Baking Soda: Chemistry, Uses, and Production
Background
Baking soda, chemically known as sodium bicarbonate (NaHCO3), is a white crystalline powder that chemists classify as an acid salt. It forms from a combination of the acid carbonic and the base sodium hydroxide, giving it mild alkaline properties. When heated above 149 °C (300 °F), it decomposes into sodium carbonate, water, and carbon dioxide.
The inherent chemical and physical characteristics of sodium bicarbonate underpin its versatility. It neutralizes odors through a true chemical reaction rather than merely masking them, which makes it ideal for bath salts, deodorant powders, and anti‑plaque mouthwashes. In aqueous solutions it maintains a pH of 8.1, even when acids or bases are added. Its tabletizing ability makes it an excellent effervescent component in antacids and denture cleaners.
In household cleaning, the crystalline structure provides gentle abrasion that lifts dirt without scratching delicate surfaces, while its mild alkalinity transforms fatty acids into a soap‑like solution that rinses cleanly. As a leavening agent, it releases carbon dioxide when paired with an acid—such as lemon juice—causing baked goods to rise.
Beyond domestic use, baking soda’s CO₂ release and neutralizing power give it key industrial roles. It smothers flames in fire extinguishers, absorbs sulfur dioxide in air‑pollution control, and is employed in abrasive blasting, chemical synthesis, leather tanning, oil‑well drilling fluids, rubber and plastic manufacturing, paper production, textile processing, and water treatment where it helps reduce lead and other heavy metals.
Imported from England, baking soda first appeared in America during the colonial era. The United States began domestic production in 1839. In 1846, Dr. Austin Church of Connecticut and farmer John Dwight of Massachusetts founded a factory in New York that produced baking soda under the name Vulcan Spice Mills. The Roman‑god logo of an arm and hammer became the recognizable brand that today is known as Arm & Hammer.
The original manufacturing route was the Leblanc process, developed by French chemist Nicolas Leblanc. It produced soda ash (Na2CO3) from sodium chloride, sulfuric acid, coal, and limestone. In the late 1800s, Ernest Solvay introduced a cleaner, more efficient method that uses sodium chloride, ammonia, and carbon dioxide to precipitate crude sodium bicarbonate, which is then converted to soda ash and refined to United States Pharmacopoeia (USP) purity. Environmental concerns over the Solvay process have spurred a shift toward refining soda ash from trona ore—a naturally occurring, ancient mineral deposit.
Raw Materials
Sodium bicarbonate is sourced from soda ash produced either by the Solvay process or by refining trona ore. Trona, a hard crystalline mineral that formed 50 million years ago in the Green River Basin of Wyoming, has yielded a 200‑billion‑ton deposit that can satisfy global demand for thousands of years. Companies such as Church & Dwight Co. Inc. and FMC Corporation increasingly rely on trona mining to mitigate the pollution associated with the Solvay method. FMC’s mine shafts, extending 4,022.5 km (2,500 mi) and covering 24 sq mi, feature 15‑ft‑wide, 9‑ft‑tall tunnels that accommodate heavy equipment and vehicles.
Manufacturing Process
Making soda ash
- 1. Soda ash can be manufactured chemically via the Solvay process or extracted from trona ore. When trona is used, it is first mined, then transported to processing plants where it is refined into a slurry of sodium sesquicarbonate—a mixture of sodium carbonate and sodium bicarbonate.
Making baking soda
- 2. The intermediate solution is fed into a centrifuge to separate crystals from liquid. The crystals are dissolved in a bicarbonate solution in a rotary dissolver to create a saturated mixture. After filtration, the solution is pumped to a carbonating tower.
- 3. Purified carbon dioxide is introduced at the tower’s base under pressure. As the saturated solution descends, it cools and reacts with CO₂ to crystallize sodium bicarbonate. The crystals are collected, centrifuged again to remove excess solution, washed, and dried on a belt conveyor or flash dryer. Church & Dwight reports a 90‑95 % theoretical yield, producing >99 % pure baking soda.
- 4. The dried cake is sorted by particle size, packaged, and shipped. An illustration of the process appears below.
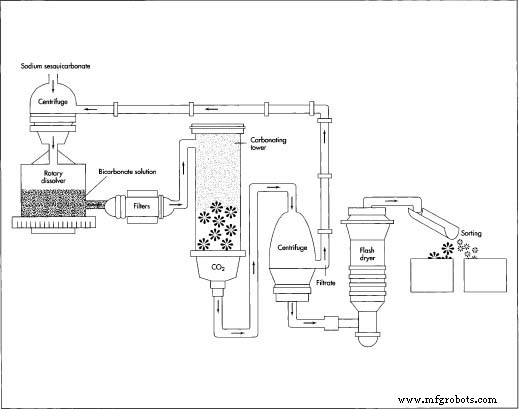
In the carbonating tower, the descending saturated solution cools and reacts with CO₂ to form sodium bicarbonate crystals—baking soda. After filtering, washing, and drying, the crystals are graded by particle size and packaged for specific applications.
Sorting and Storing
- 5. After drying, sodium bicarbonate crystals are sorted into grades based on particle size. Powdered #1 and fine granular #2 are widely used in food, chemical, and pharmaceutical products. Granular grades #4 and #5 appear in dough, cleaning agents, and more. Industrial grade is reserved for oil‑well drilling fluids, fire extinguishers, and water treatment.
- 6. Each grade is stored in a controlled environment that manages atmosphere, CO₂, and moisture to “cure” the product before packaging and shipment.
Quality Control
Quality assurance spans every step of production. FMC, for instance, selects materials and equipment that meet the strictest standards for pharmaceutical‑grade sodium bicarbonate. Statistical Process Control (SPC) is employed to maintain consistent daily quality, and key operating parameters are charted for continuous oversight. Products are catalogued by lot number, and samples are retained for two to three years.
All USP grades satisfy the United States Pharmacopoeia and Food Chemicals Codex specifications for pharmaceutical and food applications. Food‑grade sodium bicarbonate also meets the U.S. Food and Drug Administration’s Generally Recognized as Safe (GRAS) designation.
The Future
In the early twentieth century, 53,000 tons (48,071 MT) of baking soda were sold annually. By 1990, sales had declined to approximately 32,000 tons (29,024 MT) per year, largely due to the rise of self‑rising flour and pre‑mixed cake and biscuit blends that reduced the need for baking soda as a leavening agent. Nevertheless, demand remains strong among commercial bakers—especially cookie manufacturers—and across pharmaceutical and industrial sectors. Sodium bicarbonate’s ability to release CO₂ upon heating ensures its continued relevance for baking, medicine, and a broad array of applications.
Manufacturing process
- What is VMC Machining? An Expert Overview of Vertical Machining Centers
- Expert Guide to Aluminum Laser Marking: Precision, Durability, and Industry Applications
- MIG vs. TIG Welding: Selecting the Right Arc Welding Technique for Your Project
- Comprehensive Guide to Laser Marking: Types, Benefits, and Applications
- Key Considerations for High‑Volume Swiss Machining
- Baking Powder Explained: History, Chemistry, and Production
- Baking Soda: Chemistry, Uses, and Production
- The History, Production, and Recycling of PET Soda Bottles
- Revolutionizing Baking: How Robotic Automation Optimizes Production
- Soda Ash Explained: Uses, Benefits, and Safety Tips



