Alumina Nanorods Synthesized from Chromium‑Containing Alumina Sludge: Influence of Cr, Fe, and Mg Doping on Crystal Transformation
Abstract
Alumina nanorods were produced from chromium‑containing alumina sludge, and the role of dopants—Cr, Fe, and Mg—was systematically investigated. The doped Cr constrained the transition of alumina from the metastable θ-Al2O3 phase to the stable α-Al2O3 during calcination, whereas Fe and Mg facilitated this transformation. Co‑doping from the sludge further restricted the phase change, producing predominantly θ-Al2O3 with reduced crystallinity. Cr mainly entered the lattice as Cr(III), while Fe and Mg were incorporated at lower levels. These findings provide a technical foundation for converting hazardous chromium sludge into valuable alumina nanorods, thereby addressing environmental concerns and creating economic value.
Background
Low‑dimensional alumina—such as nanofibers and nanorods—offers exceptional strength, high elastic modulus, chemical stability, low thermal conductivity, and superior insulation, making it attractive for composites, catalysts, adsorbents, membranes, coatings, and battery anodes. However, the high cost of production limits widespread adoption. Numerous synthesis routes exist, including solid‑phase, vapor‑phase, and liquid‑phase methods. Among them, the liquid‑phase precipitation route is preferred for its mild conditions, homogeneous products, and low cost.
Chromium‑containing alumina sludge is a hazardous waste generated during non‑calcium roasting of chrome ore. Each ton of chrome product produces roughly 7 t of sludge, composed of 55–65 % Al2O3, 7–13 % chromium (as Cr(III) or the carcinogenic Cr(VI)), and trace silicon, iron, magnesium, and sodium. Conventional treatment involves detoxification—converting Cr(VI) to Cr(III)—or direct utilization of the sludge in refractory materials, both of which either occupy land or generate secondary pollution. Converting the sludge into high‑value alumina nanorods offers a dual benefit: pollution abatement and resource recovery.
In this study, alumina nanorods were prepared from the sludge via precipitation–calcination. Undoped, Cr‑doped, Fe‑doped, and Mg‑doped samples were also synthesized to isolate the effect of each ion. The resulting materials were characterized by XRD, FT‑IR, TG‑DSC, TEM, EDS, and XPS to elucidate phase evolution, microstructure, and chemical state changes.
Methods
Materials
Analytical‑grade reagents (aluminum sulfate octadecahydrate, chromium sulfate, ferric sulfate, magnesium sulfate, sodium hydroxide, sulfuric acid, sodium dodecyl benzene sulfonate) were used. Chromium‑containing alumina sludge was supplied by CITIC Jinzhou Metal Co., Ltd. All solutions were prepared with de‑ionized water.
Treatment of the Chromium‑Containing Alumina Sludge
The sludge was first washed and filtered (solid–liquid ratio 1:5 g/mL) to remove soluble Cr(VI). The residue was dissolved in sulfuric acid (ratio 1:3 g/mL), then oxidized with H2O2 to convert any remaining Cr(VI) to Cr(III). The resulting acidic solution was analyzed by titration and VIS spectroscopy (Varian 721N) and its composition is shown in Table 2.
Synthesis of Alumina Nanorods
A 0.25 M Al2(SO4)3 solution was mixed with 2 M NaOH and dodecyl benzene sulfonate under stirring at 85 °C. The pH was adjusted to 9.0, the mixture was stirred for 5 h, then aged for 20 h. The precipitates were collected, washed with de‑ionized water and ethanol, and vacuum‑dried at 40 °C for 15 h. Calcination was performed in a programmable furnace: 250 °C / 1 h, 400 °C / 1 h, 770 °C / 1 h, 900 °C / 1 h, 1050 °C / 2 h. Undoped samples used pure Al2(SO4)3; doped samples incorporated Cr, Fe, or Mg chlorates at concentrations matching those in the sludge (Table 2). Samples derived directly from the sludge were labeled “sludge‑derived.”
Characterization
Phase identification: XRD (Rigaku D/MAX‑RB, Cu Kα, 2θ = 10–70°, 2°/min). Functional groups: FT‑IR (Thermo Electron Scimitar 2000, 4000–400 cm−1). Thermal stability: TG‑DSC (NETZSCH STA449F3, 15–1200 °C, 10 °C/min, air). Morphology and lattice: FETEM (JEOL Jem‑2100F). Surface chemistry: XPS (ESCAMABMKLL, Al Kα). Elemental distribution: EDS (JEOL).
Results and Discussion
XRD Characterization of the Alumina Nanorods
The XRD patterns (Fig. 1) reveal the presence of both α- and θ-Al2O3 phases. In undoped rods, θ-Al2O3 peaks are weak, indicating partial transformation to α-Al2O3 during calcination. Cr‑doped rods exhibit stronger θ-Al2O3 peaks and weaker α-Al2O3, confirming that Cr retards the phase transition. Conversely, Fe‑ and Mg‑doped rods show pronounced, sharper α-Al2O3 peaks, reflecting accelerated crystallization. Sludge‑derived rods display dominant θ-Al2O3 with low crystallinity, due to the high impurity load that impedes phase conversion.
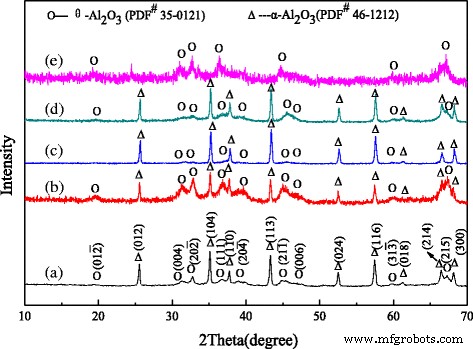
XRD patterns of alumina nanorods doped with different ions: a undoped alumina, b Cr-doped alumina, c Fe-doped alumina, d Mg-doped alumina, and e sludge-derived alumina.
FT‑IR Spectra of the Alumina Nanorods
All samples exhibit O–H stretching (3500–3300 cm−1) and H2O bending (1635 cm−1), indicating adsorbed water. The 2360 cm−1 band corresponds to CO2. In the fingerprint region, undoped rods show characteristic α-Al2O3 vibrations at 829, 589, and 449 cm−1 and θ-Al2O3 at 762, 663, and 488 cm−1. Cr doping reduces the α-Al2O3 signals, while Fe and Mg doping weaken the θ-Al2O3 bands and shift peaks slightly, reflecting changes in bonding. Sludge-derived rods lack α-Al2O3 features, confirming the absence of the stable phase.
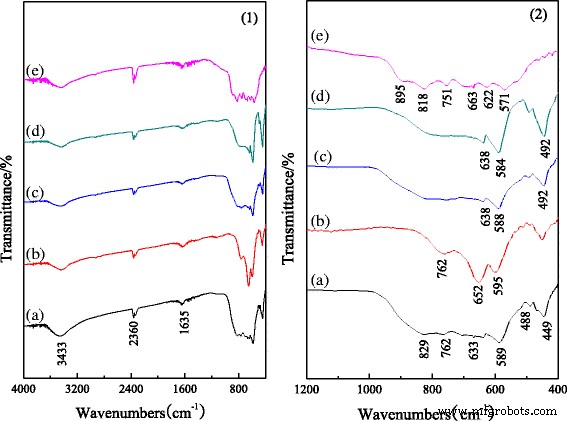
FT‑IR spectra of nano alumina rods doped with different ions: a undoped alumina, b Cr-doped alumina, c Fe-doped alumina, d Mg-doped alumina, and e sludge-derived alumina.
TG‑DSC of Alumina Nanorods
The TG–DSC curves (Fig. 3) show three mass‑loss stages for undoped precursors: 40 % loss below 250 °C (moisture), 35 % between 250–730 °C (dehydroxylation and transformation to θ-Al2O3), and a minor loss above 730 °C (conversion to α-Al2O3). Doping shifts the endothermic peaks to higher temperatures and broadens them, indicating that Cr, Fe, and Mg alter the kinetic pathways of phase transformation. These thermal trends align with XRD and FT‑IR observations.
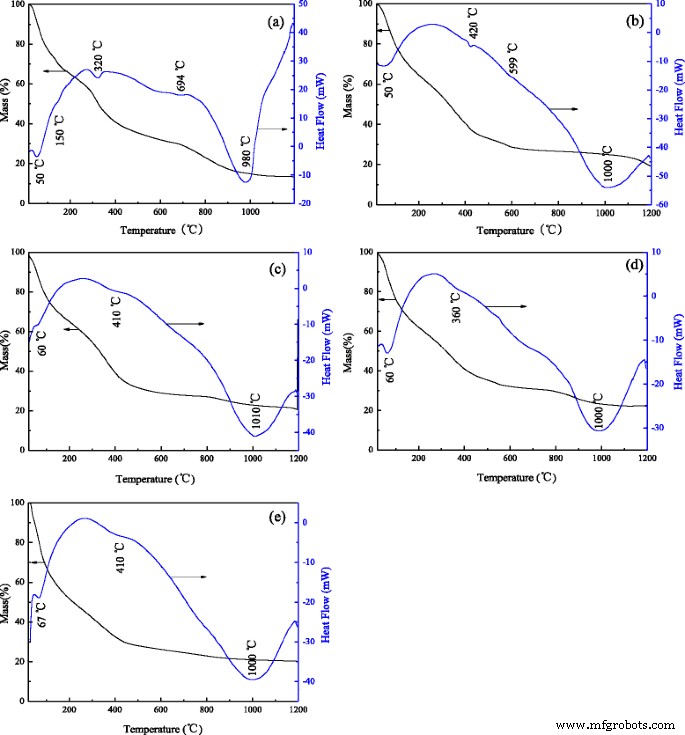
TG and DSC of the nano alumina rod precursors doped with different ions: a undoped alumina, b Cr-doped alumina, c Fe-doped alumina, d Mg-doped alumina, and e sludge-derived alumina.
TEM, SAED, and HRTEM Images of Alumina Nanorods
Undoped rods are uniformly dispersed with diameters of 4–6 nm and lengths of 20–60 nm. SAED patterns confirm mixed θ- and α-Al2O3 phases, and HRTEM shows interplanar spacings of 0.273 and 0.284 nm (θ) and 0.255 and 0.348 nm (α). Cr-doped rods are longer (50–120 nm) but maintain similar lattice spacings, indicating that Cr does not drastically alter the crystal lattice. Fe- and Mg-doped rods exhibit improved crystallinity with sharper diffraction spots and slightly altered interplanar distances, consistent with their accelerated phase transition. Sludge-derived rods are dispersed, 4–6 nm in diameter and 50–100 nm in length, but lack any α-Al2O3 spots, reflecting the suppression of stable phase formation by co‑dopants.
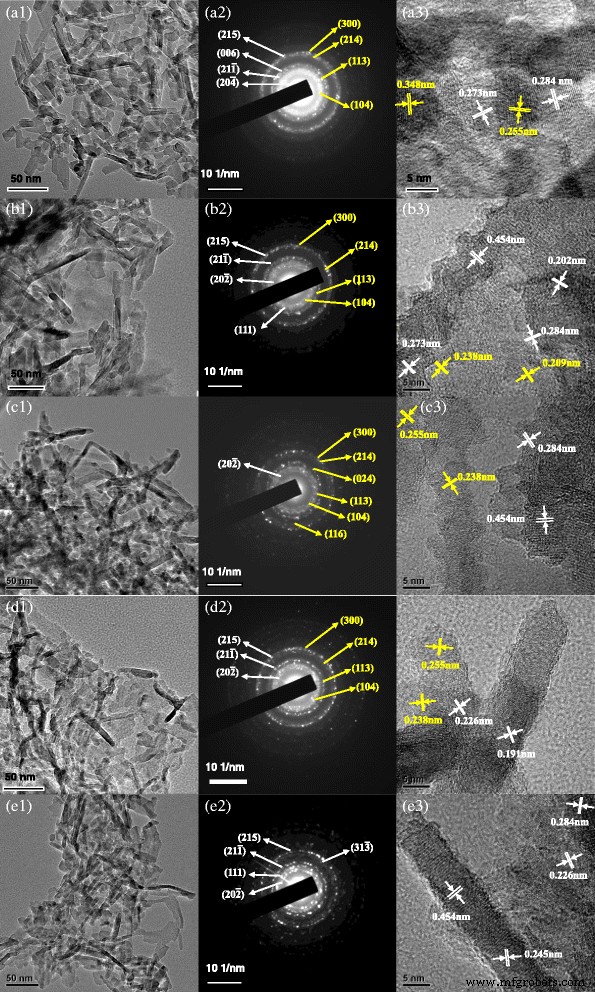
TEM, SAED, and HRTEM of alumina nanorods doped with different ions: a undoped alumina, b Cr-doped alumina, c Fe-doped alumina, d Mg-doped alumina, and e sludge-derived alumina. (1) TEM; (2) SAED; (3) HRTEM.
EDS Characterization of Alumina Nanorods Precursor Doped with Different Ions
EDS quantification (Table 3) shows dopant levels of 2.06 % Cr, 0.99 % Fe, and 0.58 % Mg in single‑doped precursors—close to the target amounts. In sludge‑derived precursors, Cr is 2.11 %, Fe 0.14 %, and Mg 0.96 %, confirming that Cr and Mg dominate the incorporation, while Fe is largely suppressed.
XPS Characterization of Nanometer Alumina Fibers Doped with Different Ions
O 1s spectra reveal binding energies of 531.90 eV (undoped), 531.85 eV (Cr-doped), 531.15 eV (Fe-doped), 531.20 eV (Mg-doped), and 532.00 eV (sludge-derived). These values indicate a slight shift toward higher energy for samples with more θ-Al2O3. Al 2p peaks at 74.00, 74.25, 74.75, 74.38, and 73.90 eV correspond to the respective samples, confirming the Al3+ oxidation state. Fe 2p spectra show both Fe(II) and Fe(III) signals in Fe-doped rods, whereas Cr 2p spectra display Cr(VI) and Cr(III) components, with Cr(VI) largely absent in sludge-derived rods. Mg 2p indicates MgO formation in Mg-doped rods, but is weak in sludge-derived samples, consistent with low Mg content.
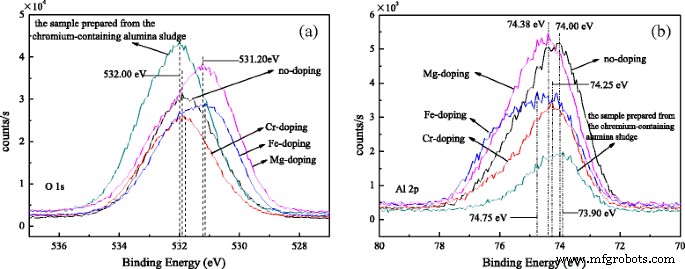
XPS spectra of a O 1s and b Al 2p for alumina nanorods doped with different ions.
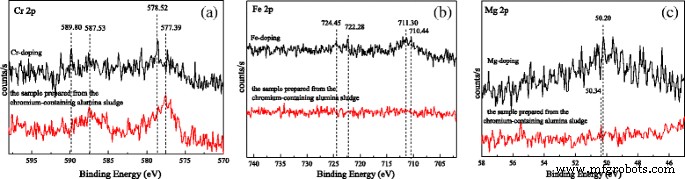
XPS spectra of a Cr 3+ 2p, b Fe 3+ 2p, and c Mg 2+ 2p.
Conclusions
Chromium, iron, and magnesium are successfully incorporated into alumina nanorods. Cr doping impedes the θ→α transformation, while Fe and Mg accelerate it. Co-doping from the sludge severely restricts phase conversion, yielding predominantly θ-Al2O3 with limited crystallinity. The dopants modify the lattice, alter O–Al bonding, and influence thermal behavior, as confirmed by XRD, FT‑IR, TG‑DSC, TEM, EDS, and XPS. These insights demonstrate that chromium-containing alumina sludge can be valorized into high‑performance alumina nanorods, offering a sustainable solution to waste management and resource recovery.
Abbreviations
- BE
Binding energy
- DSC
Differential scanning calorimetry analysis
- EDS
Energy‑dispersive spectrometer
- FETEM
Field‑emission transmission electron microscopy
- FT‑IR
Fourier transform infrared spectra
- HRTEM
High‑resolution transmission electron microscopy
- SAED
Selected area electron diffraction
- TEM
Transmission electron microscopy
- TG
Thermogravimetric analyzer
- XPS
X‑ray photoelectron spectroscopy
- XRD
X‑ray powder diffraction
Nanomaterials
- Professional Guide to Producing High‑Purity Rhenium Powder
- Nanocellulose from Blue‑Green Algae: A Sustainable Pathway to High‑Purity Biomaterials
- Bovine Serum Albumin–Coated Gold Nanorods Enable Low‑Dose NIR‑II Photothermal Therapy
- Small-Angle Scattering Reveals Structure of Deterministic Nanoscale Fat Fractals
- Palladium(II)-Imprinted Polymeric Nanospheres for Efficient Removal of Pd(II) from Water
- Optimizing Zinc Oxide/Porous Anodic Alumina Composite Films for Superior Antibiofilm Performance
- Ultra‑Smooth Copper Substrates Enhance Graphene Quality: Annealing + Electro‑Polishing
- Hollow Polyaniline Microparticles Deliver 127.9 mg/g Cr(VI) Removal for Sustainable Wastewater Treatment
- Hemp‑Stem‑Derived Nanoporous Carbon Anodes for High‑Capacity Lithium‑Ion Batteries
- Japan's Pioneering Journey into Robotics



