Controlling Contact Geometry in Carboxylic‑Acid Molecular Junctions via Side‑Group Position
Abstract
We demonstrate that the contact geometry of single‑molecule junctions can be tuned by the position of a methoxy side‑group on carboxylic‑acid ligands. Using electrochemical jump‑to‑contact scanning‑tunneling‑microscopy break‑junction (ECSTM‑BJ) on Cu electrodes, 2‑methoxy‑1,3‑benzenedicarboxylic acid (2‑M‑1,3‑BDC) exhibits a conductance of ~10–3.65 G0, whereas 5‑methoxy‑1,3‑benzenedicarboxylic acid (5‑M‑1,3‑BDC) shows ~10–3.20 G0, identical to 1,3‑benzenedicarboxylic acid (1,3‑BDC). 1,3‑benzenedicarboxaldehyde (1,3‑BDCA) contacts Cu through a single oxygen atom and yields the same conductance as 2‑M‑1,3‑BDC, confirming that the 2‑position methoxy group forces the carboxylate to bind via one oxygen. These findings establish a straightforward strategy to dictate contact configurations, a critical parameter for future molecular electronic devices.
Background
Understanding electron transport in single‑molecule junctions is essential for molecular electronics [1–14]. Multiple factors—molecular structure, anchoring groups, electrode material, and contact geometry—shape conductance [15–28]. Yet, controlling contact geometry remains challenging, limiting reproducibility and device design [29–31]. Experimental reports of distinct conductance peaks for a single molecule indicate multiple contact configurations [20,30]. While mechanical manipulation can switch between these states [31], it introduces additional variability. Recent work suggests that adding side‑groups can lock a junction into a single configuration [28], opening a path to controlled contact geometries. Here we explore this concept with carboxylic‑acid ligands on Cu electrodes.
Carboxylic acids reliably form junctions with Cu and Au electrodes [19,24,30,32], and Cu is advantageous electrochemically for preventing oxidation [33]. We selected four benzene‑based carboxylic acids: 2‑M‑1,3‑BDC, 1,3‑BDC, 5‑M‑1,3‑BDC, and 1,3‑BDCA (Fig. 1). ECSTM‑BJ was employed to construct and measure these junctions.
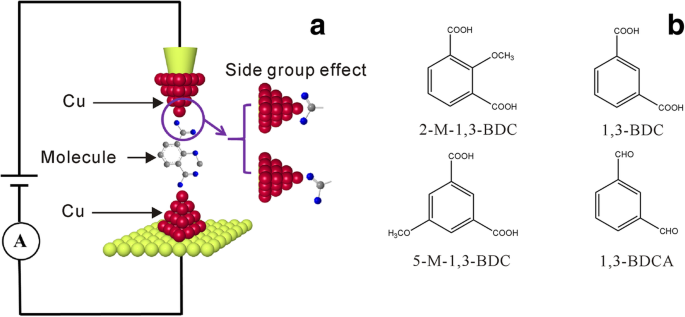
Schematic of the ECSTM‑BJ setup and molecular structures: a ECSTM‑BJ illustration (red Cu, green Au, blue O, gray C). b Target molecules: 2‑M‑1,3‑BDC, 1,3‑BDC, 5‑M‑1,3‑BDC, 1,3‑BDCA.
Methods
Reagents were purchased from Alfa‑Aesar, Sigma‑Aldrich, and TCI, used without further purification. Au(111) single‑crystal beads served as substrates; Pt‑Ir tips were insulated with thermosetting epoxy. Cu and Pt wires functioned as counter and reference electrodes, respectively.
Conductance measurements employed a modified Nanoscope IIIa STM (Veeco) in 1 mM CuSO4 + 50 mM Na2SO4 + 1 mM target molecule. The tip and substrate biases were –5 mV and +45 mV vs. Cu wire, promoting Cu deposition on the tip only. After approach, a jump‑to‑contact occurred; the tip was retracted at 20 nm s–1, recording conductance until junction rupture. Thousands of traces formed a histogram without data curation, as detailed in previous studies [23,34,35].
Computational analysis used DFT‑based NEGF. Structures were relaxed with 3–4 buffer layers and 15 Å vacuum. PBE exchange–correlation and DZP (molecule and surface Cu) or SZP (bulk Cu) basis sets were applied, with a (4,4) k‑point grid. Calculations were carried out in SHINE (Shanghai Integrated Numeric Engineering) [36,37].
Results and Discussion
Conductance of 2‑M‑1,3‑BDC (methoxy at the 2‑position)
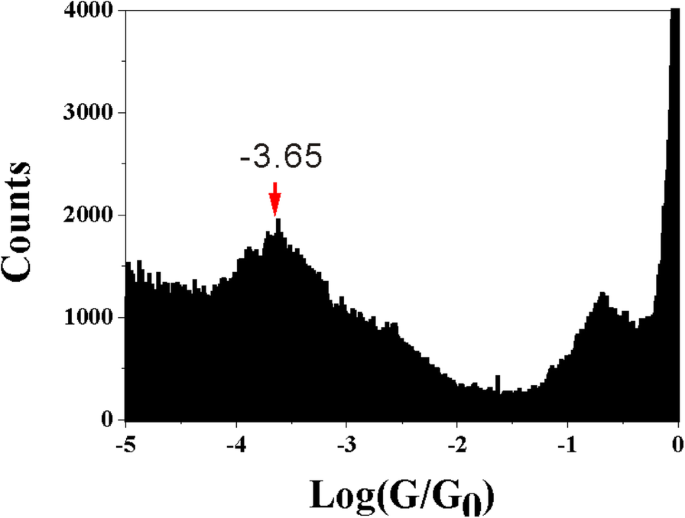
In 1 mM 2‑M‑1,3‑BDC + 1 mM CuSO4 + 50 mM Na2SO4, ECSTM‑BJ produced Cu clusters (Fig. 2a). Typical conductance traces (log scale) reveal a plateau at ~10–3.65 G0 (Fig. 2b). A histogram built from ~1,500 traces shows a dominant peak at the same value (Fig. 2c), indicating a single, well‑defined contact configuration.
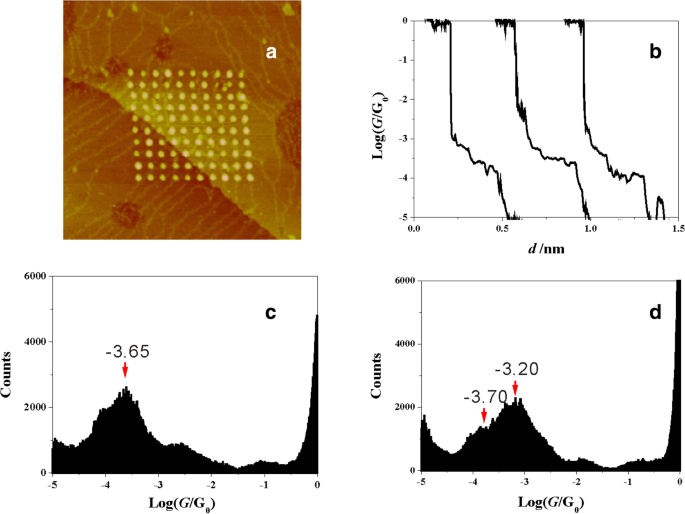
STM image and conductance of 2‑M‑1,3‑BDC (a) Cu cluster array; (b) representative traces; (c) histogram for 2‑M‑1,3‑BDC; (d) histogram for 1,3‑BDC.
Contrastingly, 1,3‑BDC exhibits a peak at ~10–3.20 G0 (Fig. 2d). The methoxy group does not contribute directly to electrode binding, confirming that the carboxylate anchors the junction. The ~300 % conductance disparity indicates that side‑group position, not electronic donation alone, dictates contact geometry, since literature reports <20 % changes for single side‑group substitutions [38].
Conductance of 5‑M‑1,3‑BDC (methoxy at the 5‑position)
Adding the methoxy group at the 5‑position, far from the anchoring groups, yields a histogram with a peak at ~10–3.20 G0 (Fig. 3), matching 1,3‑BDC. This demonstrates that side‑group proximity, rather than mere presence, governs contact configuration.
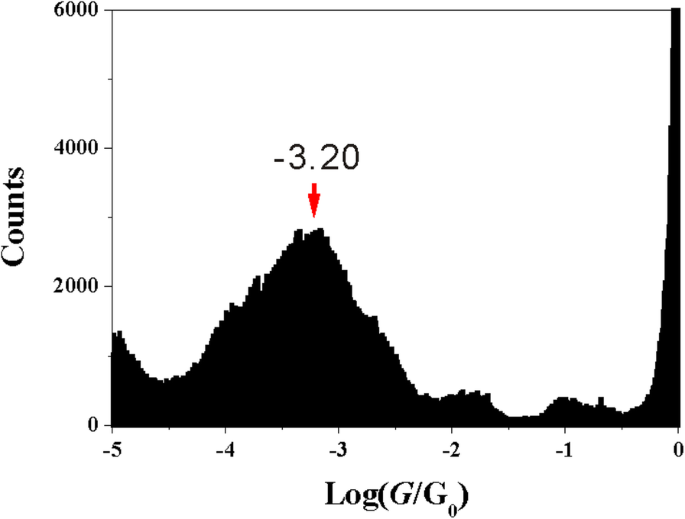
Histogram of 5‑M‑1,3‑BDC (1500 traces).
Origin of the Conductance Difference
Destructive quantum interference (DQI) can lower meta‑benzene conductance by orders of magnitude [39,40]. However, 1,3‑BDC (10–3.20 G0) exceeds the para‑benzene analog 1,4‑BDC (10–3.40 G0) [35], indicating no DQI. Calculated transmission spectra for junctions where the carboxylate binds via two oxygens show no conductance drop for either 2‑M‑1,3‑BDC or 5‑M‑1,3‑BDC (Fig. 4), ruling out DQI effects.
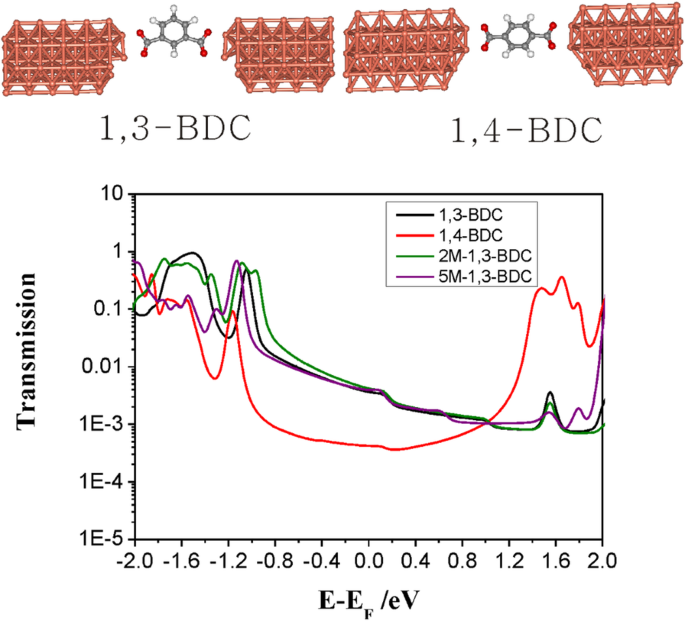
Transmission spectra for 1,3‑BDC, 1,4‑BDC, 2‑M‑1,3‑BDC, and 5‑M‑1,3‑BDC contacting Cu via two‑oxygen carboxylate.
The key lies in contact geometry: the carboxylate can bind Cu through one or two oxygens. For 1,3‑BDC and 5‑M‑1,3‑BDC, the dominant peak at 10–3.20 G0 corresponds to two‑oxygen binding. In 2‑M‑1,3‑BDC, the adjacent methoxy sterically hinders this mode, forcing a single‑oxygen contact and yielding the lower conductance of 10–3.65 G0.
Validation with 1,3‑BDCA
1,3‑BDCA, which possesses only a carbonyl anchor capable of single‑oxygen binding, shows a histogram peak at ~10–3.65 G0 (Fig. 5). This matches 2‑M‑1,3‑BDC, confirming that the 2‑position methoxy enforces the one‑oxygen contact. A minor shoulder at 10–3.70 G0 in the 1,3‑BDC histogram likely arises from one‑oxygen binding, while the main peak stems from two‑oxygen binding.
Histogram of 1,3‑BDCA (1100 traces).
Table 1 summarizes all conductance values, reinforcing that contact geometry is dictated by side‑group position. Steric hindrance from a neighboring methoxy prevents two‑oxygen binding, offering a practical handle to design junctions with desired conductance.
Conclusions
We have shown that positioning a methoxy side‑group on a carboxylic‑acid ligand controls whether the Cu electrode engages via one or two oxygen atoms. The 2‑position methoxy forces a single‑oxygen contact, reducing conductance to ~10–3.65 G0; the 5‑position methoxy preserves the two‑oxygen mode, yielding ~10–3.20 G0. This strategy offers a straightforward route to engineer contact geometries in single‑molecule electronics, potentially enhancing device reproducibility and performance.
Availability of Data and Materials
The datasets generated during this study are available from the corresponding author upon reasonable request.
Abbreviations
- 1,3‑BDC
1,3‑benzenedicarboxylic acid
- 1,3‑BDCA
1,3‑benzenedicarboxaldehyde
- 1,4‑BDC
1,4‑benzenedicarboxylic acid
- 2‑M‑1,3‑BDC
2‑methoxy‑1,3‑benzenedicarboxylic acid
- 5‑M‑1,3‑BDC
5‑methoxy‑1,3‑benzenedicarboxylic acid
- DQI
Destructive quantum interference
- ECSTM‑BJ
Electrochemical jump‑to‑contact STM break junction
Nanomaterials
- IBM Scientists Measure Thermal Conductance of Single-Atom Gold Contacts at Room Temperature
- Molecular Dynamics Investigation of Ultra‑Precision Diamond Cutting of Cerium
- Low Tunneling Decay Observed in Iodine-Terminated Alkane Single-Molecule Junctions
- Shape Stability of Metallic Nanoplates: Insights from Molecular Dynamics Simulations
- Optimizing Ohmic Contacts on p‑GaAs Nanowires for Low‑Resistance, High‑Reliability Devices
- Optimizing DNA Translocation in Solid‑State Nanopores for Enhanced Sequencing Accuracy
- Influence of Hydrophilicity on Single-File Water Flux Through Sub‑Nanometer Pores in 2D Nanoporous Membranes
- Exploring Tip-Based Nanomachining with Molecular Dynamics: A Comprehensive Review
- Ensuring Product Integrity: Molecular Tagging for End-to-End Supply Chain Traceability
- Linfox Leverages Digital Transformation to Boost Efficiency & Customer Trust



