How Batteries Convert Chemistry into Electricity
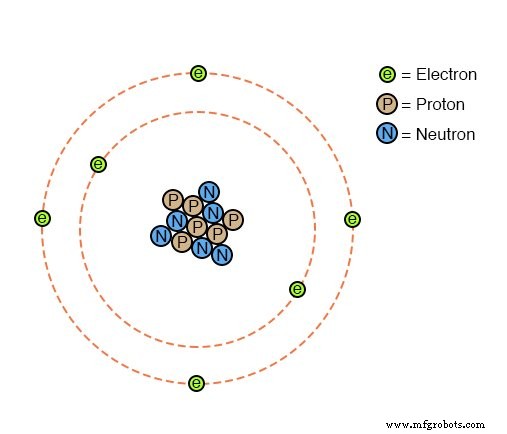
While earlier chapters introduced the atom and its constituent particles—electrons, protons, and neutrons—this section dives into the practical side of batteries. We explore the chemistry that turns chemical energy into electrical energy, and examine the real-world considerations for power systems.
Electrons are the active agents in electrical activity, but an atom’s chemical identity—and therefore its conductivity—is governed by the number of protons in its nucleus. The protons are tightly bound, making the elemental identity of an atom remarkably stable. In contrast, electrons can be displaced more readily, allowing for friction‑induced charge transfer (e.g., glass and silk) or thermal effects such as thermocouple voltage generation.
Types of Chemical Bonds
Electrons not only shuttle between atoms; they also mediate bonds that hold atoms together. These chemical bonds give rise to molecules with unique physical properties. The most common bond types are covalent and ionic:
- Covalent bond: electrons are shared between atoms. For example, a simple molecule of two identical atoms shows shared electron density.
- Ionic bond: one atom donates electrons, becoming positively charged, while another accepts electrons, becoming negatively charged. Electrostatic attraction holds the ions together.
In reality, molecules often contain a mix of covalent and ionic character, as illustrated by sodium hydroxide (NaOH). The Na⁺ ion forms an ionic bond with the hydroxide ion, while the O–H pair within the hydroxide is covalently bonded.
Atomic oxidation states are indicated by superscripts or Roman numerals (e.g., Pb²⁺, Fe³⁺). Knowing the oxidation state clarifies which atoms are being reduced or oxidized during a reaction. The mnemonic OIL RIG (Oxidation Is Loss, Reduction Is Gain) helps remember that oxidized species lose electrons, while reduced species gain electrons.
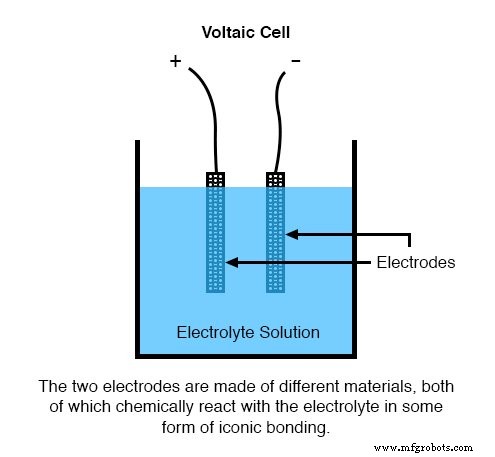
How a Voltaic Cell Works
A voltaic cell—or simply a cell—converts the movement of electrons during redox reactions into usable electric current. The typical configuration includes two metal electrodes immersed in an electrolyte that facilitates ion transport. When the electrodes are connected externally, electrons flow from the anode to the cathode, generating current.
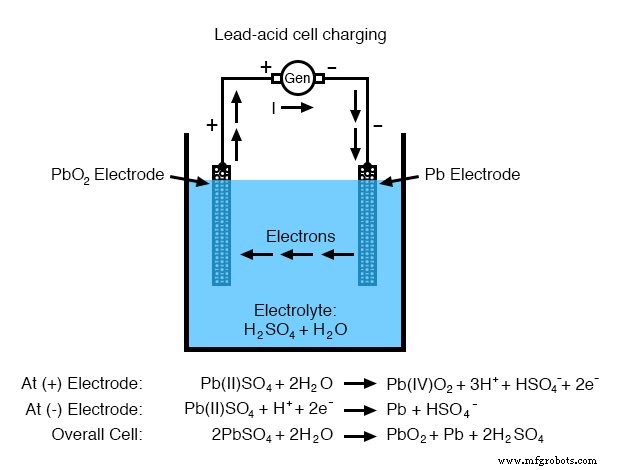
Consider the lead‑acid battery, ubiquitous in automotive applications. The negative electrode is metallic lead (Pb), and the positive electrode is lead(IV) dioxide (PbO₂). The electrolyte is a dilute solution of sulfuric acid (H₂SO₄). During discharge, PbO₂ at the cathode accepts electrons to form PbO, while water is generated from H⁺ and O²⁻. Simultaneously, Pb at the anode releases electrons, forming PbSO₄ that combines with sulfate ions in the electrolyte. The overall cell voltage is approximately 2.04 V under fully charged, optimal conditions.
Note on nomenclature: In lead chemistry, PbO refers to lead(II) oxide, whereas PbO₂ (often called lead dioxide or plumbic oxide) denotes lead(IV) oxide. The context usually clarifies which compound is meant.
When a lead‑acid battery is fully discharged, the electrolyte becomes largely water, and both electrodes are coated with PbSO₄, halting further current flow. The state of charge can be monitored with a hydrometer that measures the specific gravity of the electrolyte—higher density indicates a higher acid concentration and a more charged cell.
Not all cells are reversible. Primary cells, such as the Edison cell (NiO/Fe/KOH), cannot be recharged because the electrochemical reaction is effectively irreversible under practical conditions. Secondary cells, like lead‑acid or lithium‑ion batteries, allow the reaction to be reversed by applying an external current, thereby restoring the original electrode materials.
Key Takeaways
- Atoms bound by electrons form molecules.
- Ionic bonds arise when one atom donates electrons and another accepts them.
- Redox reactions transfer electrons and can be harnessed to generate electric current.
- A voltaic cell converts chemical reactions into electrical energy.
- Discharged cells have depleted their chemical energy; recharging restores it in secondary cells.
- Lead‑acid charge can be gauged via electrolyte density measured with a hydrometer.
Related Worksheets
- Atomic Structure Worksheet
Industrial Technology
- Understanding Electron Tubes: Components and Functionality
- Quantum Physics and the Atom: Foundations for Modern Electronics
- Understanding Electrons, Holes, and Doping in Semiconductors
- A Primer on Vacuum (Electron) Tubes: Foundations of Modern Electronics
- Understanding Conductors, Insulators, and the Science of Electron Flow
- Understanding Conductors and Insulators: From Quantum Mechanics to Practical Applications
- CPI Maintenance Spending Rises: 2008 Turnarounds Exceed $521 M, Up 17% in H2
- Polyurethane: Advanced Chemical Resistance for Durable Applications
- Polyurethane vs Polyethylene: Choosing the Right Plastic for Your Design
- In-Depth Tutorial: Understanding the Fundamentals of Electron Emission



