Understanding Conductors, Insulators, and the Science of Electron Flow
Electrons in different atoms exhibit varying degrees of freedom. In metals, outer electrons are loosely bound, moving chaotically between atoms under the influence of room‑temperature heat. These free electrons can drift freely, giving metals their high conductivity.
Conductors vs Insulators
Materials like glass hold their electrons tightly, so they hardly move. While mechanical rubbing can force some electrons to detach and transfer to adjacent atoms, internal mobility remains limited. This relative ease of electron movement is known as electric conductivity and depends on the atomic composition and bonding structure of the material.
Materials with abundant free electrons are conductors; those with few or none are insulators. Common examples include:
- Conductors
- silver
- copper
- gold
- aluminum
- iron
- steel
- brass
- bronze
- mercury
- graphite
- dirty water
- concrete
- Insulators
- glass
- rubber
- oil
- asphalt
- fiberglass
- porcelain
- ceramic
- quartz
- (dry) cotton
- (dry) paper
- (dry) wood
- plastic
- air
- diamond
- pure water
Not all conductors are equal. Silver has the highest conductivity (≈6.3×107 S/m), followed closely by copper (≈5.96×107 S/m). Non‑metal conductors such as dirty water or concrete are far less conductive than metals. Likewise, insulators vary in their resistance; glass is a better insulator than most plastics, and even clear fiberglass still blocks electric flow more effectively than window glass.
Materials can change their electrical behavior with temperature. Glass, an excellent insulator at room temperature, becomes conductive when heated to extreme temperatures. Gases like air, normally insulating, ionize and conduct at high heat. Metals generally lose conductivity as temperature rises, whereas superconductors emerge at cryogenic temperatures, achieving zero resistance.
Electron Flow / Electric Current
In a conductor, free electrons move randomly. However, an external electric field aligns their motion, creating a uniform drift—this is what we call electricity or electric current. It is the dynamic counterpart to static electricity, where charge accumulates without motion.
Imagine a pipe filled with marbles: inserting one marble at the left end forces another to exit at the right almost instantaneously, regardless of pipe length. In reality, the electric signal travels at the speed of light (≈186,000 miles per second), while each electron drifts slowly. This phenomenon explains why circuits respond so quickly.
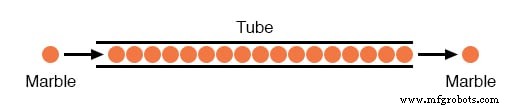
Electron Flow Through Wire
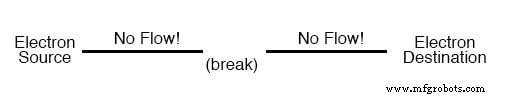
To guide electrons in a desired direction, we install wires—highly conductive metals such as copper or aluminum—of appropriate gauge. Current exists only where a continuous conductive path is present. A closed loop is essential; a break in the wire blocks flow, just as a capped pipe stops water.

In the diagram, the solid line represents a continuous copper wire. Adding a hypothetical Electron Source on the left and a Destination on the right shows current flowing when the path is intact. If the wire is severed, as illustrated, the flow stops completely.
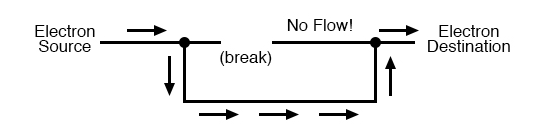
Electrical Continuity
Air gaps act as insulators, breaking continuity. Restoring contact—by touching two wire ends together—reestablishes a complete circuit, allowing electrons to flow again. This is akin to adding a tee fitting to reconnect a broken pipe.
Unlike water pipes, metal wires do not suffer wear from electron flow; however, the friction of electrons (resistive heating) can generate heat, which we will discuss in a later section.
Review
- In conductive materials, outer electrons are free to move (free electrons).
- In insulating materials, outer electrons are tightly bound.
- All metals are electrically conductive.
- Dynamic electricity (electric current) is the coordinated drift of electrons.
- Static electricity is stationary charge accumulation on insulators.
- Continuous current requires an unbroken conductive path.
Related Worksheets
- Conductors and Insulators Worksheet
Industrial Technology
- Valence Electrons, Crystal Structures, and Their Role in Conductivity
- Understanding Electrons, Holes, and Doping in Semiconductors
- Understanding Voltage and Current: The Foundations of Electrical Flow
- Conventional vs. Electron Flow: What Engineers Use and Why
- How Batteries Convert Chemistry into Electricity
- Understanding Conductors and Insulators: From Quantum Mechanics to Practical Applications
- NASA Goddard’s Lab‑On‑Chip Sensor Delivers Real‑Time Flow & Temperature Monitoring
- In-Depth Tutorial: Understanding the Fundamentals of Electron Emission
- Water Flow Sensors: How They Work & How to Use Them
- Understanding Pressure vs. Flow in Compressed Air Systems



