Understanding Conductors and Insulators: From Quantum Mechanics to Practical Applications
Fundamental Concepts of Electrical Conductivity
Electrical conductivity depends on how freely electrons can move within a material. Materials that allow electrons to travel easily are called conductors, while those that restrict electron motion are called insulators.
Classical “planetary” models of electrons are inadequate; quantum mechanics shows that electrons occupy discrete energy zones—shells and subshells—around the nucleus. These zones can only hold electrons up to specific energy limits, creating a quantized structure that determines whether a material conducts or resists electrical flow.
Excited-State vs. Ground-State Electrons
When an electron occupies a higher-energy shell, it may detach from its atom and become part of an electric current—this is typical of conductive materials. Conversely, if quantum restrictions confine an electron to its ground state, it remains bound and cannot readily contribute to current flow—characteristic of insulators.
Beyond Valence Electrons: The Complexity of Conductivity
While textbook explanations often link conductivity to the number of valence electrons, real materials exhibit much richer behavior. Conductivity must be evaluated in the context of molecular bonding and the overall electronic structure of compounds.
Case Study: Carbon Allotropes
Graphite and diamond are both made of carbon atoms (6 p, 6 n, 6 e), yet they differ dramatically in conductivity. Graphite’s planar layers allow delocalized electrons to move freely, making it a decent conductor. Diamond’s tetrahedral structure localizes electrons, rendering it an insulator (in pure form, a semiconductor that conducts only under high temperature or doping).
Doping and Semiconductors
Introducing impurities—known as doping—modifies a semiconductor’s electrical, optical, and structural properties. For instance, silicon carbide (SiC) exhibits a negative differential resistance: its resistance drops as voltage increases. Hydrocarbon oils are excellent insulators, illustrating that valence counts alone are insufficient predictors of conductivity.
Metallic Conductivity
All metals conduct electricity effectively because their valence electrons form an “electron gas” or “electron sea,” freely moving between nuclei. This mobility explains metals’ high thermal conductivity, malleability, ductility, and luster.
Practical Considerations: Resistance and Voltage Drop
Every conductor has measurable resistance, even if it is small. In circuits, resistance causes a voltage drop along the conductor. Although ideal calculations often assume zero resistance, real-world designs must account for these drops to predict performance accurately.
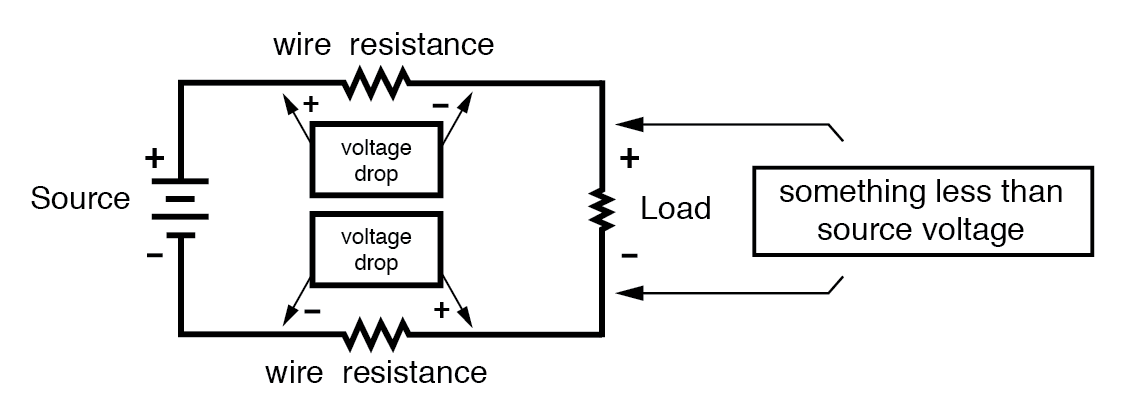
Review
- Conductivity is governed by electron configuration in atoms and molecules.
- All ordinary conductors exhibit some resistance.
- Current through a conductor with resistance produces a measurable voltage drop.
Related Worksheets
- Conductors and Insulators Worksheet
- Specific Resistance of Conductors Worksheet
By understanding these principles, engineers can design efficient electrical paths and appropriate insulation for modern technology.
Industrial Technology
- Valence Electrons, Crystal Structures, and Their Role in Conductivity
- Understanding Electrons, Holes, and Doping in Semiconductors
- Diodes and Rectifiers: Fundamentals, Operation, and Key Parameters
- Understanding Conductors, Insulators, and the Science of Electron Flow
- Understanding Voltage and Current: The Foundations of Electrical Flow
- How Batteries Convert Chemistry into Electricity
- Understanding Conductance, Susceptance, and Admittance in AC Circuits
- The Revolution of Semiconductors: A Comprehensive Introduction
- 4047 IC Explained: Mastering Monostable & Astable Multivibrators
- Understanding PCB Gerber Files: Key Role in Electronics Manufacturing



