Understanding Band Theory of Solids: How Electron Energy Levels Shape Electrical Conductivity
Quantum mechanics describes electrons in atoms using four quantum numbers, which specify the allowed energy states—much like seats in a stadium. Each electron occupies a unique set of quantum numbers, analogous to a spectator assigned to a specific row and seat.
Electrons can move between these states when energy is supplied or released, just as a spectator might switch rows. Transitions between different shells demand significant energy, while moving within a shell or between subshells requires less.
When atoms bond to form a solid, their outer shells, subshells, and orbitals merge, creating a dense array of energy levels that coalesce into nearly continuous bands. This is illustrated in the figure below.
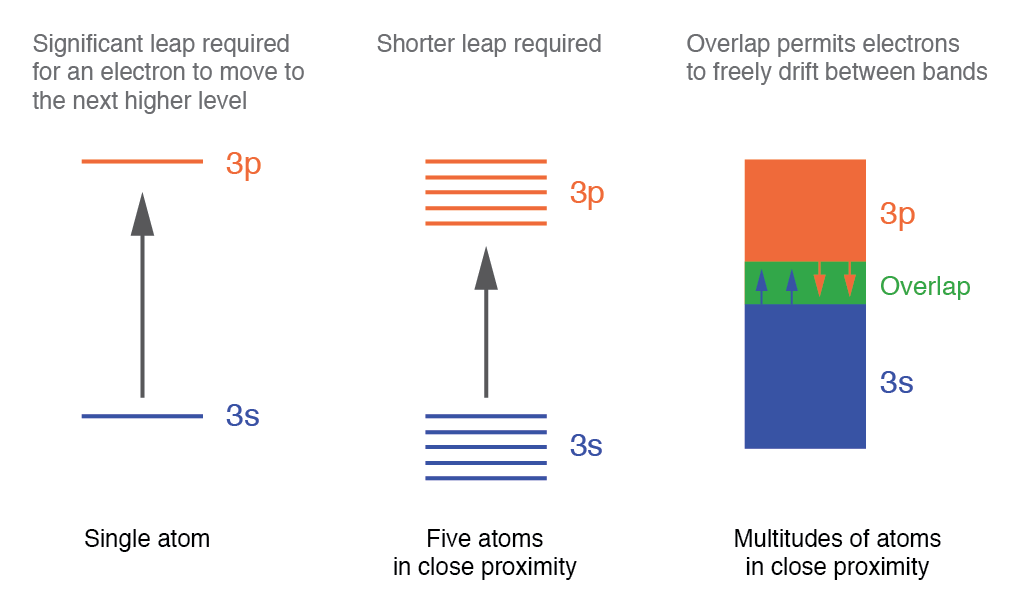
In metals, the conduction band overlaps with the valence band, allowing electrons to flow freely with minimal external energy. These “free” electrons are the carriers that give metals their excellent conductivity.
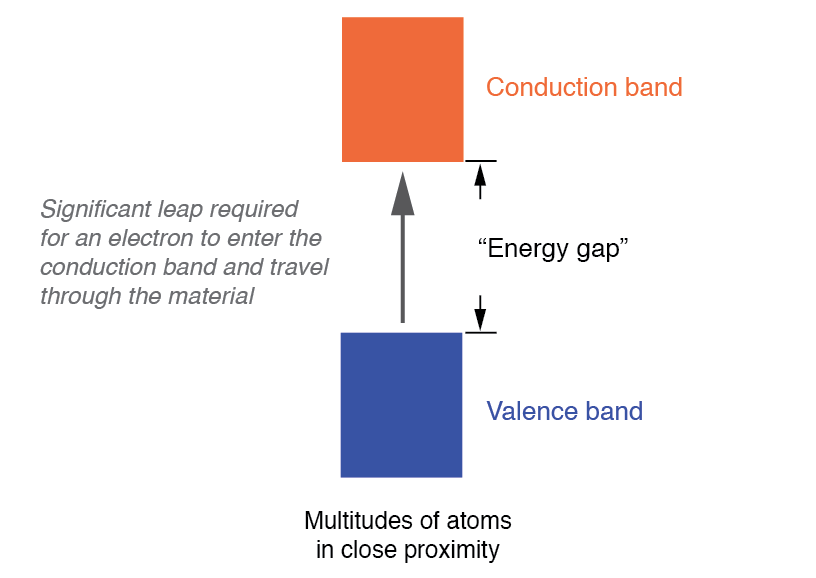
In contrast, insulators exhibit a substantial band gap between the highest filled band (valence band) and the next empty band (conduction band). Without enough energy to cross this gap, valence electrons remain bound, preventing electrical conduction. See the figure below.
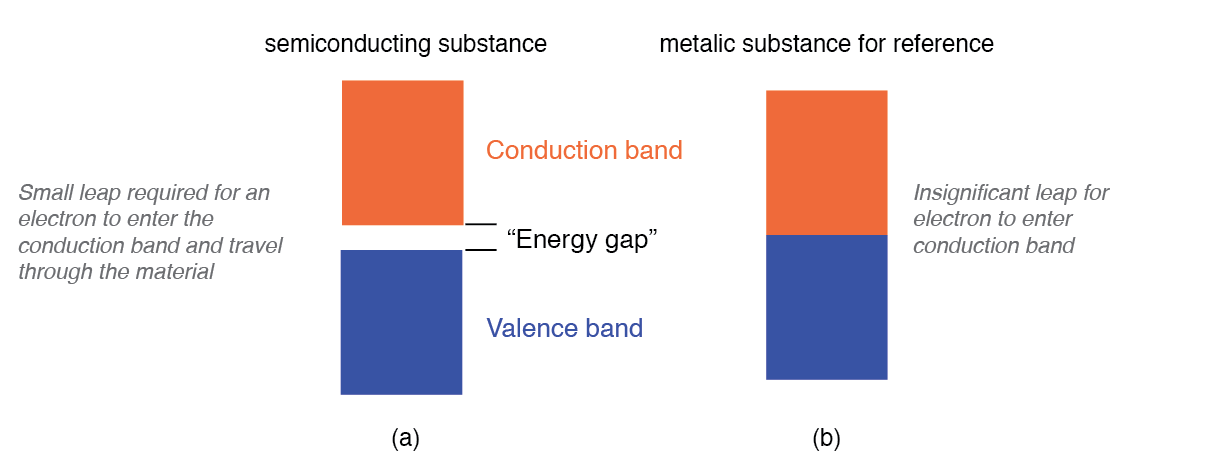
Semiconductors sit between these extremes, featuring a narrow band gap. At low temperatures, few electrons possess the thermal energy needed to cross the gap, so the material behaves like an insulator. As temperature rises, more electrons reach the conduction band, dramatically increasing conductivity. The figure below compares semiconductor and metallic band structures.
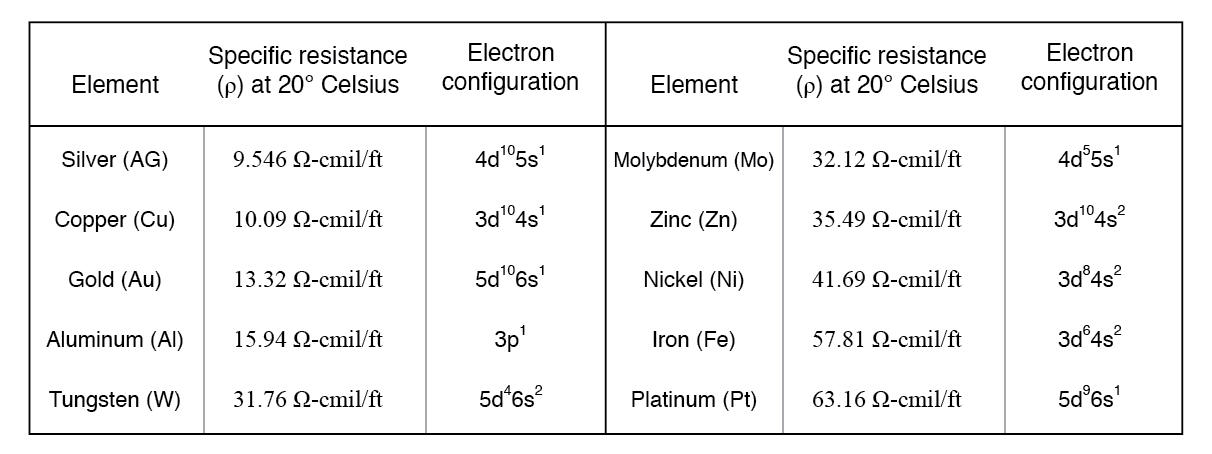
It is not always possible to predict a material’s conductivity from the electron configurations of its constituent atoms. For instance, while the best conductors—silver, copper, and gold—each have a single valence s‑electron, conductivity depends on how their bands overlap, not merely on electron count.
Key Takeaways
- Energy is required to promote an electron from the valence band to the conduction band; larger gaps mean higher energy.
- In metals, band overlap allows electrons to move with minimal energy, making them excellent conductors.
- Insulators have wide band gaps that prevent electron mobility, resulting in negligible conductivity.
- Semiconductors possess a small, non‑overlapping gap, enabling controlled conductivity that varies with temperature or doping.
Further Learning
Industrial Technology
- Amplifiers: Harnessing Active Devices to Boost Power
- Quantum Physics and the Atom: Foundations for Modern Electronics
- Understanding Band Theory of Solids: How Electron Energy Levels Shape Electrical Conductivity
- Understanding Electrons, Holes, and Doping in Semiconductors
- Understanding Voltage and Current: The Foundations of Electrical Flow
- Essential Electrical Safety: Zero Energy State & Lock‑Out/Tag‑Out Procedures
- Energy Band Alignment in Atomic‑Layer‑Deposited ZnO/β‑Ga₂O₃ (over 2 01) Heterojunctions Revealed by XPS
- On-Site Energy Solutions for a Carbon-Free Future
- 50‑Million‑Electron Pulse Enables Self‑Powered Sensors to Operate for Over a Year
- In-Depth Tutorial: Understanding the Fundamentals of Electron Emission



