Static Electricity Explained: How Rubbing Materials Generates Charge
Long before modern physics, scientists observed that certain materials attracted each other after being rubbed. For example, rubbing silk against glass makes both surfaces cling together, and the attraction persists even when the objects are separated.
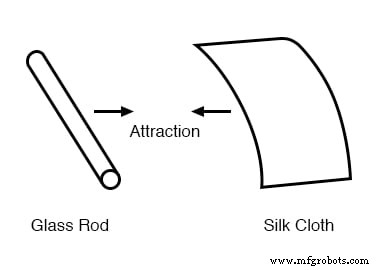
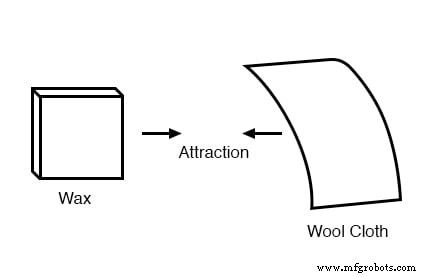
Glass and silk aren’t the only materials that behave this way. Anyone who has brushed a latex balloon against their hair knows how the balloon will cling to the hair. Early experimenters also noted that rubbing paraffin wax with wool produced a similar attraction.
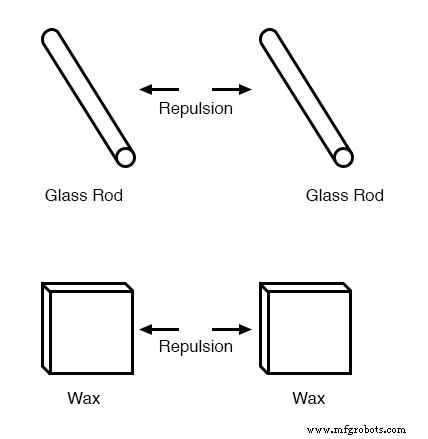
The phenomenon became even more intriguing when researchers discovered that identical materials, after being rubbed with their own cloths, always repel each other.
Furthermore, a piece of glass rubbed with silk attracted a piece of wax rubbed with wool.
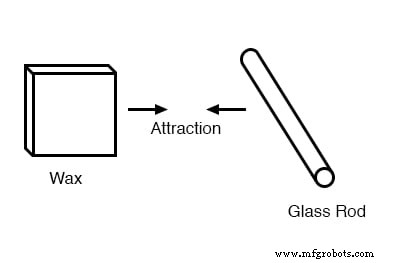
All materials that showed attraction or repulsion could be grouped into two categories: those attracted to glass and repelled by wax, and those repelled by glass and attracted to wax. No material was ever found to be attracted to both or repelled by both.
Attention then shifted to the cloths used for rubbing. Rubbing two glass pieces with two silk cloths caused both the glass and the cloths to repel each other. The same pattern appeared when wool rubbed wax.
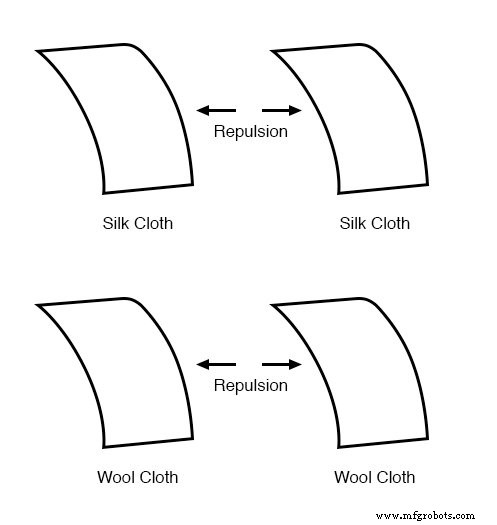
What’s remarkable is that none of these objects showed visible changes after rubbing, yet their interactions altered dramatically. The underlying change was invisible.
Early scientists speculated that an invisible “fluid” was transferred during rubbing, exerting a force over a distance. Charles Dufay was among the first to demonstrate that rubbing produced two distinct effects—attraction and repulsion—leading to the concept of charge.
Benjamin Franklin proposed that a single fluid was exchanged between rubbed objects, and that the two observed charges were simply an excess or deficiency of this fluid. Experiments with wax and wool suggested that wool removed fluid from wax, leaving wool with an excess and wax with a deficiency, thereby creating attraction as the fluid tried to rebalance.
Franklin’s hypothesis explained why all rubbed materials fell into one of two opposing categories and why they always attracted each other. He also named the charge of wax “negative” (deficiency) and wool “positive” (excess), a terminology that persists today.
In the 1780s, French physicist Charles Coulomb quantified electrical charge using a torsional balance, establishing the unit “coulomb.” Two point charges of 1 C, 1 m apart, exert a force of about 9 × 109 N (≈ 2 × 109 lb). One coulomb equals roughly 6.25 × 1018 electrons, making the elementary charge ≈ 1.60 × 10-19 C.
Later research revealed that this “fluid” is in fact electrons—tiny particles named after the Greek word for amber, a material that also shows charge when rubbed.
The Composition of the Atom
All objects consist of atoms, which in turn are made of protons, neutrons, and electrons. Most atoms contain all three particles, but some, like protium (¹H¹), have only a single proton and electron. Though invisible, atoms can be pictured as a dense nucleus surrounded by sparse electrons.
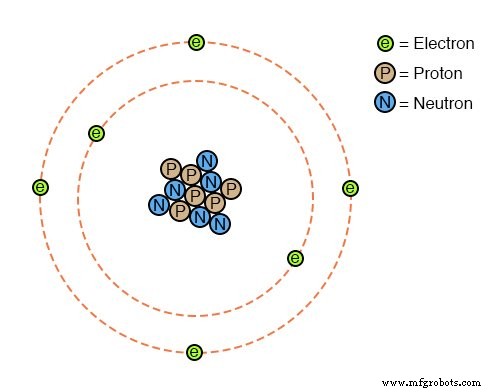
Between the nucleus and the electrons lies a large amount of empty space. The nucleus—comprising tightly bound protons and neutrons—determines an element’s identity: changing the proton count changes the element, as alchemists once hoped to do by turning lead into gold.
Neutrons influence mass and can render an element radioactive, but they do not affect chemical behavior. Electrons, however, can be easily removed or added with relatively little energy. When electrons leave or enter an atom, the balance of positive (protons) and negative (electrons) charges shifts, giving the atom an overall charge.
In an atom, protons and electrons attract each other over distance, while identical charges repel. The strong nuclear force binds protons and neutrons together within the nucleus at extremely short ranges. Because electrons carry a negative charge and protons a positive one, an equal number of each results in a neutral atom. Extra or missing electrons create a net charge, making the atom interact with other charged particles.
When two materials are rubbed together, electrons are transferred from one to the other—exactly the “fluid” Benjamin Franklin described.
What Is Static Electricity?
Static electricity arises when electrons are unevenly distributed between objects. The term “static” reflects that the displaced electrons remain largely stationary on insulating surfaces. For example, experiments showed that electrons move from wool to wax, contrary to Franklin’s original assumption. The resulting charge designations—negative for excess electrons, positive for a deficiency—are still in use.
In 1832, Michael Faraday demonstrated that static electricity is essentially the same as electricity produced by batteries or generators. Although often considered a nuisance, static electricity can damage semiconductor circuits and ignite flammable powders. Nonetheless, it has practical uses in xerography, electrostatic air filtration, and Van de Graaff generators.
REVIEW:
- All materials are composed of atoms.
- Atoms contain protons, neutrons, and electrons, except for the hydrogen-1 isotope.
- Electrons carry a negative charge; protons, a positive charge; neutrons are neutral.
- Electrons can be removed or added more easily than protons or neutrons.
- The proton count defines an element’s identity.
RELATED WORKSHEETS:
- Static Electricity Worksheet
- Atomic Structure Worksheet
Industrial Technology
- Sensitive Static‑Electricity Detector Using a JFET Switch
- Amber: From Ancient Resin to Scientific Treasure – History, Properties, and Modern Uses
- The Guillotine: History, Design, and Legacy
- Silicon: Properties, Production, and Market Outlook
- The Ukulele: From Portuguese Roots to Modern Craftsmanship
- The Complete Guide to the U.S. Chicken Industry: History, Production, and Quality Control
- Batteries: From Volta’s Pile to Modern Lithium Cells
- Suzie Model One: Precision CNC PCB Cutter
- Protect Your Business: Safeguard Against Ransomware Attacks
- Transforming Lives with Custom Running Chairs



