Practical Battery Bank Design: Series vs Parallel, Protection, and Charging Best Practices
When building larger battery banks, selecting compatible cells is essential to avoid capacity loss and safety hazards.
Batteries in Series
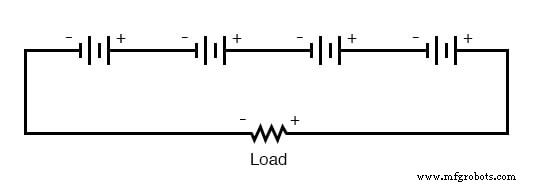
Connecting cells in series raises the overall voltage while keeping the amp‑hour capacity unchanged. Because the same current flows through every cell, all cells must share the same amp‑hour rating; otherwise the weaker cells will deplete first, compromising the entire bank’s performance.
Batteries in Parallel
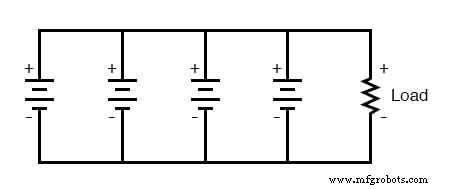
Parallel connections increase the available current (lowering internal resistance) and add amp‑hour capacity. In a parallel circuit the voltage across each branch is identical, so all cells must have the same nominal voltage. Mismatched voltages cause large circulating currents that can damage cells and wiring.
Overcurrent Protection
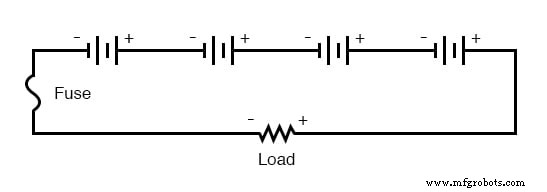
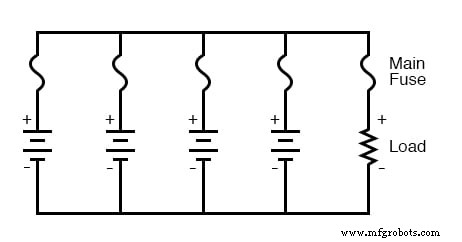
Proper fuse or breaker placement protects both wiring and individual cells. For a series bank, a single fuse on the main supply line stops all current if a cell fails. For a parallel bank, a load fuse protects the external circuit, while individual fuses on each cell guard against internal short‑circuits that could force dangerous currents through neighboring cells.
Charging Considerations
Secondary cells require careful charging. Manufacturers’ instructions are the most reliable guide. Two key concerns are:
- Cycling – the process of charging a cell to full capacity and then discharging it to a lower state. Each battery has a finite cycle life, and the allowable depth of discharge varies by design.
- Overcharging – allowing current to continue once the cell is fully charged. For lead‑acid cells, overcharging produces electrolysis, boiling the electrolyte and drastically shortening lifespan.
Hydrogen Gas Safety
Water‑based electrolytes generate hydrogen during overcharge or normal operation. Hydrogen is odorless, colorless, and highly flammable, especially when mixed with the oxygen produced by electrolysis. Even a small spark—such as from disconnecting a charger—can ignite the gas, as witnessed in a documented lead‑acid battery explosion in a high school automotive shop. Protective measures include:
- Always make the last connection or first disconnection at a point away from the battery, at least one foot from the case, to avoid spark‑ignition of hydrogen.
- Install vent caps on each cell in permanent installations and route venting ducts to exhaust the gas safely outside the battery enclosure.
- For sealed lead‑acid designs, ensure adequate ventilation as a precaution against leaks.
Key Takeaways
- Series wiring increases voltage but not amp‑hour capacity; all cells must share the same amp‑hour rating.
- Parallel wiring increases both voltage and amp‑hour capacity; all cells must share the same voltage rating.
- Proper overcurrent protection requires a main fuse for series banks and both a load fuse and individual fuses for parallel banks.
- Follow manufacturer charging guidelines to prevent overcharging and excessive cycling.
- Never allow hydrogen gas to accumulate; use venting and safe connection practices.
Additional Resources
Industrial Technology
- Optimizing Battery Performance: Parallel & Series‑Parallel Configurations for High‑Load Lamps
- Key ADC Design Factors: Resolution, Sampling, and Practical Performance
- Determinism and Fault Tolerance: Essential Design Principles for Industrial Control Networks
- Practical Considerations for Operational Amplifiers
- Practical Considerations for Selecting and Using Capacitors
- Practical Considerations for Selecting and Using Inductors
- Practical Considerations in Transformer Design: Power, Losses, and Performance
- Key Testing Parameters for Electric Vehicle Batteries
- Graphene Batteries Explained: The Future of Long-Lasting Energy
- Essential Guide to Battery Types for Electronics Projects



