Hofmeister Anion‑Induced Tunable Rheology of Self‑Healing Supramolecular Hydrogels
Abstract
We explored the physical gelation of a series of d‑gluconic acetal derivatives functionalized with fatty alkyl amine groups. One derivative forms robust, self‑healing hydrogels in water. Remarkably, the elastic modulus and tensile strength of these gels can be finely adjusted by adding Hofmeister salts. Spectroscopic analyses (FT‑IR, ^1H‑NMR, XRD) reveal that π–π stacking between benzene rings drives the self‑assembly. Our study presents a straightforward strategy to modulate the mechanical properties of d‑gluconic acetal‑based hydrogels.
Background
Low‑molecular‑mass gelators (LMMGs) self‑assemble through supramolecular interactions—hydrogen bonding, van der Waals forces, π–π stacking—to form gels that are lightweight, stimuli‑responsive, and often exhibit self‑healing behavior.1–24 These materials have attracted interest for biomedical, sensing, and soft‑robotics applications.25–30 Despite advances in polymeric self‑healing gels, their lack of biodegradability limits biomedical use.31–34 Recent progress in LMMG‑based organogels and hydrogels offers a promising alternative, yet their rheological tuning remains underexplored.35–44 Hofmeister’s “specific ion” concept, first described in 1888, links ion type to protein solubility and has since been applied to colloids, polymers, and hydrogels.45–60 Studies show that kosmotropic (salt‑out) ions strengthen gel networks, whereas chaotropic (salt‑in) ions weaken them.61–68 For polymeric hydrogels, Hofmeister salts have been used to control mechanical properties, but their effect on LMMG hydrogels is largely unknown.69–71 Here, we design a series of d‑gluconic acetal derivatives (Gn, n = 1–4) bearing alkyl amine groups and investigate their gelation, self‑healing, and salt‑mediated rheology.
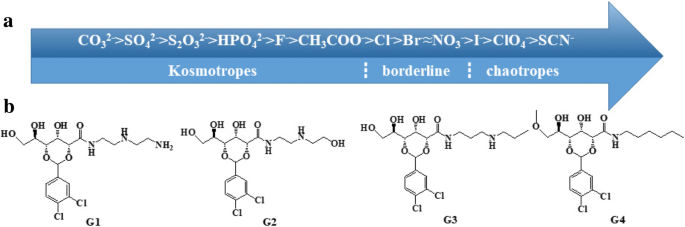
a Hofmeister anions. b Chemical structures of gelator Gn (n = 1–4).
Initial gelation tests (Additional file 1: Table S1) showed that G1–G4 are potent gelators in selected organic solvents; G1, G2, and G3 also form hydrogels, with G1 exhibiting pronounced self‑healing. G1’s critical gelation concentration (CGC) in water is 25 mM, making it the focus of subsequent salt‑effect studies. The CGC of G1 increases gradually when 0.5 M salts spanning the Hofmeister series are added, reflecting ion‑specific modulation of gelation propensity.
Methods/Experimental
Materials
All reagents were commercially sourced and used without further purification. The key precursor, 2,4-(3,4‑dichloro) benzylidene methyl‑d‑gluconate, was synthesized per literature. Detailed synthetic routes and characterisation data are in Additional file 1.
Gel Preparation
Gelation was evaluated by tube inversion. Gelators were dissolved in 1 mL solvent, heated to dissolve, cooled to room temperature, and inverted. A homogeneous, non‑flowing state was labelled G; partial gels (PG), clear solutions (S), precipitation (P), or insoluble (I) were also noted. CGC is the minimal concentration required to immobilise 1 mL solvent.
Spectroscopic and Structural Characterisation
^1H‑NMR (Bruker DPX 400 MHz) was recorded in D_2O-d6 at 25 °C. FT‑IR spectra were collected with an FTS3000 spectrometer (KBr pellets). Powder X‑ray diffraction (PXRD) used a Bruker D8‑S4 (CuKα, λ = 1.546 Å). SEM images were obtained with a Hitachi S‑4800 at 3–5 kV; samples were vacuum‑dried and Au‑coated.
Rheology Measurements
Frequency sweeps (0.1–100 rad s^−1) and strain sweeps (0.01–1000%) were performed on an Anton Paar Physica MCR 301 with a 15 mm steel‑coated parallel‑plate (gap 0.5 mm). Time‑sweep tests applied 0.1% strain to establish baseline, 100% strain to rupture, then 0.1% to monitor recovery. Measurements were conducted at 20 °C to prevent solvent loss.
Results and Discussion
Specific Ion Effects on Gelation
G1’s CGC in water rises systematically with 0.5 M salts from kosmotropes (SO_4^2−, S_2O_3^2−, HPO_4^2−, F^−) to chaotropes (I^−, ClO_4^−, SCN^−). The correlation between CGC and the viscosity B coefficient of each anion (Fig. 2a) yields a linear fit (slope = 0.543). Removing outliers (ClO_4^−, F^−, S_2O_3^2−) improves the fit to 0.932 (Fig. 2b). Kosmotropes, which strengthen water hydrogen bonds, reduce free water and promote gelator self‑assembly, lowering CGC. Chaotropes have the opposite effect.
Gel transition temperature (T_gel) follows the same trend (Fig. 2c,d). For most anions at 0.5 M, T_gel decreases from kosmotropes to chaotropes, reflecting a stronger fibrillar network in the presence of kosmotropic ions. Increasing salt concentration from 0 to 1 M generally raises T_gel, suggesting a denser 3‑D network.
Salt‑Induced Tunable Rheology
G1 gels display rapid self‑healing: a 2.0% (w/v) gel can recover from mechanical disruption within 10 min and re‑join when cut (Fig. 3a). Rheological analysis (2.5% w/v, 0.5 M salts) shows that G′ spans 110 kPa to 350 kPa, and G″ ranges 11 kPa to 65 kPa, with a 68–83% variation across the Hofmeister series (Fig. 3b). Kosmotropic anions elevate G′ and G″, whereas chaotropes diminish them. Time‑sweep tests reveal recovery ratios above 80% after each rupture cycle, confirming excellent thixotropy and self‑healing.
Spectroscopic Insights
FT‑IR spectra (Fig. 4a) reveal shifts in O–H, N–H, and C=O bands upon addition of SO_4^2− and S_2O_3^2−, indicating weakened hydrogen bonding but improved overall gel stability. ^1H‑NMR (Fig. 4b) shows upfield shifts of benzene protons with kosmotropic ions, reflecting enhanced π–π stacking, and downfield shifts with chaotropes, indicating weakened stacking. These trends align with the observed mechanical behavior.
Microstructure
SEM images (Fig. S3) illustrate rope‑like left‑handed helical fibers (~30–50 nm) forming dense 3‑D networks in the presence of SO_4^2−, whereas SCN^− yields loosely bundled, broken fibers, correlating with weaker mechanical properties.
PXRD and Packing Mode
WXRD patterns (Fig. 4c) display characteristic d‑spacings of 0.38 nm (π–π stacking) and 0.46 nm (hexagonal close packing). The addition of SO_4^2− or SCN^− does not alter the fundamental packing motif, suggesting that ion effects primarily influence network density rather than molecular packing.
Proposed Self‑Assembly Mechanism
Energy‑minimised G1 (Fig. 4d) suggests a 1.46 nm length and a face‑to‑face π–π stacking orientation. Nanofibers assemble into helical bundles via van der Waals interactions, culminating in a 3‑D intertwined network that entrains water.
Conclusions
We have developed a d‑gluconic acetal‑based gelator that forms self‑healing hydrogels with tunable rheology through Hofmeister anion addition. FT‑IR, ^1H‑NMR, and PXRD confirm π–π stacking as the primary self‑assembly driver. This work provides a facile route to tailor the mechanical and self‑healing properties of low‑molecular‑weight supramolecular hydrogels, with implications for biomedical and soft‑material applications.
Nanomaterials
- The 555 Integrated Circuit: A Timeless Benchmark in Analog Design
- Ultra‑Sensitive, Label‑Free Immunoassay Using Gold Nanoparticle–Graphene Oxide Hybrid Plasmonic Biosensors
- Ultra‑Broadband, Tunable Terahertz Absorber Based on Multi‑Layer Graphene Ribbons
- Poly(acrylic acid)/Boron Nitride Composite Hydrogels with Superior Mechanics and Rapid Self‑Healing via Hierarchical Physical Interactions
- Double‑Gated Nanohelix as a Tunable Binary Superlattice: Band Engineering and Optoelectronic Prospects
- Precise All‑Optical Control of Light Transmission in WSe₂‑Coated Microfibers
- Large-Area Extrinsic Chiral Metal Nanocrescent Arrays Deliver Giant, Tunable Circular Dichroism
- Design and Simulation of Tunable Metasurface Color Filters with Aluminum and Lithium Niobate
- Advanced Approaches to Noble Metal Nanoparticle Synthesis
- Engineered Nickel‑Cobalt Layered Double Hydroxides: Tunable Thin‑Layer Nanosheets for Superior Supercapacitor Performance



