Ultra‑Sensitive, Label‑Free Immunoassay Using Gold Nanoparticle–Graphene Oxide Hybrid Plasmonic Biosensors
Abstract
This work presents a novel AuNP‑GO nanocomposite that combines the high biocompatibility of graphene oxide with the localized surface plasmon resonance (LSPR) of gold nanoparticles. The hybrid platform enables label‑free detection of protein interactions with unprecedented sensitivity. Using the AuNP‑GO sensor, anti‑BSA was detected at concentrations as low as 145 fM in the presence of 10 ng ml⁻¹ hCG interference. The dynamic range spanned five orders of magnitude (1.45 nM to 145 fM). These results demonstrate the potential of AuNP‑GO plasmonic biosensors for rapid, point‑of‑care diagnostics and biomarker screening.
Background
Carbon‑based nanomaterials—graphene, carbon nanotubes, and graphene oxide (GO)—have become staples in biosensing due to their exceptional electrical, optical, and surface chemistry. GO’s two‑dimensional structure and oxygen‑rich functional groups (epoxides, hydroxyls, carboxyls) confer high protein affinity and enable covalent coupling, making it an attractive substrate for electrochemical, fluorescence, SPR, and LSPR sensors. Gold nanoparticles (AuNPs) bring tunable plasmonic absorption, biocompatibility, and facile surface chemistry, allowing them to act as signal amplifiers or optical tags. When AuNPs are decorated on GO, the hybrid inherits the rapid binding of GO and the intense LSPR of AuNPs, leading to enhanced sensitivity in immunoassays, DNA detection, and enzymatic assays. Despite extensive electrochemical and SERS studies, no rapid, colorimetric, or label‑free immunoassays based on naked‑eye AuNP‑GO hybrids have been reported to date. This study fills that gap by developing a chemisorption‑based, LSPR‑enhanced immunoassay that operates in aqueous buffer and tolerates common interferents.
Methods/Experimental
Materials
Graphite (Graphene Supermarket), GO (modified Hummer’s method, 0.1–1 µm flakes, 1.1 nm thick), cystamine dihydrochloride (Cys, 96 %), HAuCl₄·3H₂O (99.99 %), sodium citrate, BSA, anti‑BSA (rabbit IgG), NHS, and EDC were all obtained from Sigma‑Aldrich or Alfa Aesar. All reagents were used without further purification.
Synthesis of AuNPs
AuNPs were synthesized by citrate reduction: 15 mL of 1 mM HAuCl₄ was refluxed at 550 °C, 1100 rpm; 1.8 mL of 38.8 mM sodium citrate was added. After 5 min, the mixture was boiled at 400 °C, 900 rpm for 30 min and cooled to room temperature. This procedure yields ~15 nm spherical AuNPs; using 0.8 mL citrate produces ~60 nm particles. The reaction: HAuCl₄ + C₆H₅O₇Na₃ → Au(s) + CO₂ + HCOOH.
Preparation of GO‑BSA Antigen Target
GO sheets (0.1 g L⁻¹) were activated with EDC (400 µM) and NHS (100 µM) (1:1 volume ratio). BSA (20 µL, 100 µg mL⁻¹) was covalently coupled via amide bond formation and purified by repeated centrifugation. Figure 1 illustrates the GO‑BSA conjugation workflow.
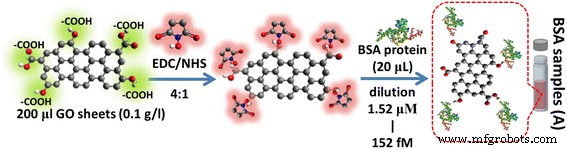
GO‑BSA interaction. The carboxyl group of GO sheets can be activated using EDC/NHS reaction and preparation of the GO based on antigen target for GO‑BSA
Preparation of AuNP‑antiBSA Probe
AuNPs were functionalized with Cys (10 mM, 2 h, RT) to introduce exposed –NH₂ groups. Anti‑BSA (20 µL, 100 µg mL⁻¹) was then coupled via amide bonds without additional activation, followed by centrifugation to remove unbound antibody. Serial dilutions from 100 µg mL⁻¹ to 1 pg mL⁻¹ produced a range of AuNP‑antiBSA probes (Fig. 2a). AuNP‑GO‑antiBSA composites were assembled by EDC/NHS activation of GO carboxyls and covalent attachment of Cys‑modified AuNPs, then antibody coupling (Fig. 2b).
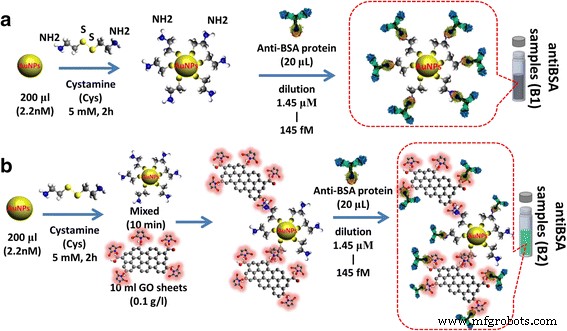
AuNP‑antiBSA and AuNP‑GO‑antiBSA interactions. Preparation of the a AuNPs and b AuNP‑GO based on antibody probe
Characterization Techniques
Structural and optical characterization employed FE-TEM, HR‑TEM, SEM (JEOL JSM‑7800F), UV‑vis spectroscopy (U‑2900, Hitachi), Raman microscopy (MRI, Protrustech), FTIR (Bruker Vertex 80v ATR), and XPS (National Synchrotron Radiation Research Center). AuNP size, distribution, and surface chemistry were confirmed across all samples.
Results and Discussion
Morphology and Composition
SEM and TEM revealed uniform 60‑nm AuNPs evenly distributed on wrinkled GO sheets (Fig. 3). XPS confirmed the presence of C–C (sp²), C–C (sp³), C–O, C=O, and O–C=O functional groups on GO, while Au 4f peaks verified AuNP integration.

Surface morphology analysis of the nanocomposite. a SEM images of a GO sheet and b TEM image of AuNP‑GO composite. c The TEM image of the ERGO AuNP‑GO film
Spectroscopic Characterization of GO
High‑resolution C 1s XPS, Raman, and ATR‑FTIR spectra confirm the characteristic oxygenated functional groups of GO (sp² C, epoxides, hydroxyls, carbonyls, carboxyls). Raman peaks at 1355 cm⁻¹ (D), 1614 cm⁻¹ (G), and 2714 cm⁻¹ (2D) corroborate the layered structure (Fig. 4).
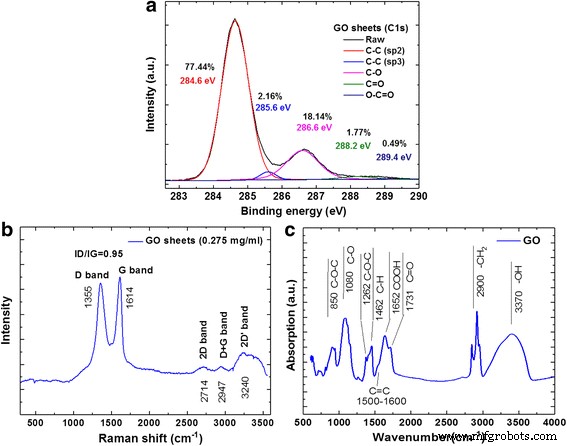
Spectral analysis of GO sheets. a XPS high‑resolution scan in the C1s region, b Raman, and c FTIR
Protein Interaction and LSPR Response
UV‑vis spectra of AuNPs (15 nm and 60 nm) show LSPR peaks at 520 nm and 540 nm, respectively. GO alone displays π–π* absorption at 230 nm and n–π* at 300 nm. After functionalization, AuNP‑Cys‑antiBSA conjugates exhibit dual peaks at 540 nm (AuNP) and 755 nm (AuNP‑Cys‑antiBSA) with a concentration‑dependent shift (Fig. 5). Calibration curves (y = 2.43 + 0.25 x, R² = 0.91 for 540 nm; y = 2.56 + 0.31 x, R² = 0.96 for 760 nm) confirm linearity across 1.45 nM to 145 fM anti‑BSA.
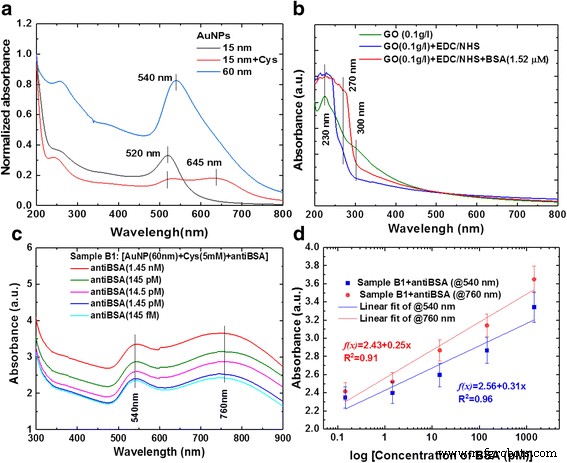
Analysis of UV‑vis absorption spectra for AuNP with antiBSA interaction response. a SP absorption spectra of AuNPs, b GO‑bound BSA, c AuNP‑anti‑BSA probe, and d Calibration curves for AuNP with antiBSA interaction response at dilution different concentrations of antiBSA protein from 1.45 nM ~ 145 fM
AuNP‑GO‑antiBSA hybrids further amplify the LSPR signal. Upon addition of GO‑BSA targets, the 540‑nm peak shifts linearly with anti‑BSA concentration (1.45 nM–145 fM), yielding a LOD of 145 fM (Fig. 6). Calibration for the AuNP‑GO probe (f(x) = 0.918 + 0.124 x, R² = 0.94) and for GO alone (f(x) = 0.791 + 0.057 x, R² = 0.954) demonstrate superior sensitivity of the hybrid system.
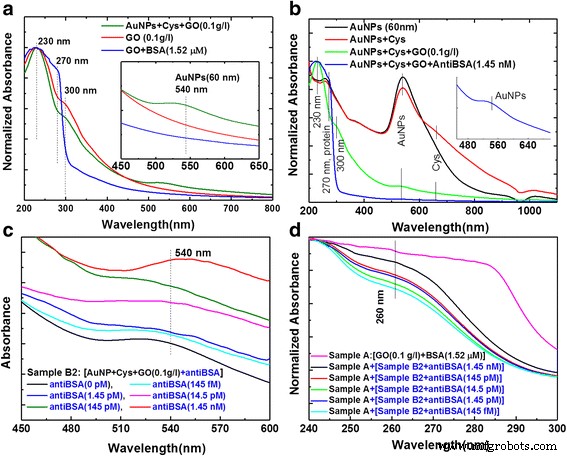
Analysis of UV‑vis absorption spectra for immune response. a AuNP‑bound GO and GO‑bound BSA, b AuNPs and GO‑bound anti‑BSA, c AuNP‑GO‑antiBSA probe, and d AuNP‑GO‑antiBSA probe and GO‑BSA target for immune response
Interference studies with 10 ng ml⁻¹ hCG showed minimal cross‑reactivity (R² = 0.89 for AuNP‑GO; 0.73 for GO alone), confirming assay robustness. The sensor retained performance after four regeneration cycles, indicating high surface stability.
Conclusions
We have engineered a biocompatible AuNP‑GO hybrid that leverages LSPR to achieve label‑free detection of anti‑BSA at 145 fM. The nanocomposite exhibits high bioaffinity, rapid response, and resilience to common interferents, making it a compelling platform for point‑of‑care immunoassays. Future work will explore multiplexed detection, integration into microfluidic devices, and adaptation to clinically relevant biomarkers.
Abbreviations
- Ag:
Silver
- AntiBSA:
Bovine serum albumin antibody
- Au:
Gold
- AuNP:
Gold nanoparticle
- BSA:
Bovine serum albumin
- Cys:
Cystamine
- EDC:
1‑Ethyl‑3‑(3‑dimethylaminopropyl)carbodiimide
- FEG‑TEM:
Field‑emission gun transmission electron microscope
- FTIR:
Fourier‑transform infrared spectrometer
- GO:
Graphene oxide sheet
- HR‑TEM:
High‑resolution transmission electron microscope
- LOD:
Limit of detection
- LSPR:
Localized surface plasmon resonance
- MOA:
8‑Mercaptooctanoic acid
- NHS:
N‑Hydroxysuccinimide
- Pd:
Palladium
- POCT:
Point‑of‑care testing
- Pt:
Platinum
- RGO:
Reduced graphene oxide
- SAMs:
Self‑assembled monolayers
- SERS:
Surface‑enhanced Raman scattering
- UV‑vis:
Ultraviolet‑visible
- XPS:
X‑ray photoelectron spectroscopy
- ZnO:
Zinc oxide
Nanomaterials
- Plasmonic Nanoparticles: Harnessing Surface Plasmons for Advanced Photothermal Applications
- Gold Nanoparticles as Advanced Chemosensors: Enhancing Electrochemical Detection
- Gold Nanoparticles: Advancing Diagnostic and Therapeutic Applications in Medicine – A Comprehensive Review
- Boosting Nonlinear Optical Response in Hybrid Liquid Crystal Cells with Photonic Crystal Integration
- Hybrid TiO₂ Nanocomposite Coating Achieves 80% Diffuse Reflectance and Suppressed Specular Glare
- Smartphone-Enabled Plasmonic ELISA for Ultra‑Sensitive Myoglobin Detection at Point of Care
- Hierarchical Au@CdS‑CdS Nanoflowers: Stepwise Fabrication and Superior Photocatalytic Performance
- Enhanced Visible-Light Photocatalysis by Anchoring Plasmonic Ag@AgCl Nanocrystals on ZnCo₂O₄ Microspheres
- Gold Nanoparticle‑Based Immunofluorescence Enables Rapid, Sensitive Detection of Inflammation‑Associated Cancer Biomarkers
- Gold Nanoparticles Boost Electron Transport in PF‑NR2 Cathode Interface for High‑Performance iPLEDs



