Quantum Physics and the Atom: Foundations for Modern Electronics
“I think it is safe to say that no one understands quantum mechanics.” — Physicist Richard P. Feynman
Semiconductor technology is often called the cornerstone of the digital age. Its development unlocked miniaturized computers, advanced medical imaging, and ubiquitous communication devices. Behind this technological leap lies a deeper scientific revolution: quantum physics. Understanding the behavior of electrons at the atomic scale is essential for grasping how semiconductor devices work. Classical physics offers an incomplete picture, and without quantum insight, the extraordinary performance of modern electronics would remain unexplained.
Atom
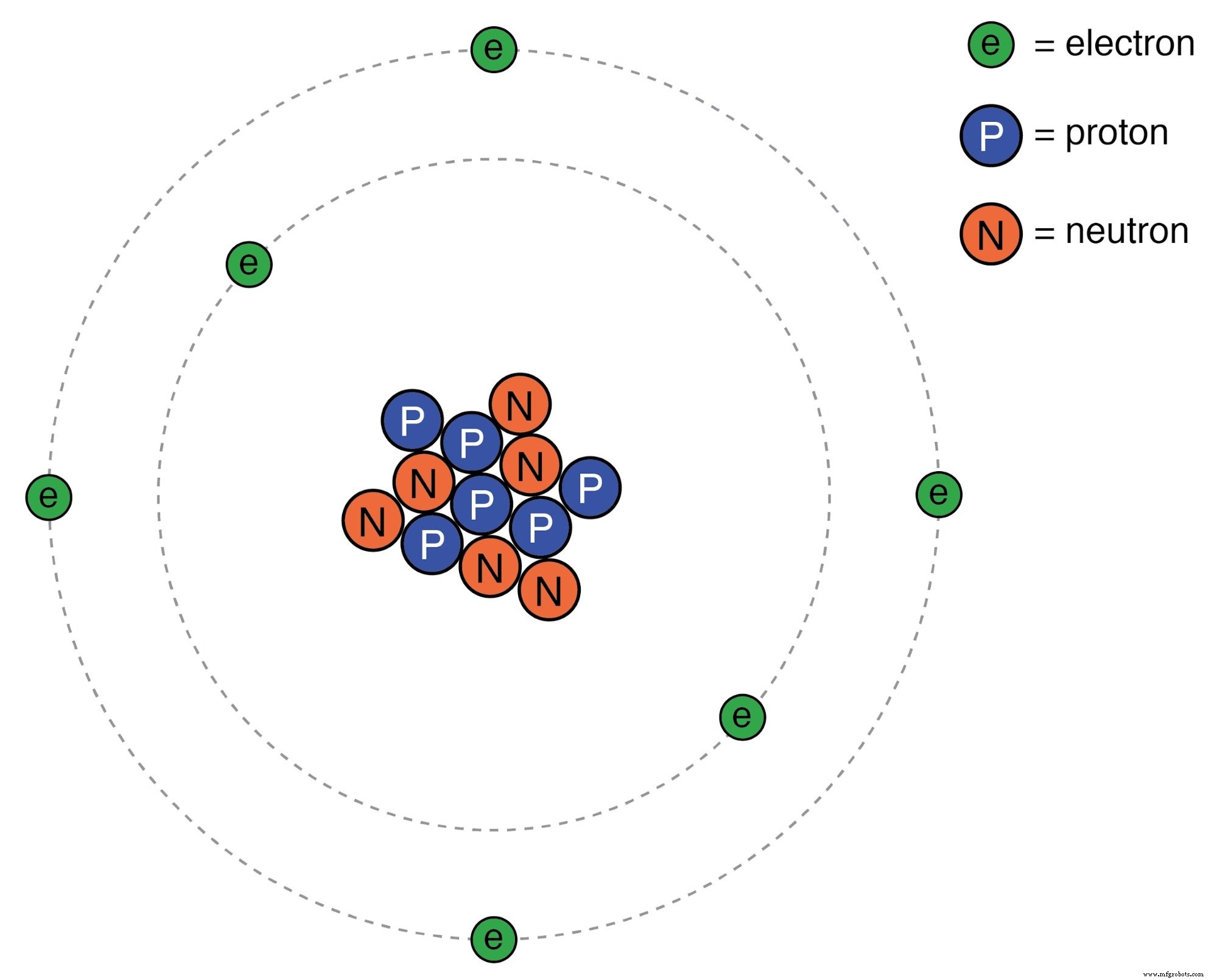
Early atomic models portrayed a miniature solar system, with negatively charged electrons orbiting a positively charged nucleus. This representation stems from Ernest Rutherford’s 1911 experiment, which revealed that the atom’s charge is concentrated in a tiny, dense core. Although the model captured the overall charge distribution, it ignored the quantum nature of electrons.
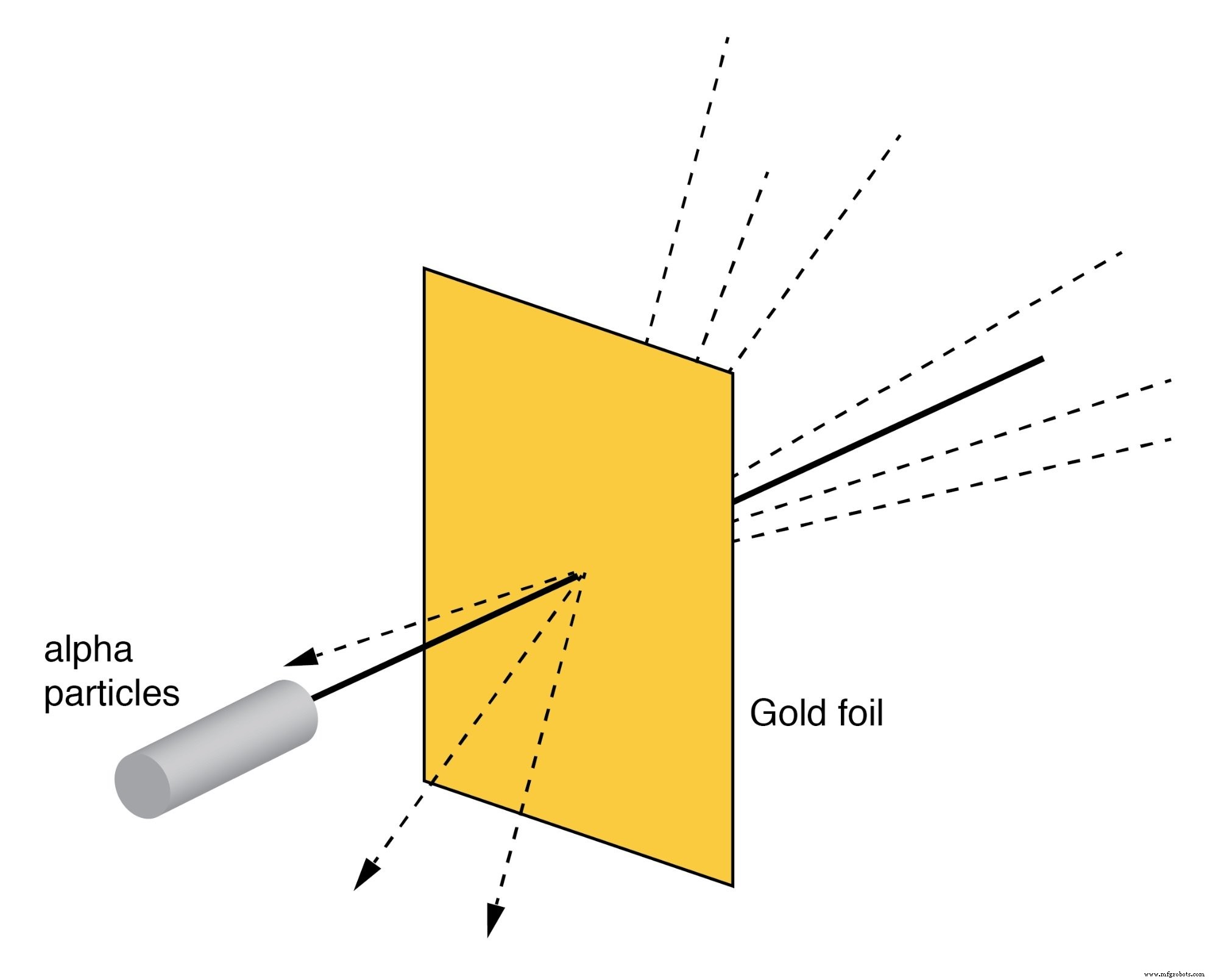
Rutherford’s Scattering
Rutherford’s scattering experiment bombarded a thin gold foil with alpha particles. Most particles passed through undeflected, indicating that atoms are largely empty space. However, a small fraction was deflected at large angles or even back-scattered, proving the existence of a tiny, positively charged nucleus. This experiment, performed by H. Geiger and E. Marsden, revolutionized our understanding of atomic structure.
Despite its success, the Rutherford model struggled to explain why electrons do not spiral into the nucleus. Classical explanations invoked a centrifugal force that balances electrostatic attraction, yet such a force does not exist for orbiting bodies. Moreover, accelerating charges should radiate energy—an effect observed in synchrotrons—but atomic electrons remain stable, contradicting classical expectations.
Excited Atoms
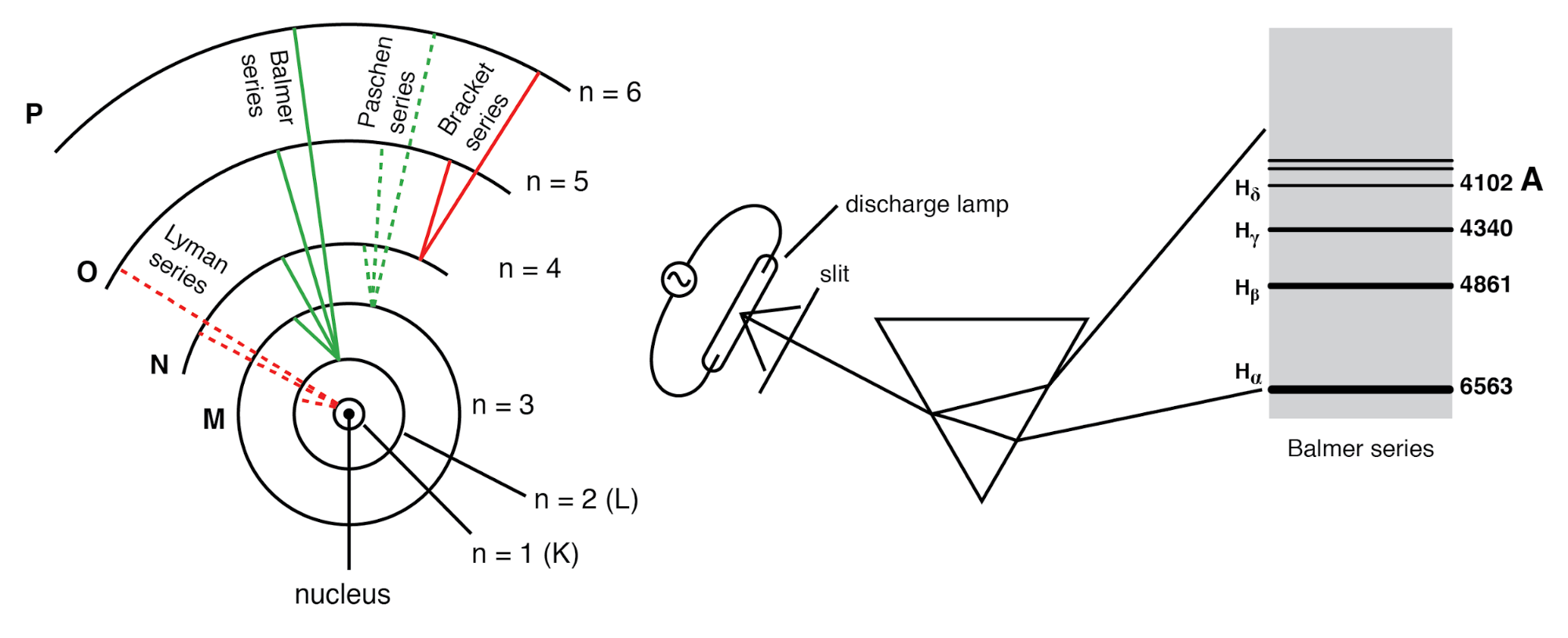
Experiments with excited atoms revealed that emitted light appears only at discrete frequencies. When an atom absorbs energy and later releases it, the resulting spectrum consists of sharp lines unique to each element—a phenomenon known as atomic spectroscopy. Classical physics predicts a continuous spectrum, so the discrete lines signal a fundamental quantum property of matter.
Bohr Model
In 1913, Niels Bohr incorporated Planck’s quantization into the atomic model. He proposed that electrons occupy specific, quantized orbits—stationary states—each associated with a discrete energy level. Bohr’s theory successfully explained the hydrogen Balmer series and other spectral lines. However, the model lacked a deeper physical mechanism for the quantization.
De Broglie Hypothesis
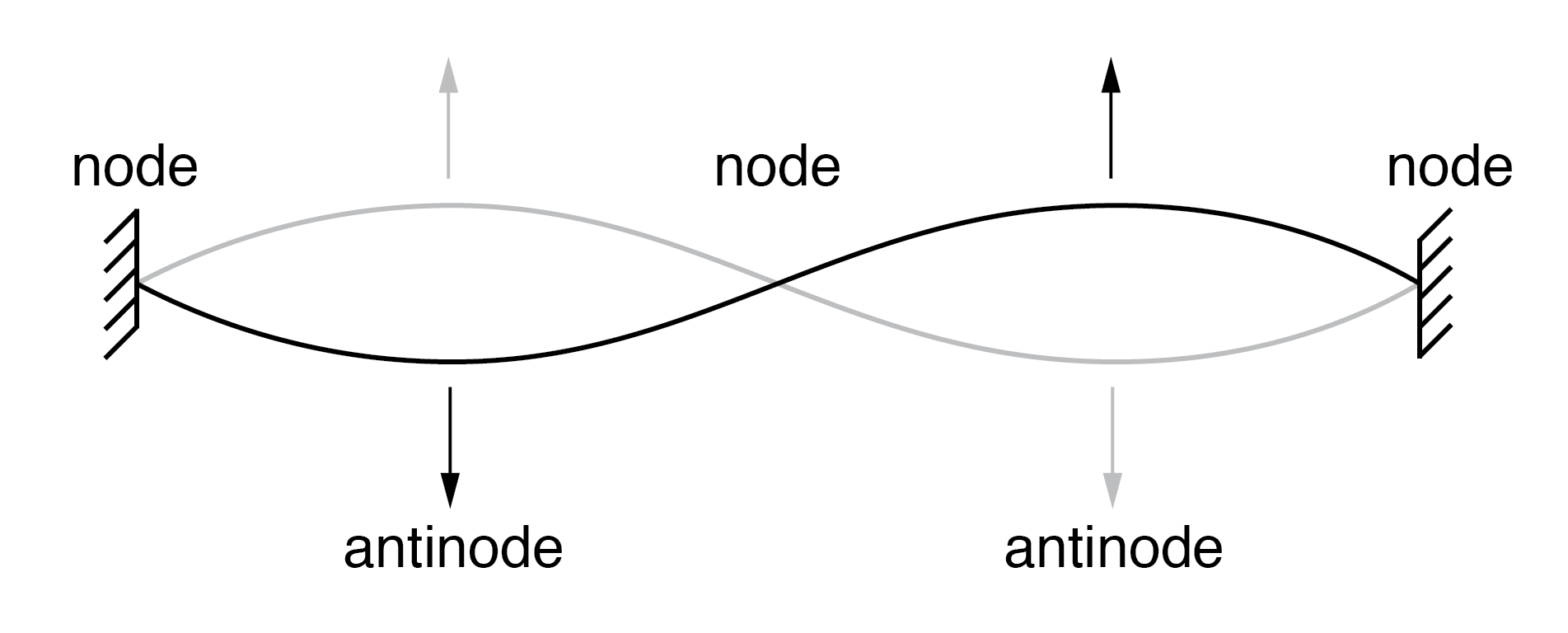
Louis de Broglie extended the wave–particle duality to matter in 1924, suggesting that electrons exhibit wave-like properties. He proposed that an electron’s standing wave pattern around the nucleus must fit an integer number of wavelengths, naturally leading to quantized orbits. This insight bridged the gap between Bohr’s postulates and the emerging wave mechanics.
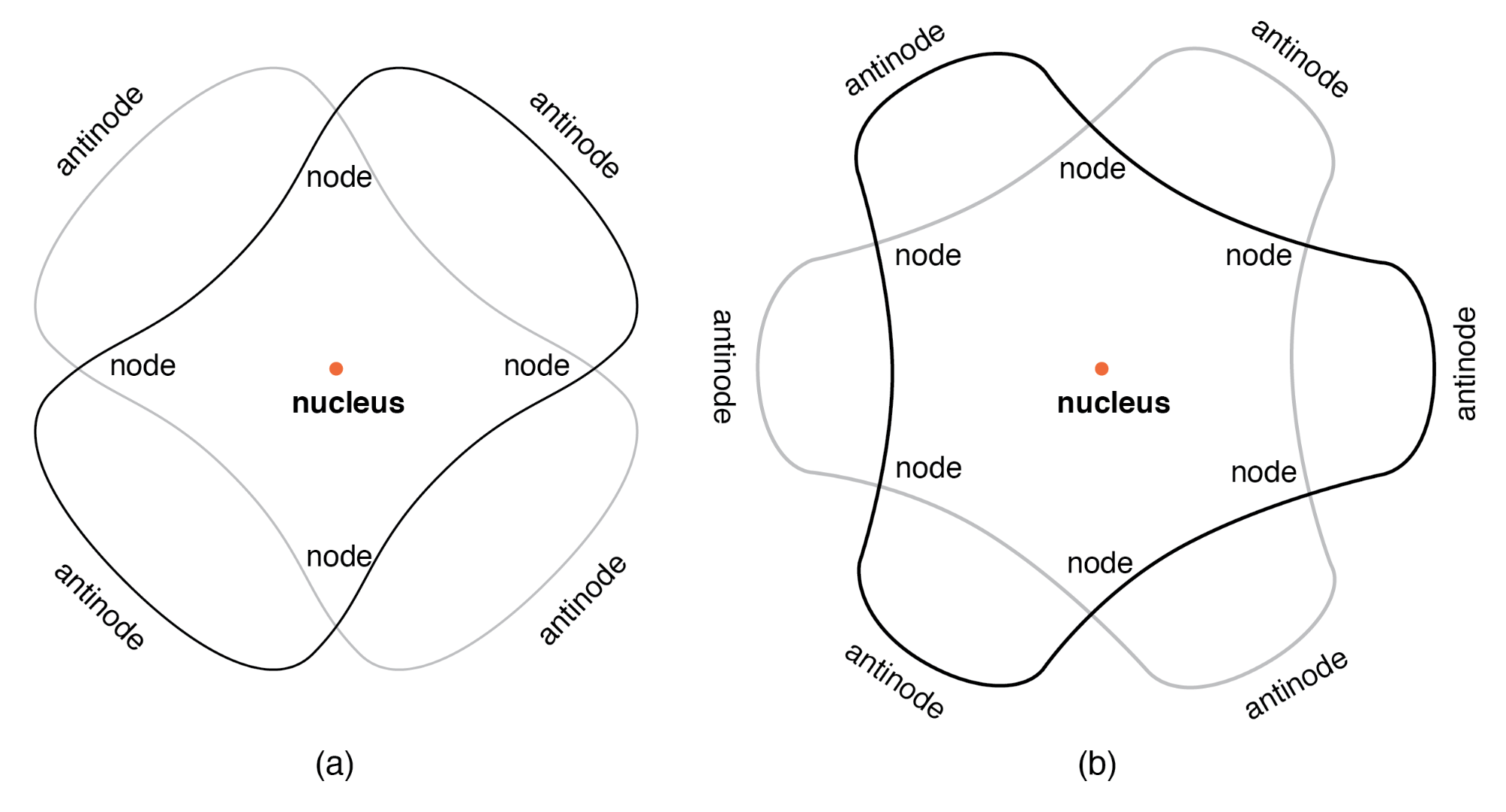
Quantum Mechanics
The formal development of quantum mechanics by Werner Heisenberg (matrix mechanics, 1925) and Erwin Schrödinger (wave mechanics, 1926) provided a mathematically rigorous framework. A central feature is the Heisenberg uncertainty principle, which states that position and momentum cannot both be known with arbitrary precision: ±ΔxΔp >= ½ħ. This principle is not a measurement limitation but an intrinsic property of quantum systems, giving rise to electron “clouds” rather than fixed orbits.
These probabilistic wavefunctions retain discrete energy levels, reconciling the quantized spectra observed experimentally with the inherently fuzzy nature of electron positions and momenta.
Four Quantum Numbers
Each electron in an atom is uniquely described by four integers or half‑integers:
- Principal quantum number (n): identifies the energy shell (n = 1, 2, 3, …). The maximum electron count in a shell is 2n2.
- Angular momentum quantum number (l): defines the subshell shape (l = 0 to n-1), corresponding to s, p, d, f, … orbitals.
- Magnetic quantum number (ml): specifies orbital orientation (ml = -l … +l).
- Spin quantum number (ms): represents intrinsic spin (+½ or -½).
These numbers explain the structure of electron clouds, the Pauli exclusion principle, and the chemical behavior of elements.
Pauli Exclusion Principle
Wolfgang Pauli formalized the rule that no two electrons in the same atom can share an identical set of quantum numbers. Consequently, each orbital can accommodate at most two electrons with opposite spins. This principle underlies the periodic arrangement of elements and the filling of electron shells.
Examples of electron configurations:
- Hydrogen: 1s1
- Helium: 1s2
- Lithium: 1s22s1
In spectroscopic notation, fully filled lower shells are often omitted to emphasize the valence configuration that determines chemical reactivity.
Periodic Table
The periodic table arranges elements by increasing atomic number, revealing trends in chemical behavior. Elements with completely filled outer shells (noble gases) exhibit minimal reactivity, whereas those with partially filled valence shells readily form bonds. Mendeleev’s original table, organized by atomic mass, was later refined to reflect atomic number, enabling accurate predictions of undiscovered elements.
Quantum Mechanics From Hypothesis to Theory
From its speculative beginnings, quantum mechanics has evolved into a predictive, experimentally validated theory. While some aspects remain incomplete—particularly regarding the transition from microscopic to macroscopic behavior—its success in describing atomic spectra, semiconductor physics, and countless other phenomena underscores its foundational role in modern science.
Review
- Electrons exist as probabilistic clouds, not orbiting particles.
- Four quantum numbers uniquely define an electron’s state.
- Principal quantum number (n) determines shell energy and radius.
- Angular momentum quantum number (l) defines subshell shape.
- Magnetic quantum number (ml) specifies orbital orientation.
- Spin quantum number (ms) can be +½ or -½.
- Pauli’s exclusion principle limits electron occupancy per orbital.
- Spectroscopic notation concisely represents electron configurations.
- Chemical behavior is governed by valence electrons; filled shells are inert.
- Elements with filled outer shells are noble gases.
Related Worksheets
- Atomic Structure Worksheet
- Basic Electricity Worksheet
Industrial Technology
- Valence Electrons, Crystal Structures, and Their Role in Conductivity
- Understanding Band Theory of Solids: How Electron Energy Levels Shape Electrical Conductivity
- Understanding Electrons, Holes, and Doping in Semiconductors
- Quantum Devices: Beyond CMOS – Harnessing Tunneling, Quantum Dots, and Spintronics for Future Electronics
- Understanding Voltage and Current: The Foundations of Electrical Flow
- How Batteries Convert Chemistry into Electricity
- Understanding Conductors and Insulators: From Quantum Mechanics to Practical Applications
- Quantum Financial Systems: A Clear, Practical Overview
- Quantum Physics Unveiled: A Poetic Journey into Topological Computing
- What is Quantum Computing? A Beginner’s Guide



