Battery Construction Fundamentals: Cells, Internal Resistance, and Connectivity
The term battery denotes a group of similar components. In military jargon it refers to a cluster of guns; in electronics it describes a set of voltaic cells combined to deliver higher voltage and/or current than a single cell can provide.
Electrical symbols are intentionally simple: a single cell is represented by a long line and a short line, parallel to each other, with connecting wires.
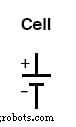
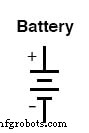
A battery symbol is just multiple cell symbols stacked in series, indicating that the cells are connected so their voltages add.
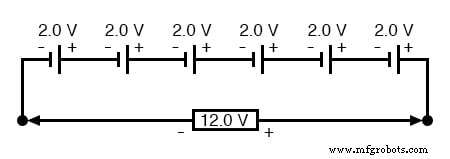
As previously noted, the voltage of a cell is fixed by its chemistry; the cell’s physical size does not alter its nominal voltage. To achieve a higher total voltage, cells must be wired in series, and the battery’s output voltage equals the sum of the individual cell voltages. For example, a standard automotive lead‑acid battery contains six 2.0‑volt cells, yielding a nominal 12.0‑volt output:
In automotive batteries, the cells share a single hard rubber housing. Thick lead bars, rather than thin wires, interconnect the cells. Each cell’s electrodes and electrolyte are isolated in partitioned sections of the case. In large‑capacity batteries, electrodes are often large metal grids or plates, commonly called “plates.”
For simplicity, schematic symbols typically show only four lines—alternating long/short—even if the real battery contains many more cells. Occasionally you will see a battery symbol with extra lines to denote unusually high voltages; each line represents an individual cell plate:

How Does Battery Size Influence Performance?
While a cell’s size does not affect its voltage, it does influence internal resistance. Larger cells provide a greater electrode surface area, reducing the contact resistance between electrodes and electrolyte. Lower internal resistance allows a cell to deliver higher current to a load.
In theory, a cell is treated as an ideal voltage source. In practice, every cell has internal resistance that limits current delivery. The following example illustrates this effect:
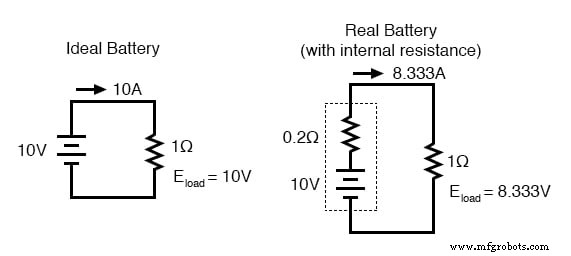
The dotted diagram shows a real battery with 0.2 Ω internal resistance powering a 1 Ω load. An ideal battery (no internal resistance) would provide 10 A (10 V / 1 Ω). The real battery, constrained by its internal resistance, delivers only 8.33 A. In a short‑circuit (0 Ω load), the ideal source would supply infinite current, whereas the real battery is limited to 50 A (10 V / 0.2 Ω). The cell’s chemistry still delivers 10 V, but part of that voltage is dropped across the internal resistance, reducing the terminal voltage available to the load.
Reducing Internal Resistance Through Parallel Connection
In many applications, a battery’s internal resistance is negligible compared to the load resistance, so its performance closely approximates an ideal source. When a lower overall resistance is required—for higher current capacity—the cells should be wired in parallel:
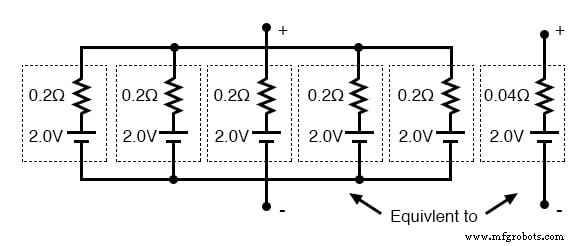
Connecting five identical cells in parallel yields a Thevenin equivalent of a single 2.0‑V source with an internal resistance that is one‑fifth of a single cell’s resistance. The total voltage remains 2.0 V, while the current capacity increases because each cell shares the load current.
Review- A battery is a cluster of cells assembled to increase voltage and/or current output.
- Series connection (polarity‑aligned) adds cell voltages.
- Physical cell size lowers internal resistance, enhancing current delivery.
- Parallel connection reduces equivalent internal resistance, raising available current.
- Batteries Worksheet
- Thevenin’s, Norton’s, and Maximum Power Transfer Theorems Worksheet
Industrial Technology
- Exploring Nonlinear Resistance in Incandescent Lamps: A Practical Lab Guide
- Voltage Divider Lab: Design, Measurement, and Kirchhoff’s Voltage Law Verification
- Build a Potato Battery: A Step‑by‑Step Guide to DIY Electrochemical Power
- Understanding Relay Construction: From Solenoids to Industrial Applications
- Understanding Electrical Resistance and Circuit Safety
- Resistors: Fundamentals, Types, and Practical Applications
- Understanding Conductance: The Inverse of Resistance
- Bridge Circuits: Wheatstone, Kelvin, and Their Role in Precise Electrical Measurements
- Calculating Wire Resistance for Voltage‑Drop‑Critical Circuits
- Measuring a Battery’s Internal Resistance: A Practical Guide



