Build a Potato Battery: A Step‑by‑Step Guide to DIY Electrochemical Power
Overview
A potato battery demonstrates the fundamentals of electrochemistry in a simple, hands‑on experiment. By inserting two dissimilar metals into a potato, you create a tiny cell that generates a measurable voltage.
Parts and Materials
- One large potato (any variety works; larger potatoes yield slightly higher voltage)
- One lemon (optional, can serve as a reference or alternative electrolyte)
- Strip of zinc, or a galvanized nail (thick, rough zinc coating preferred)
- Piece of thick copper wire or a copper strip
Scientific Background
When the zinc electrode oxidises (Zn → Zn²⁺ + 2e⁻) electrons flow through the external circuit to the copper electrode, where they reduce copper ions (Cu²⁺ + 2e⁻ → Cu). The potato’s acidic interior provides the necessary ions, and the typical cell voltage is about 0.8 V.
Cross‑References
Lessons In Electric Circuits, Volume 1, chapter 11: “Batteries and Power Systems.”
Learning Objectives
- Understand how chemical activity drives battery operation.
- Explore the impact of electrode surface area on voltage output.
Illustration
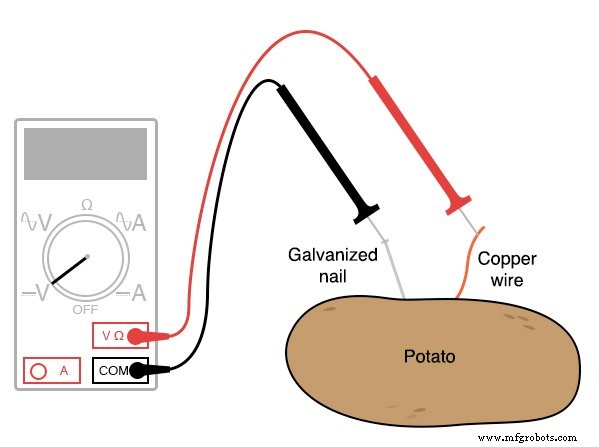
Instructions
- Insert the zinc nail and copper wire deep into opposite sides of the potato, leaving enough exposed to connect wires.
- Use a voltmeter to measure the open‑circuit voltage between the two electrodes.
- Experiment by varying electrode depth, spacing, and metal type to maximise voltage.
- Try other fruits or vegetables and compare the results using the same electrode configuration.
- Remember that a single potato cell typically cannot power a lamp or motor due to high internal resistance; you’ll need multiple cells in series or parallel to achieve sufficient voltage and current.
Tips for Better Performance
- Use a larger, rough‑textured galvanized nail to increase surface area and reduce resistance.
- For higher voltage, connect several potato cells in series (positive to negative). For higher current, connect cells in parallel (positive to positive).
- Keep electrodes from short‑circuited by adding a small insulating spacer.
- Monitor the potato’s condition; as the electrolyte depletes, voltage will drop.
By following these steps, you’ll gain hands‑on insight into how simple materials can create electrical energy, and you’ll learn how to optimise cell performance through experimental design.
Industrial Technology
- Exploring Voltage Addition with Series Battery Connections
- Voltage Divider Lab: Design, Measurement, and Kirchhoff’s Voltage Law Verification
- Thermoelectricity: Understanding Thermocouples and the Seebeck Effect
- Potentiometric Voltmeter: Precise Voltage Measurement with Minimal Loading
- Low‑Voltage AC Power Supply: Phase‑Shift Circuit Components & Best Practices
- Voltage Regulator Experiment with a 12‑Volt Zener Diode
- Voltage Follower Amplifier: Design, Build, and Measurement Guide
- Designing a High‑Gain Multi‑Stage Common‑Emitter Amplifier with Negative Feedback
- Tachogenerators: Precision Speed Measurement for Industrial Motors and Equipment
- Understanding AC Waveforms: Sine Waves, Frequency, and Oscilloscope Basics



