Understanding Battery Capacity: Amp-Hour Ratings and Condition Testing
Batteries generate current by exchanging electrons in ionic reactions. Because the number of reactable molecules is finite, a battery can only deliver a limited amount of charge before its energy reserves are depleted. While it’s theoretically possible to measure capacity by counting electrons, the resulting numbers are unwieldy. The practical unit that emerged is the amp‑hour, which ties directly to the more familiar coulomb (1 Ah = 3600 C). An ampere is a flow of one coulomb per second, so over an hour (3600 s) that equals 3600 coulombs. Using amp‑hours makes it easy to compare the energy a battery can provide to a load.
Amp‑Hour Application to Measure Battery Capacity
An amp‑hour rating tells you how long a battery can sustain a particular current before it is fully discharged. For example, a 70 Ah automotive battery, rated at 3.5 A, can supply that current for 20 hours (70 Ah ÷ 3.5 A). The relationship is linear in an ideal battery, but real cells exhibit variations. High currents raise internal temperature, altering chemical kinetics and shortening the actual runtime. Conversely, very low currents waste capacity to self‑discharge, evaporation, and leakage. Manufacturers often provide derating curves that show how capacity falls off at higher currents or temperatures.
Secondary cells use the amp‑hour figure to estimate charging times. The same 70 Ah battery would need roughly 10 hours to recharge from a dead state at a steady 7 A charge current (70 Ah ÷ 7 A).
Typical amp‑hour capacities for common batteries:
- Automotive lead‑acid: 70 Ah at 3.5 A (secondary)
- D‑cell carbon‑zinc: 4.5 Ah at 100 mA (primary)
- 9 V carbon‑zinc: 400 mAh at 8 mA (primary)
Checking Battery Condition – With and Without Load
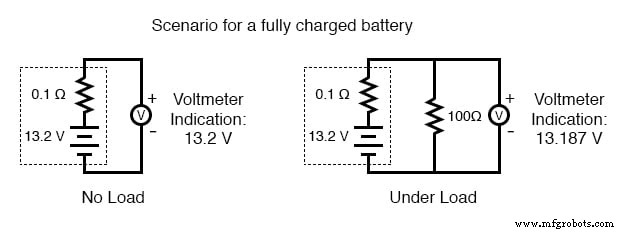
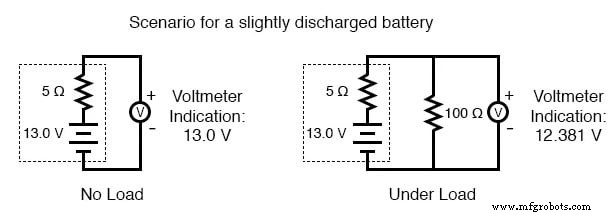
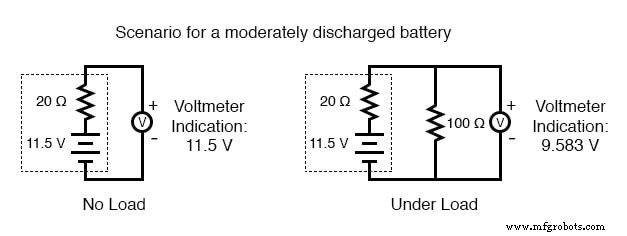
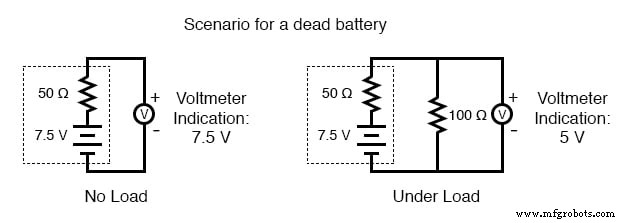
As a battery discharges, its internal resistance rises and its open‑circuit voltage falls. The most reliable indicator of health is a voltage measurement taken while the battery is supplying a substantial load. A simple voltmeter check can mask increased resistance and give a false impression of a healthy cell.
Typical measurements:
- Fully charged: 13.2 V (illustrated in the accompanying image)
- Partially discharged: 12.5 V (still usable but lower than full)
- Near dead: 7.5 V (immediately indicates a failed battery)
To perform a load test, connect a resistor rated for the expected power dissipation (several hundred watts for a car battery) and measure the voltage drop. A significant drop confirms a high internal resistance and likely reduced capacity.
Review:
- The amp‑hour is a unit of energy capacity equal to the product of continuous current and discharge time before a battery’s chemical energy is exhausted.
- Ratings are approximations and should be trusted only at the current or time specified by the manufacturer; extrapolation to very high currents or long times is unreliable.
- Discharged batteries lose voltage and increase resistance. The most accurate assessment of a dead battery is a voltage test under load.
Related Worksheets:
- Batteries Worksheet
- Basic Voltmeter Use Worksheet
Industrial Technology
- Optimizing Battery Performance: Parallel & Series‑Parallel Configurations for High‑Load Lamps
- Voltage Divider Lab: Design, Measurement, and Kirchhoff’s Voltage Law Verification
- Hands‑On Guide to Current Dividers: Build, Measure, and Simulate with a 6 V Battery
- Build a Potato Battery: A Step‑by‑Step Guide to DIY Electrochemical Power
- Comprehensive Guide to Diode Ratings & Datasheet Parameters
- Common-Emitter Amplifier Limitations: Distortion, Temperature, and High‑Frequency Challenges
- Insulated‑Gate Bipolar Transistors (IGBTs): Merging FET Precision with BJT Power
- DIAC: The Bidirectional Trigger for AC Thyristors
- Understanding Electrical Resistance and Circuit Safety
- Calculate Battery Charging Time & Current: Step‑by‑Step Guide with 120Ah Example



