Maghemite Nanoparticles Enhance Growth and Drought Tolerance in Brassica napus by Acting as Nanozymes
Abstract
Yttrium‑doped γ‑Fe₂O₃ nanoparticles were investigated as a dual‑purpose plant additive: a supplemental iron source and a nanozyme that scavenges reactive oxygen species. When delivered through irrigation to Brassica napus grown in soil, the particles lowered leaf hydrogen peroxide from 151 μM g⁻¹ to 83 μM g⁻¹ and reduced malondialdehyde (MDA) levels from 36 mM g⁻¹ to 26 mM g⁻¹ after a 5‑day drought period. Leaf elongation increased by 33 % compared with fully fertilized controls, and chlorophyll content (SPAD) rose from 47 to 52, indicating superior agronomic performance versus chelated iron fertilization.
Background
Global food security hinges on innovations that boost crop yield while reducing environmental footprints. Nanotechnology offers promising tools—from crop protection and precision fertilization to biosensing and packaging—each capable of transforming the production chain. Iron‑oxide nanoparticles (IONs) naturally occur in soil and are produced by plants and microbes. While concerns about engineered ION toxicity exist, recent studies have demonstrated their utility as iron fertilizers and as nanozymes that mimic peroxidase and catalase activities. In particular, γ‑Fe₂O₃ and Fe₃O₄ have shown enzymatic potential at low and neutral pH, respectively, and can enhance plant growth beyond that achieved with equivalent chelated iron doses. We hypothesize that the enzymatic properties of γ‑Fe₂O₃ nanoparticles will not only supply iron but also mitigate oxidative stress during drought, thereby improving plant resilience.
Results
Effect of Particles on Plant Traits
ION application increased leaf length and chlorophyll content relative to chelated‑iron controls. Leaf length rose by 33 % (p = 0.053) and SPAD values improved significantly (p = 0.000), suggesting enhanced cell division or elongation and greater photosynthetic capacity (Fig. 1a–b).
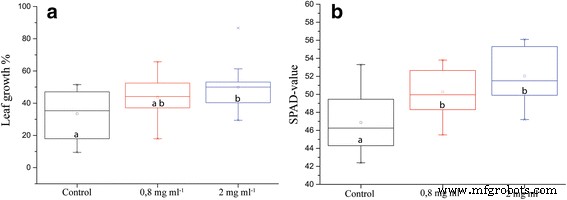
Different physiological parameters of plants grown in pots with soil irrigated with nutrients or nutrients containing IONs. a Individual leaf length increase from before until after 5 days of ION‑treatment (n = 16, p = 0.053). b Chlorophyll content in the leaves, as measured with SPAD measurement (n = 16, p = 0.000). Different letters signify statistically significant difference.
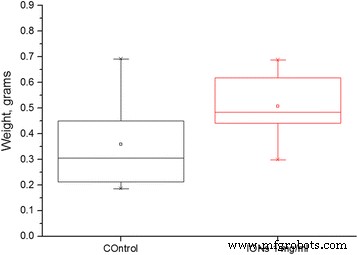
Water retention did not differ significantly, but ION treatments displayed a trend toward greater water content (Fig. 2a). Fresh weight was consistently higher for ION treatments, reaching statistical significance in several experiments (Fig. 2b). In an extended drought trial, plants treated with IONs maintained higher biomass and recovered more rapidly after rewatering (Fig. 3).
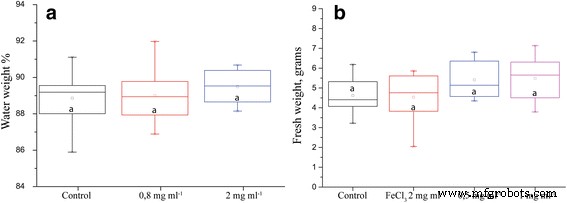
Plant parameters after drought stress. a Percent of plant weight that constitutes water. b Plant biomass after 5 days of drought (n = 8, p = 0.127). Different letters signify statistically significant difference.
Fresh weight of plants, measured after 5 days of drought. Statistically significant difference with 15 biological replicates and p = 0.01.
Control plants, receiving adequate chelated iron, displayed markedly lower drought tolerance. In contrast, ION‑treated plants coped better during stress and recovered more fully post‑rewatering (Fig. 4).

Photos of plants after rewatering following 5 days of drought stress. a Control plants irrigated with nutrient solution. b Plants irrigated with nutrient solution containing 0.8 mg ml⁻¹ IONs. c Plants irrigated with nutrient solution containing 2 mg ml⁻¹ IONs.
Effects of IONs on Leaf Hydrogen Peroxide Concentration
Leaf hydrogen peroxide was markedly lower in ION‑treated plants. While the 0.8 mg ml⁻¹ treatment showed high variability, the 2 mg ml⁻¹ concentration reduced H₂O₂ by 84 % relative to control (p = 0.004) (Fig. 5).
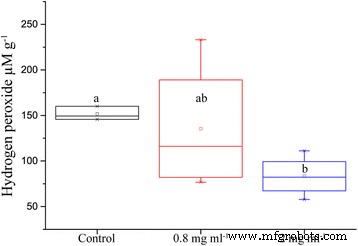
Amount of soluble hydrogen peroxide per gram of leaf tissue from oilseed rape treated with nutrient solution containing IONs and challenged with drought for 5 days (n = 16, p = 0.004).
Effects of IONs on Lipid Peroxidation
Malondialdehyde (MDA) concentrations dropped by 36 % in leaves of plants receiving 200 mg IONs, indicating reduced lipid peroxidation. Although a positive control with equivalent iron(III) ions was included, its high variability precluded definitive conclusions. Nonetheless, a trend toward lower MDA in ION‑treated plants was evident (Fig. 6).
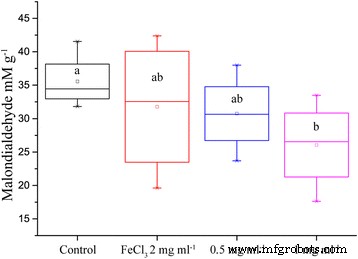
Concentration of the lipid peroxidation product MDA in the leaves of oilseed rape treated with nutrient solution containing IONs and drought for 5 days (n = 8, p = 0.052).
Plant Particle Uptake
Inductively coupled plasma atomic emission spectroscopy (ICP‑AES) revealed a significant rise in leaf iron concentration following ION application, whereas supplemental iron(III) ions actually reduced leaf iron levels (Fig. 7). This pattern supports nanoparticle uptake and a reduced propensity for iron‑induced Fenton reactions.
Iron concentration in Brassica plant leaves after treatment with maghemite nanoparticles, as compared to control with the same nutrient solution or the same nutrient solution with 1:1 M ratio of iron(III) ions. Different letters signify statistically significant difference (n = 15).
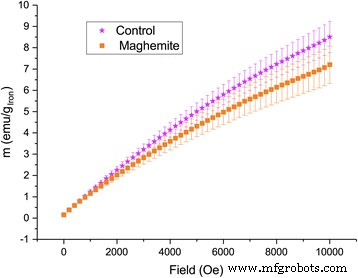
Magnetization measurements further corroborated nanoparticle presence. At 2 K, leaves from ION‑treated plants displayed higher remanent magnetization under zero field, whereas control leaves showed greater magnetization under a 10,000 Oe field, consistent with superparamagnetic behavior of γ‑Fe₂O₃ nanoparticles (Fig. 8–9). Although variability limited statistical significance, the trend aligns with the ICP‑AES data.

Low‑temperature (2 K) magnetization of ashed leaves of plants treated with maghemite nanoparticles compared to control plants. Error bars show standard error of mean (n = 6).
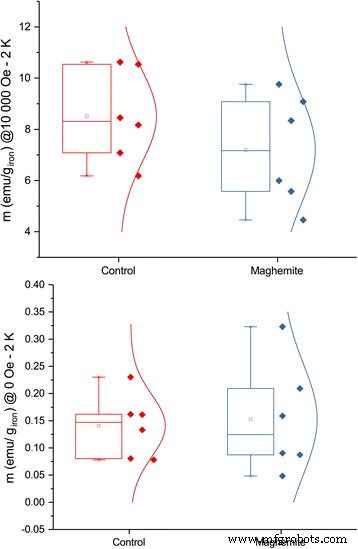
Low‑temperature (2 K) magnetization of ashed leaves under different magnetic fields. The plot at 10,000 Oe has p = 0.8, and the plot at zero field has p = 0.08 (n = 6).
Material Characterizations
The synthesis method produced a gel that, after drying and grinding, yielded a powder of γ‑Fe₂O₃ nanoparticles in the 1–10 nm size range. Scanning electron microscopy (SEM) revealed a hierarchical structure; energy‑dispersive spectroscopy (EDS) detected only iron, confirming the absence of yttrium in the dried powder (Fig. 7).
Dynamic light scattering via nanoparticle tracking analysis (NTA) showed a hydrodynamic diameter distribution up to 500 nm, with 84 % of aggregates below 300 nm and 11 % below 50 nm. In a 50‑fold diluted dispersion, 4.28 × 10⁶ particles per mL were smaller than 20 nm, implying approximately 2 × 10⁸ sub‑20 nm particles per mL in the irrigation solution.
Atomic force microscopy (AFM) images confirmed the size distribution observed by NTA and X‑ray diffraction (XRD). XRD, performed one year after synthesis, confirmed the maghemite crystal structure with a crystallite size of 3.8 nm calculated via the Scherrer equation. The 13 % yttrium weight substitution does not alter the crystalline phase but affects vibrational states (Additional file 1: Figure S3).
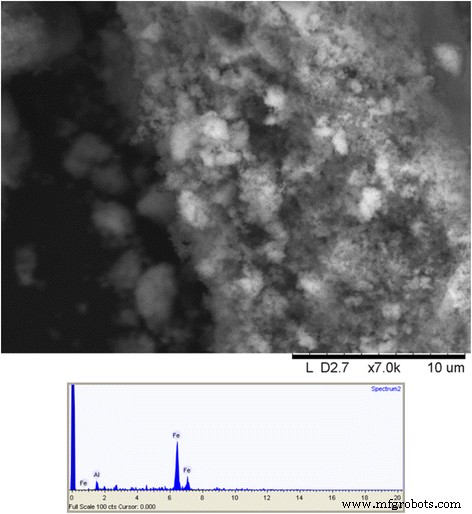
Scanning electron image of γ‑Fe₂O₃ synthesized through yttrium‑directed sol‑gel synthesis and an EDS spectrum of the same material.
Discussion
Our study provides the first evidence that γ‑Fe₂O₃ nanoparticles act as functional nanozymes in planta. Compared to chelated iron, IONs deliver iron more efficiently and simultaneously scavenge reactive oxygen species, as demonstrated by lower H₂O₂ and MDA levels. The reduction in oxidative stress correlates with improved drought tolerance and accelerated leaf growth.
The observed enzymatic effects align with known peroxidase‑ and catalase‑like activities of iron‑oxide nanoparticles at low and neutral pH, respectively. While alternative mechanisms—such as direct interaction with proteins or lipids—cannot be excluded, the combination of increased leaf iron content, altered magnetization, and decreased ROS supports a nanozyme‑mediated pathway.
Iron(III) ions alone did not enhance plant performance; in fact, they reduced leaf iron uptake, likely reflecting a plant‑initiated defense against excess iron. The yttrium dopant stabilizes the maghemite phase and suppresses transformation to hematite, thereby preserving enzymatic activity.
Enhanced chlorophyll accumulation, faster leaf elongation, and greater biomass suggest practical agronomic benefits, particularly in environments where weed competition and water scarcity are limiting factors. Future work should evaluate yield and seed quality to fully establish the commercial potential of ION fertilization.
Conclusions
γ‑Fe₂O₃ nanoparticles function as effective nanozymes in Brassica napus, reducing leaf hydrogen peroxide and lipid peroxidation while boosting growth and drought resilience. These findings support their deployment as a dual‑purpose fertilizer and stress mitigator in sustainable agriculture.
Methods
Experimental Conditions and Design
Brassica napus seeds (spring rape variety Larissa) were surface‑sterilized, germinated on agar, and transplanted into 8 × 8 cm pots containing sterilized S‑Soil. After 7 days of establishment, plants received daily irrigation with either a standard nutrient solution, the same solution plus 3.4 mg L⁻¹ chelated Fe, or the solution supplemented with 0.5, 0.8, 1, or 2 mg mL⁻¹ γ‑Fe₂O₃ (total 100–400 mg plant⁻¹). Plants were grown in a controlled chamber (16 h light, 180 µE m⁻² s⁻¹; 8 h dark; 25 °C/22 °C; 65 % RH). After 5 days of treatment, an additional 5 days of standard nutrient irrigation were followed by 4 days of drought. Hydrogen peroxide and MDA assays were performed after drought, then plants were rewatered for 3 days to assess recovery. The entire experiment was replicated four times.
Nanoparticle Synthesis and Characterization
Yttrium‑doped γ‑Fe₂O₃ nanoparticles were synthesized per Cui et al. (2013) with ~13 % wt Y. Characterization included XRD, SEM, NTA, FTIR, TGA, and AFM. SEM (Hitachi TM1000) and EDS confirmed the iron‑oxide composition. NTA (NanoSight 300) yielded hydrodynamic size distributions; FTIR (Perkin‑Elmer Spectrum 100) and TGA (Perkin‑Elmer Pyris 1) provided additional compositional data. AFM (Bruker FastScan) visualized particle morphology. XRD (Bruker Smart ApexII) confirmed maghemite crystallinity, with a 3.8 nm crystallite size calculated from the 16.197° 2θ peak.
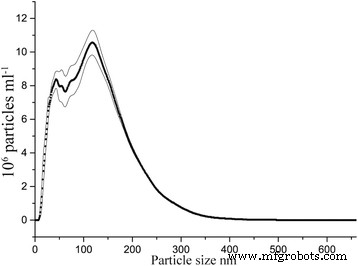
Hydrodynamic particle size distribution in water, as measured by NTA, of γ‑Fe₂O₃ synthesized through yttrium‑directed sol‑gel synthesis. Values are averaged from four repeated measurements, and the area within the thin lines represents mean error.

Maghemite nanoparticles synthesized through yttrium‑directed sol‑gel, dispersed onto silicon wafer and imaged with AFM. The same image is represented in 3D and 2D.
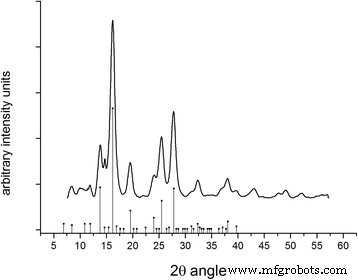
A powder diffractogram of the produced maghemite nanoparticles. The peaks align with the positions of standard maghemite from the database seen as point with drop line at the base of the figure. Crystallite size was calculated from the largest peak at 16.197 2θ degrees angle.
Plant Trait Measurements
Leaf length, chlorophyll content (SPAD), and above‑ground biomass were recorded before treatment, after 5 days of irrigation, and after drought. Measurements were averaged across 16 biological replicates. Biomass was oven‑drying at 110 °C for 72 h.
Iron Content and Magnetic Measurements
After drought, plant tissues were ashed at 450 °C, digested in 36 % HCl, diluted, and analyzed by ICP‑AES at 238.204 nm. Magnetic moments were measured on a SQUID magnetometer at 2 K, sweeping from 10,000 Oe to 0 Oe. Data were normalized to sample weight.
Hydrogen Peroxide Measurements
Leaf hydrogen peroxide was quantified using the eFOX method (Mohr’s salt reaction). Leaf samples were powdered in liquid nitrogen, extracted in phosphate buffer, and reacted with ferrous ammonium sulfate, sorbitol, xylenol orange, ethanol, and sulfuric acid. Absorbance differences between 550 nm and 800 nm were used to calculate H₂O₂ concentrations (R² = 0.9946).
Lipid Peroxidation
MDA was measured following the method of [40]. Samples were homogenized in 0.1 % TCA, and absorbance at 532 nm was corrected for turbidity at 600 nm. An extinction coefficient of 155 mM cm⁻¹ was used to calculate MDA.
Statistical Analysis
All data were analyzed with Minitab 17 using one‑way ANOVA and Fisher’s LSD for post‑hoc grouping. Student’s t‑tests identified specific significant differences between treatments.
Nanomaterials
- Semiconducting Nanoparticles: Quantum Confinement and Advanced Synthesis Techniques
- Nanoparticle-Based Cancer Therapy: Advances, Mechanisms, and Clinical Translation
- Cobalt‑Doped FeMn₂O₄ Spinel Nanoparticles: Size‑Controlled Synthesis and Magnetic Behavior
- Hydrogen‑Assisted Atomic Rearrangement in GaN‑Based Multiple Quantum Wells: Enhancing Structural Uniformity and Optical Efficiency at 750 °C
- Gold Nanoparticle–Modified 6‑Mercaptopurine and Rabies‑Derived Peptide Enhance SH‑SY5Y Neural Cell Proliferation and Neurite Outgrowth
- How Elastic Stiffness and Surface Adhesion Govern Nanoparticle Bouncing Behavior
- Impact of Gold Nanoparticle Size and Concentration on Root Development in Arabidopsis thaliana
- Al₂O₃ and SiO₂ Nanoparticles with Ultrasound Significantly Reduce Water Supercooling
- Comprehensive Review of Luminescent Silica Nanoparticles: Classification, Synthesis, and Practical Applications
- Bibliometric Trends in Drug Delivery and Magnetic Nanoparticles (1980‑2017)



