How Catalytic Converters Work: Reducing Emissions & Protecting Your Vehicle
Modern cars emit a mix of gases—often called “exhaust” or “emissions”—that can harm air quality. In response, the U.S. Clean Air Act of 1963 and its 1965 amendment, the National Emissions Standards Act, established federal vehicle emission limits. States now enforce these standards, requiring routine emissions testing for all registered vehicles.
At the heart of a vehicle’s emissions control system is the catalytic converter, a component that helped manufacturers meet federal standards and significantly cut pollution.
Who Invented the Catalytic Converter?
The idea traces back to the late 19th century, with early prototypes developed in France. In the mid‑1950s, French engineer Eugene Houdry patented a design that could convert harmful exhaust gases into less dangerous substances. Houdry’s work was driven by concerns over industrial smokestack pollution and the rising environmental crisis in Los Angeles.
After the 1960s emissions regulations, the first production catalytic converter appeared in 1973 at Engelhard Corporation. Widespread adoption followed in 1975, and the technology has evolved ever since.
What Does a Catalytic Converter Do?
A catalytic converter contains a chamber filled with a catalyst—typically platinum, rhodium, or palladium—that transforms toxic emissions into harmless gases such as water vapor, carbon dioxide, and nitrogen. The device sits beneath the vehicle, flanked by two pipes: an “input” pipe that carries raw exhaust from the engine, and an “output” pipe that releases the treated gases into the tailpipe.
When exhaust gases flow over the catalyst, a chemical reaction breaks down pollutants. The cleaner gases then travel through the output pipe and exit the vehicle.
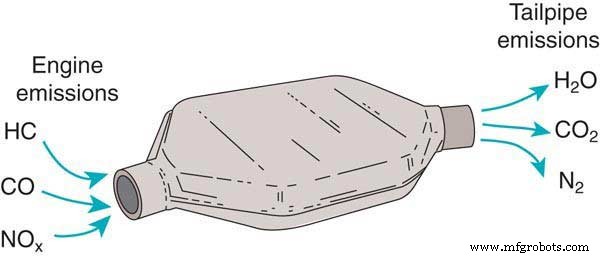
What Is Inside a Catalytic Converter?
The core of the converter is a ceramic honeycomb structure coated with precious metals. These metals perform two key reactions:
- Reduction catalysts remove oxygen from nitrogen oxides (NOx), converting them into harmless nitrogen and oxygen.
- Oxidation catalysts add oxygen to carbon monoxide (CO) and unburned hydrocarbons, turning them into carbon dioxide and water.
An adjacent oxygen sensor monitors exhaust oxygen levels and feeds data to the engine control unit (ECU). This ensures the engine delivers the correct air‑fuel mix for optimal catalytic conversion.
Types of Catalytic Converters
Two primary designs have been used in the U.S.:
- Two‑way converters (pre‑1981) contain only oxidation catalysts, converting CO and hydrocarbons into CO₂ and H₂O.
- Three‑way converters (introduced in 1981) combine oxidation with reduction catalysts, effectively handling CO, hydrocarbons, and NOx.
Diesel engines employ specialized two‑way converters that target particulate matter—soluble organic fractions bound to soot.
How to Prevent Catalytic Converter Theft
Because catalytic converters contain valuable precious metals, thieves target them, especially on vehicles with higher ground clearance. To protect your vehicle:
- Park in well‑lit, secured areas or garages.
- Weld the converter to the chassis.
- Install a metal cage or protective cover.
- Use a car alarm with vibration detection.
- Engrave the VIN on the converter for traceability.
Frequently Asked Questions
Why do people steal catalytic converters?
Rising prices for platinum, palladium, and rhodium have made converters lucrative targets for scrap dealers.
How much precious metal is in a converter?
A typical converter contains 3‑7 g of platinum, 2‑7 g of palladium, and 1‑2 g of rhodium.

How much is a catalytic converter worth?
Recyclers pay $50‑$250 for a standard converter, but values can rise to $800‑$1,500 for hybrids. A replacement can cost around $2,000.
Signs Your Catalytic Converter Is Failing
When the converter becomes clogged or damaged, you may notice:
- Reduced fuel economy—the engine burns more fuel to compensate for restricted exhaust flow.
- Check Engine Light—especially on vehicles built after 1996, diagnostic codes can pinpoint converter issues.
- “Rotten egg” odor—unconverted sulfur compounds emit a sulfuric smell.
- Hard starting or stalling—excess exhaust pressure hampers engine performance.
- Poor acceleration—trapped exhaust causes jerky or stalled acceleration.
- Failed emissions test—a common symptom of a malfunctioning converter.
Learn About Catalytic Converters and Exhaust Systems at UTI
Curious about automotive technology? The Universal Technical Institute’s Automotive Technology program trains you in 51 weeks to become a certified technician, covering everything from exhaust systems to advanced diagnostics.
Contact UTI at 1‑800‑834‑7308 or request more information online to start your career today!
Industrial equipment
- What Is a Toggle Press? How It Works and Why It Matters
- Compression Molding: Process, Benefits, and Applications for Rubber Parts
- Demagnetizers Explained: How They Add or Remove Magnetism from Tools
- Abrasive Jet Machining: How It Works, Benefits, and Practical Applications
- Understanding Gate Hinges: Types, Functions, and How to Choose the Right One
- Transfer Molding Explained: Process, Benefits, and Variants
- What Is a Solar Inverter? How It Converts Solar Power into Usable Electricity
- Transmission Fluid Explained: Purpose, Importance, and Maintenance Guide
- Understanding Vehicle Transmissions: Types, Functions, and How They Operate
- Understanding Industrial Clutches: Definition, Function, and Applications



