention and disease containment. We present a single‑step, antibody‑based colorimetric assay that employs 15 nm gold nanoparticles (GNPs) and a salt‑induced aggregation mechanism to achieve a detection limit of 1.25 pM. The method exploits the red spectral shift of dispersed GNPs when the ESAT‑6/antibody complex is present, while maintaining stability in the presence of non‑specific proteins such as 10‑kDa culture filtrate protein (CFP‑10). The optimal antibody concentration was determined to be 60 nM; exceeding this threshold caused a 680‑fold loss of sensitivity due to steric crowding. This assay demonstrates high specificity, minimal biofouling, and offers a rapid, visual readout suitable for point‑of‑care diagnostics of TB.
Background
TB is caused by Mycobacterium tuberculosis and poses a severe threat to global health. Early, latent infection can progress to active disease once host immunity wanes, emphasizing the need for sensitive, rapid diagnostics at the latent stage. Conventional tests—skin tuberculin, interferon‑γ release assays, chest radiography, sputum cytology, and culture—are laborious, expensive, and often delayed by months. Thus, a simple, affordable, and rapid detection strategy is urgently required.
Gold nanoparticle (GNP)–based colorimetric sensors have emerged as powerful tools for disease diagnostics due to their strong surface plasmon resonance and tunable optical properties. Monodispersed 15 nm GNPs exhibit a characteristic red hue that shifts to blue upon aggregation triggered by divalent ions, such as NaCl. This color change, driven by a red spectral shift, forms the basis of many rapid detection assays for viruses, bacteria, and small molecules. However, the performance of DNA or aptamer‑based GNP assays can be limited by electrostatic interactions and probe–target incompatibilities, especially with longer nucleic acid sequences.
To overcome these challenges, we developed a single‑step antibody‑based colorimetric assay that targets the 6 kDa ESAT‑6 protein. By pre‑complexing ESAT‑6 with its specific polyclonal antibody before exposure to GNPs, we prevent non‑specific binding and enable a highly sensitive, visual detection platform. Figure 1 illustrates the assay workflow and the specificity controls using CFP‑10.
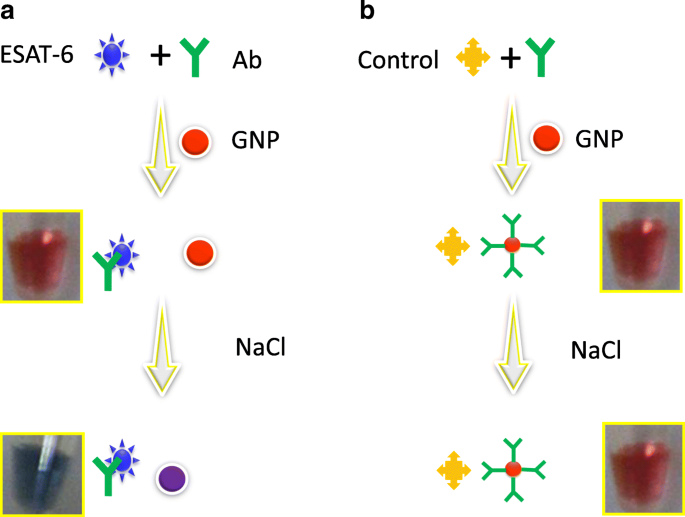
Schematic representation of the single‑step pre‑complexed antibody‑based colorimetric assay. a Antibody pre‑complexed with ESAT‑6. b Antibody pre‑complexed with CFP‑10 (negative control).
Methods
Reagents and Biomolecules
ESAT‑6 and 10‑kDa CFP‑10 were purchased from Sino Biological Inc. (Beijing, China). Anti‑ESAT‑6 polyclonal antibody was obtained from Santa Cruz Biotechnology (USA). 15 nm citrate‑capped GNPs were sourced from Sigma Aldrich (USA). All reagents were used without further purification.
Optimization of Divalent Ions for GNP Aggregation
To identify the optimal NaCl concentration for inducing GNP aggregation, a range of NaCl (50–800 mM) was added to a fixed volume of GNPs (1 O.D.). After 15 min, the color change was recorded and the absorbance spectrum (400–800 nm) was measured using a Nanophotometer.
Assessment of ESAT‑6 Biofouling on GNP Surface
To evaluate non‑specific adsorption of ESAT‑6 on GNPs, varying concentrations (1.5–100 nM) were incubated with GNPs followed by the addition of optimal NaCl. Colorimetric and spectral changes were observed. CFP‑10 was used to confirm specificity.
Optimization of Anti‑ESAT‑6 Antibody Concentration on GNP Surface
Anti‑ESAT‑6 antibody concentrations (30–500 nM) were mixed with GNPs, incubated for 30 min, and challenged with increasing NaCl to identify the antibody level that maximizes dispersion stability.
Detection of ESAT‑6 via Antibody‑Based Colorimetric Red Spectral Shift
1 µL of 1 µM ESAT‑6 (final 500 nM) was mixed with 1 µL of the optimized antibody concentration. After 30 min, 20 µL GNPs were added and incubated for another 30 min. NaCl was then added to a final concentration of 800 mM, and the absorbance spectrum (400–800 nm) was recorded. A serial dilution of ESAT‑6 (0–500 nM) was used to determine the detection limit, spanning picomolar to nanomolar ranges.
Results and Discussion
The assay leverages the red spectral shift that occurs when GNPs aggregate in the presence of the ESAT‑6/antibody complex. In the absence of ESAT‑6, the antibody coats the GNP surface, preserving the red hue even after NaCl addition, whereas the presence of ESAT‑6 triggers aggregation and a blue shift.
NaCl Optimization
GNPs remained red at NaCl concentrations below 100 mM, but began to aggregate at 100 mM, producing a red spectral shift centered at ~600 nm. For maximal sensitivity, 800 mM NaCl was selected to ensure complete aggregation in the absence of antibody coverage.
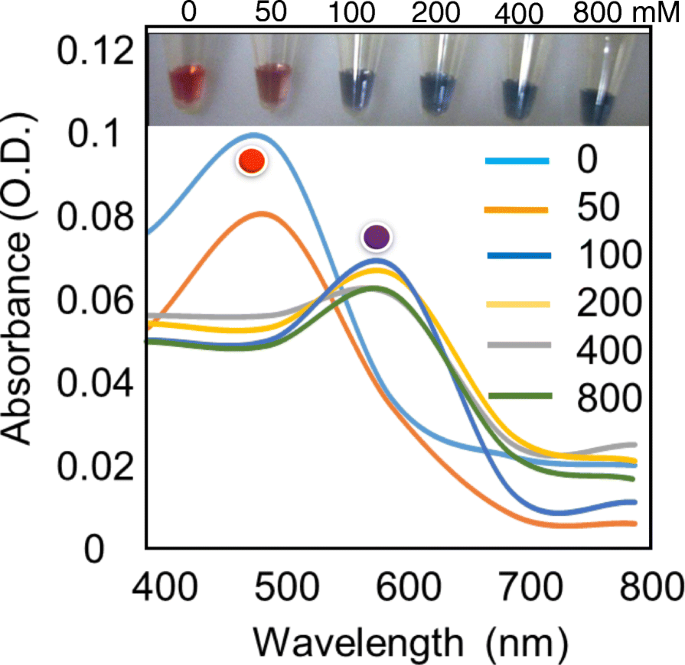
Optimization of NaCl concentration for GNP aggregation. (a) Spectral changes (400–800 nm) and (b) visual photographs illustrating the transition from red to blue at 100 mM NaCl.
Antibody Concentration Optimization
At 30 nM antibody, GNPs still aggregated even with 800 mM NaCl, indicating insufficient surface coverage. Starting at 60 nM, the GNPs maintained a red color under the same salt challenge, demonstrating effective dispersion. Concentrations above 60 nM provided no additional benefit and risked antibody crowding.
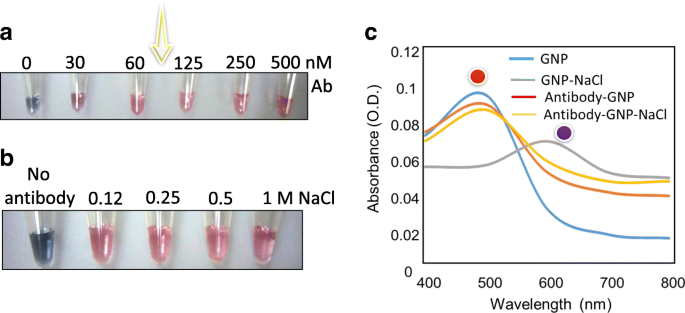
Antibody optimization and GNP stability. (a) Aggregation/dispersion trend across antibody concentrations (0–500 nM). (b) Stability of 60 nM antibody‑GNP conjugates against increasing NaCl (0.012–1000 mM). (c) Corresponding absorbance spectra.
ESAT‑6 Detection and Biofouling Validation
ESAT‑6 (up to 100 nM) did not bind to GNPs, preventing false positives/negatives. When pre‑complexed with 60 nM antibody, the assay displayed a clear color shift from red to blue at as low as 0.5 nM ESAT‑6, with full saturation observed between 15–500 nM. Spectra confirmed a red shift towards 600 nm at concentrations above 1.25 pM.

ESAT‑6 fouling assessment. GNPs remained stable up to 100 nM ESAT‑6, confirming minimal non‑specific adsorption.
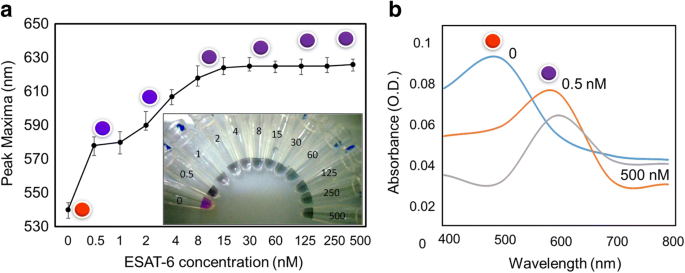
Detection of ESAT‑6 across concentrations. (a) Visual readout from 0.5 nM to 500 nM. (b) Spectral shifts (400–800 nm) indicating red shift at >1.25 pM.
Limit of Detection
By titrating ESAT‑6 from 1.25 pM to 5 nM, the assay maintained a clear blue color change and a spectral shift to ~600 nm. The limit of detection (LOD) was established at 1.25 pM, surpassing many existing colorimetric TB diagnostics.
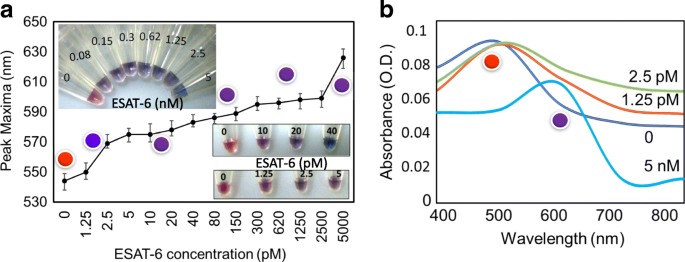
LOD assessment. (a) Visual progression from 1.25 pM to 5 nM. (b) Corresponding absorbance spectra demonstrating the red shift.
Specificity Confirmation
Using 75 nM antibody, the LOD increased to the nanomolar range, confirming the critical role of antibody concentration. CFP‑10, despite being a TB antigen, did not trigger aggregation, validating assay specificity.
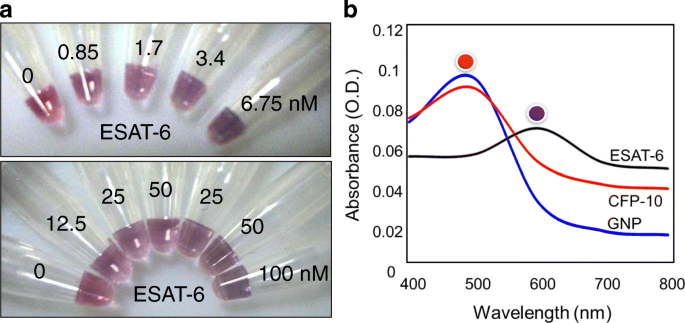
Specificity testing with 75 nM antibody. (a) Color change with varying ESAT‑6 concentrations. (b) Negative control using CFP‑10.
Conclusions
This study demonstrates a rapid, single‑step, gold‑nanoparticle–based colorimetric assay that achieves a 1.25 pM detection limit for the TB antigen ESAT‑6. The method’s high specificity, confirmed by CFP‑10 controls, and its reliance on pre‑complexed antibody/antigen reduce biofouling and enhance sensitivity. The assay’s simplicity, visual readout, and minimal equipment requirements make it a strong candidate for point‑of‑care TB screening, especially in resource‑limited settings. Future work will explore extending this platform to other small‑protein biomarkers and integrating it into portable diagnostic devices.
Abbreviations
- CFP-10:
10 kDa culture filtrate protein
- DNA:
Deoxyribonucleic acid
- ESAT-6:
6 kDa early secretory antigenic target
- GNP:
Gold nanoparticle
- NaCl:
Sodium chloride
- O.D:
One optical density (unit of GNP concentration)
- RNA:
Ribonucleic acid
- TB:
Tuberculosis
- UV:
Ultraviolet
Nanomaterials
- Revolutionizing Early Disease Detection with Nano‑Scale Biotech
- Polystyrene Core–Shell Nanocomposites with Lauryl Sulfate‑Doped Polyaniline: Synthesis, Characterization, and Sensing Potential
- GoldMag: PSS‑MA Coating Enhances Stability & Optical Precision for Protein Detection
- Highly Sensitive Nonenzymatic Glucose Sensing with Hollow Porous Nickel Oxide
- Wireless Magnetoelastic Nanobiosensor Achieves Ultra‑Sensitive Atrazine Detection
- Smartphone-Enabled Plasmonic ELISA for Ultra‑Sensitive Myoglobin Detection at Point of Care
- Silicon‑Based Photodetectors with Resonant Cavities for Near‑Infrared Polarimetric Sensing
- Enhanced Gene Delivery Using Gold Nanoparticles Functionalized with Chitosan, N‑Acylated Chitosan, and Chitosan Oligosaccharide
- Gold‑Nanoparticle‑Enhanced ELISA and Interdigitated Electrodes for Ultra‑Sensitive Detection of Human Factor IX Deficiency
- Fugro Partners with Australian Space Agency to Build Advanced Robotics Control Hub



