Targeted Anti‑Tumor Delivery of Methotrexate via Chondroitin Sulfate‑Based Nanogels
Abstract
Self‑assembled nanogels (NGs) were fabricated by covalently conjugating methotrexate (MTX) to the glycosaminoglycan chondroitin sulfate (CS). The MTX‑CS NGs exhibit dramatically enhanced aqueous solubility and improved biodistribution, thanks to CS’s affinity for the CD44 receptor that is over‑expressed on many tumor cells. In vivo experiments confirm that MTX‑CS NGs deliver higher intratumoral drug concentrations while markedly reducing systemic toxicity compared with free MTX. These results establish CS‑based conjugates as a powerful platform for targeted delivery of sparingly soluble anticancer agents.
Background
Methotrexate (MTX), a folate analogue first introduced in the 1950s, remains a cornerstone of chemotherapy for various malignancies. Its clinical utility, however, is limited by poor water solubility, rapid clearance, and dose‑dependent adverse effects, particularly mucositis and myelosuppression.1‑3 Recent evidence indicates that targeted delivery can overcome these barriers by enhancing tumor uptake while sparing normal tissues.4‑6
Nanotechnology offers a versatile toolkit for improving drug pharmacokinetics. Self‑assembly of drug–polymer conjugates into nanogels provides a protective hydrophilic shell, extended circulation time, and controlled release.7‑13 Chondroitin sulfate (CS), a naturally occurring glycosaminoglycan, binds the CD44 receptor with high affinity, a pathway frequently exploited by metastatic cancers.14‑18 CS‑based nanoparticles have already shown promise in tumor targeting,19‑22. Building on these advances, we engineered a CS‑MTX nanogel aimed at maximizing therapeutic benefit while minimizing toxicity.
Methods
Materials and Samples
Chondroitin sulfate (MW 100–200 kDa) was sourced from Dalian Meilun Biotech Co., Ltd. Other reagents, including 4‑methylmorpholine, tetrahydrofuran (THF), and 2‑chloro‑4,6‑dimethoxy‑1,3,5‑triazine, were obtained from Sun Chemical Technology. Fetal bovine serum (FBS) was purchased from HyClone. Lewis rats (Sprague‑Dawley, 200 ± 10 g) were used for in vivo studies.
Synthesis of DMT‑MM
DMT‑MM was prepared by reacting 2‑chloro‑4,6‑dimethoxy‑1,3,5‑triazine (0.769 g) with 4‑methylmorpholine (18.79 mL) in THF (200 mL) under stirring for 30 min. The resulting white precipitate was filtered, washed thrice with THF, and vacuum‑dry for 24 h.
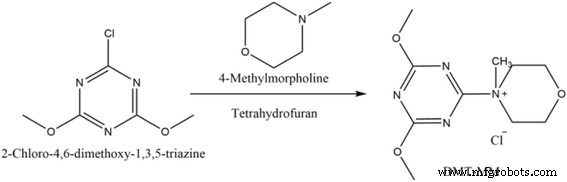
Synthetic pathway for DMT‑MM
Synthesis of MTX‑CS
CS (1.0 g) was dissolved in 20 mL ultrapure water and activated with DMT‑MM (0.769 g). After 30 min at room temperature, MTX (0.5 g) was added and the mixture stirred for 24 h. The conjugate was dialyzed (MWCO 3.5 kDa) against water for 48 h, with frequent exchanges, then lyophilized to a yellow powder. FTIR (400–4000 cm⁻¹) and ¹H‑NMR (400 MHz) confirmed successful conjugation.
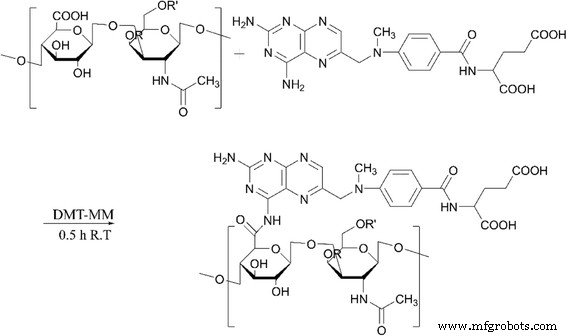
Synthetic pathway for MTX‑CS
Cytotoxicity of MTX‑CS Nanogels
A549T, HeLa, and HUVEC cells were seeded at 5 × 10³ cells/well in 96‑well plates and incubated 24 h (37 °C, 5% CO₂). Cells were then treated with MTX‑CS NGs at concentrations ranging from 0 to 400 µM (based on MTX content) for an additional 48 h. Cell viability was quantified via MTT assay: 20 µL CCK‑8 buffer was added, plates rested 4 h, and absorbance measured at 490 nm (reference 570 nm) on a MULTISKAN GO reader.
Animal and Experimental Design
Male Sprague‑Dawley rats were randomized into three groups (n = 6): (i) saline control, (ii) free MTX (1.25 µmol kg⁻¹ day⁻¹), and (iii) MTX‑CS NGs (equivalent to 1.25 µmol kg⁻¹ day⁻¹ MTX, 25 mg kg⁻¹ day⁻¹ NGs). Treatments were administered intraperitoneally over 14 days (7 injections). Body weight and survival were monitored; animals were sacrificed post‑treatment for organ collection.
Histological Study
Spleens were excised, rinsed with PBS, fixed in 4% paraformaldehyde (pH 7.4) for 24 h, embedded in paraffin, sectioned, and stained with hematoxylin & eosin. Slides were examined under a light microscope for evidence of tissue damage.
Results and Discussion
Characterization of MTX‑CS Nanogels
FTIR spectra (Fig. 1) display characteristic peaks of MTX (3355, 2951, 1646, 1600, 1540, 1493, 1403, 1207 cm⁻¹) that persist in the conjugate, confirming successful attachment. ¹H‑NMR (Fig. 2) reveals signals corresponding to MTX’s benzoyl (δ ≈ 6.9 ppm, 7.8 ppm) and pteridinyl (δ ≈ 8.7 ppm) moieties, while CS signals (δ 3.2–5.4 ppm) remain unchanged, further validating conjugation. The conjugation efficiency, determined by UV‑vis at 309 nm, was 13.65 % MTX per mass of nanogel.
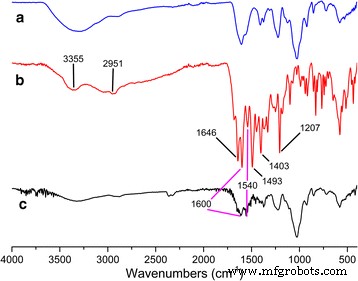
Figure 1 : FTIR spectra of CS (a), MTX (b), and MTX‑CS (c).
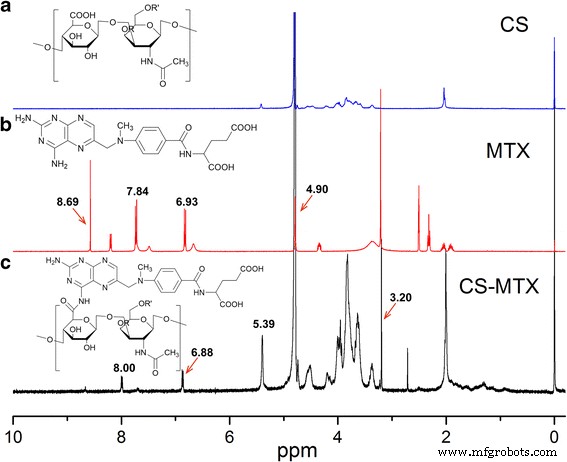
Figure 2 : ¹H‑NMR spectra of CS (a), MTX (b), and CS‑MTX (c).
Dynamic light scattering (DLS) revealed a hydrodynamic diameter of ~200 nm (Fig. 3b), while atomic force microscopy (AFM) and transmission electron microscopy (TEM) confirmed spherical morphology with diameters of 200–240 nm (Fig. 3c‑e). The nanoscale size is optimal for enhanced permeability and retention (EPR) effect and CD44-mediated endocytosis.
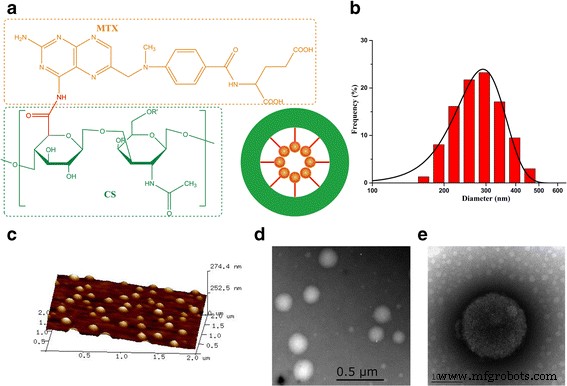
Figure 3 : (a) Schematic of MTX‑CS NGs; (b) DLS size distribution; (c) AFM image; (d‑e) TEM micrographs.
In Vitro Cytotoxicity
MTX‑CS NGs markedly reduced viability of A549T and HeLa cells compared to free MTX and MTX mixed with CS (Fig. 5). For HeLa, cell viability dropped from 73.8 % (free MTX) to 60.2 % (MTX‑CS NGs) at 10 µM, while for A549T the decrease was 80.2 % to 46.0 % at 50 µM. In contrast, the free drug exhibited minimal activity at comparable concentrations, and the MTX+CS mixture even promoted cell growth. These findings underscore the role of CD44‑mediated uptake in enhancing intracellular MTX delivery. HUVEC assays showed a 9.9 % increase in viability at 10 µM MTX‑CS NGs versus 63.6 % for free MTX, indicating reduced off‑target toxicity.
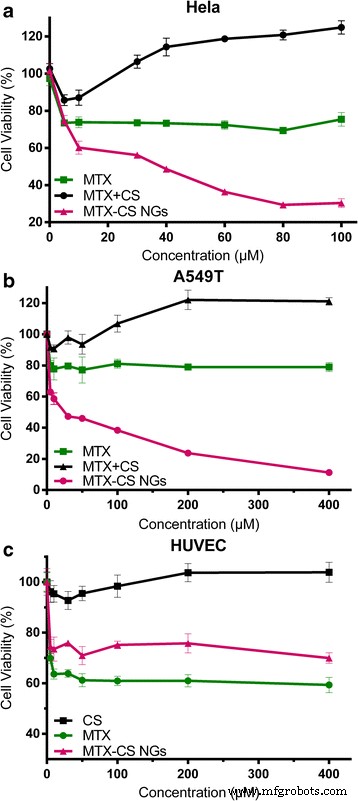
Figure 5 : Cell viability after 48 h treatment with free MTX, MTX+CS, and MTX‑CS NGs in A549T (a), HeLa (b), and HUVEC (c).
In Vivo Toxicity and Efficacy
Body‑weight monitoring over 14 days revealed that rats receiving free MTX lost significant weight, whereas the MTX‑CS NG group maintained near‑baseline weight (Fig. 6). Histological examination of spleens showed severe congestion and hemosiderin deposition in the MTX group, but only mild changes in the MTX‑CS NG group, confirming reduced systemic toxicity (Fig. 7).
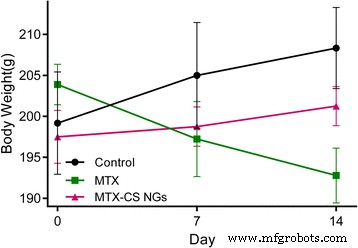
Figure 6 : Body‑weight trajectory for saline, free MTX, and MTX‑CS NG groups.
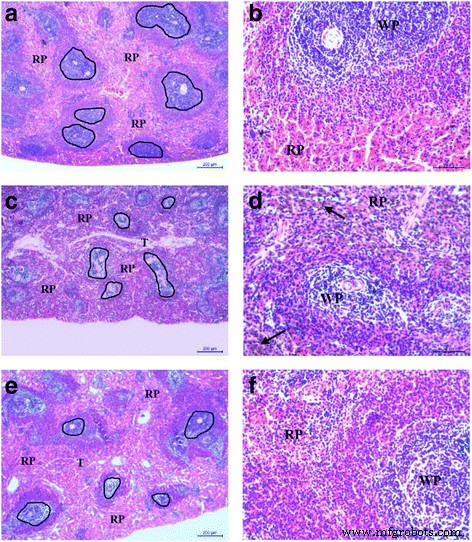
Figure 7 : H&E‑stained spleen sections – (a,b) control, (c,d) free MTX, (e,f) MTX‑CS NG.
Conclusions
We successfully engineered a 200‑nm MTX‑CS nanogel that combines high solubility, CD44‑mediated targeting, and reduced systemic toxicity. In vitro and in vivo data demonstrate superior anti‑tumor potency relative to free MTX, with markedly lower adverse effects. These findings validate CS‑based drug conjugates as a versatile strategy for delivering poorly soluble anticancer agents with improved therapeutic index.
Abbreviations
- 1H NMR:
1H Nuclear magnetic resonance
- AFM:
Atomic force microscope
- CDMT:
2‑Chloro‑4,6‑dimethoxy‑1,3,5‑triazine
- CS:
Chondroitin sulfate
- DLS:
Dynamic light scattering
- DMT‑MM:
4‑(4,6‑Dimethoxy‑1,3,5‑triazin‑2‑yl)-4‑methylmorpholinium chloride
- FTIR:
Fourier transform infrared
- MTT:
3‑(4,5‑Dimethyl‑2‑thiazolyl)-2,5‑diphenyl‑2‑H‑tetrazolium bromide
- MTX:
Methotrexate
- MTX‑CS NGs:
Methotrexate‑chondroitin sulfate nanogels
- NGs:
Nanogels
- NMM:
4‑Methylmorpholine
- TEM:
Transmission electron microscope
- THF:
Tetrahydrofuran
- UV‑vis:
Ultraviolet‑visible spectroscopy
Nanomaterials
- Nicotine Patch: Science, Manufacturing, and Future Innovations
- Nanofiber & Filament-Based Nanocarriers: Advancing Precision Drug Delivery
- Cell‑Based Drug Delivery Systems for Advanced Cancer Therapy
- Nanoparticle-Enhanced Wormlike Micellar System: Design, Rheology, and Mechanistic Insights
- Acidic Micro‑Environments in Liposomes Significantly Enhance Curcumin Stability and Anticancer Efficacy
- First‑Principles Insights into Transition‑Metal Adsorption on Black Phosphorene: Implications for Catalysis and Spintronics
- Lanthanum Strontium Manganite Nanoparticles: Airway Epithelial Cell Toxicity and Mucus Secretion Impact
- Temperature‑ and pH‑Responsive PNIPAM‑Based Nanogels for Dual‑Stimuli Controlled Delivery of β‑Lapachone
- Bibliometric Trends in Drug Delivery and Magnetic Nanoparticles (1980‑2017)
- Exploring Tip-Based Nanomachining with Molecular Dynamics: A Comprehensive Review



