Propane: Production, Quality Standards, and Environmental Benefits
Background
Propane (C3H8) is a naturally occurring hydrocarbon formed during the long‑term decomposition of organic matter. It is extracted from petroleum reservoirs and refined for commercial use. As a liquefied petroleum gas (LPG), propane condenses into a dense liquid at modest pressures, making it highly transportable and storage‑efficient. The United States consumes roughly 15 billion gallons (57 billion liters) of propane each year, with the chemical and manufacturing sectors leading the demand. Residential homes and commercial facilities also rely on propane for heating, cooking, and power generation.
Petroleum’s utility has been recognized for millennia—ancient Mesopotamians used tar‑like products derived from crude oil for masonry and jewelry adhesives, while Arabian scholars in the 1st century CE first distinguished petroleum fractions by boiling point. Modern refining began in 1859 with the discovery of oil in Pennsylvania, and propane’s commercial significance emerged in 1910 when chemist Dr. Walter Snelling identified the gas’s evaporative losses from gasoline.
Today, the U.S. propane industry is valued at approximately $8 billion, underscoring its importance in the energy landscape.
Raw Materials
Propane is not synthesized from other materials; it is extracted from the complex mixtures found in subsurface petroleum reservoirs. These reservoirs consist of porous sandstone and carbonate rocks that trap hydrocarbons formed from ancient organisms preserved in oxygen‑free sedimentary layers. Over millions of years, diagenesis (below 122 °F / 50 °C) and catagenesis (122–424 °F / 50–200 °C) convert the organic matter into a spectrum of hydrocarbons, including propane.
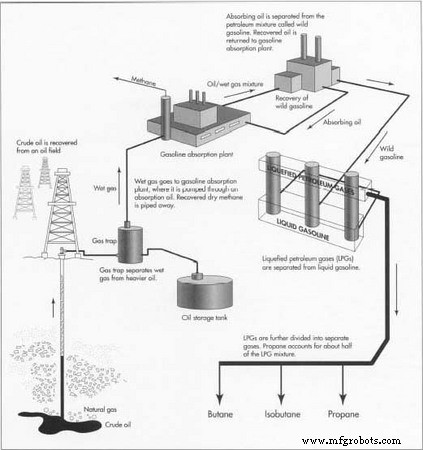
The Manufacturing Process
Propane production involves two primary extraction routes: separation from natural gas streams and distillation from crude oil. The general steps are:
- Oil wells are drilled to bring petroleum and associated gases to the surface. The mixture is routed to a gas trap where crude oil and “wet” gas separate.
- Crude oil, the denser phase, is stored for later refinery processing. Although a small fraction of propane is produced from crude oil, it serves as a refinery fuel or feedstock for LPG and ethylene.
- The wet gas, rich in natural gasoline and LPGs, is directed to a gasoline absorption plant. Cooling and absorption remove natural gasoline, leaving a dry gas stream (≈90 % methane) for distribution.
- Hydrocarbons released from the absorbing oil are condensed into “wild gasoline.” The clean oil is recycled back to the absorber.
- Wild gasoline is sent to stabilizer towers, where liquid gasoline is extracted and LPGs are drawn from the top.
- From the LPG stream—about 10 % of the total gas mixture—propane (≈5 %) is isolated along with butane and isobutane.
Quality Control
Propane must be meticulously purified to meet strict specifications, as impurities can hinder liquefaction and compromise safety. The American Society for Testing and Materials (ASTM) defines standards for commercial propane, including a maximum vapor pressure of 200 psig at 100 °F (38 °C) and a residual matter limit of 0.0017 oz (0.05 ml). These criteria ensure low corrosivity, minimal sulfur, and negligible moisture.
Strict quality control also enhances propane’s environmental performance. EPA studies indicate that propane engines emit up to 45 % less ozone‑forming potential and 29 % fewer total hydrocarbons than gasoline engines. Carbon monoxide, hydrocarbon, and nitrogen‑oxide emissions are reduced by 93 %, 73 %, and 57 % respectively, meeting or exceeding current federal clean‑air standards.
Byproducts and Co‑Products
During propane extraction, a suite of co‑products—solids, liquids, and gases—are generated. Solids such as bitumen and hydrogen sulfide find use as fuels, while liquid fractions include light and heavy crudes that feed asphalt production and petrochemical synthesis. Valuable intermediates like propylene and butylene support synthetic rubber and gasoline refining.
The Future
Ongoing research focuses on enhancing propane recovery, particularly from remote wells where natural gas is currently flared. Converting excess gas into condensable LPGs could reduce waste and improve supply efficiency. Moreover, propane’s low emissions, coupled with economic viability, position it as a preferred alternative fuel under the Clean Air Act’s 1990 designation of LPGs as clean‑burning options.
Manufacturing process
- What is VMC Machining? An Expert Overview of Vertical Machining Centers
- Expert Guide to Aluminum Laser Marking: Precision, Durability, and Industry Applications
- MIG vs. TIG Welding: Selecting the Right Arc Welding Technique for Your Project
- Propane: Production, Quality Standards, and Environmental Benefits
- Understanding Asphalt Cement: Production, Applications, and Future Innovations
- Smart IoT Propane Tank Level Monitoring – Reliable Wireless Solution
- Elevate Fuel Monitoring with IoT: Transforming Petroleum Operations
- IoT Fuel Level Monitoring Boosts Operational Efficiency in the Petroleum Sector
- Essential Guide to Inspecting Propane Forklifts for Safety and Efficiency
- A Comprehensive Guide to Propane Fittings: Types, Uses, and Safety



