Biocompatible 5‑Aminolevulinic Acid / Gold Nanoparticle‑Loaded Ethosomal Vesicles: A Synergistic Photodynamic/Photothermal Transdermal Strategy for Hypertrophic Scar Treatment
Abstract
We report a biocompatible ethosomal vesicle (A/A‑ES) co‑loaded with 5‑aminolevulinic acid (ALA) and gold nanoparticles (AuNPs) prepared by ultrasonication without toxic reagents. A/A‑ES exhibits a pronounced plasmonic absorption band at 600–650 nm, enabling simultaneous photodynamic (PDT) and photothermal (PTT) activation using a 632 nm laser. In vitro transdermal studies confirm that A/A‑ES delivers both ALA and AuNPs efficiently into hypertrophic scar (HS) tissue, achieving 48 % ALA retention after 2 h. Cellular assays with hypertrophic scar fibroblasts (HSF) demonstrate that A/A‑ES markedly elevates reactive oxygen species (ROS) production and heat generation, leading to enhanced apoptosis and necrosis compared to PDT or PTT alone. These results highlight A/A‑ES as a promising topical platform for synergistic PDT/PTT of HS.
Background
Hypertrophic scars (HS) arise from excessive dermal collagen deposition and are characterized by thickened, fibrotic skin that can impair function and aesthetics. Conventional treatments—including intralesional injections, pressure garments, and laser therapy—often suffer from limited efficacy, discomfort, or adverse effects such as hypopigmentation and tissue atrophy. Photodynamic therapy (PDT) using 5‑aminolevulinic acid (ALA) offers selective cytotoxicity via ROS generation, yet its clinical utility is hampered by poor skin penetration and low ROS yield. Photothermal therapy (PTT), particularly with gold nanoparticles (AuNPs) that convert light to heat through localized surface plasmon resonance (LSPR), can enhance ROS production and selectively ablate scar tissue, but high laser powers risk damage to surrounding skin.
Combining PDT and PTT in a single platform promises synergistic benefits: ALA‑mediated ROS generation can be amplified by the heat of AuNPs, while the photothermal effect can increase local temperature, further boosting ROS yields. Ethosomal vesicles (ES) provide a deformable, lipid‑based carrier that enhances transdermal delivery of hydrophilic and lipophilic drugs alike. Prior work demonstrated that ALA‑loaded ES (ALA‑ES) significantly outperformed aqueous solutions in delivering ALA to HS tissue. However, integrating AuNPs into ES while maintaining biocompatibility and efficient delivery remains challenging. Here, we present a facile, ultrasonication‑based synthesis of A/A‑ES that simultaneously encapsulates ALA and AuNPs without toxic solvents, achieving high ALA entrapment (≈20 %) and strong 600–650 nm absorption.
Results and Discussions
The Characterization of A/A‑ES
Ultrasonication served two purposes: (1) it generated AuNPs in situ via radical‑mediated reduction of AuCl₄⁻, obviating toxic reducing agents; (2) it induced lipid bilayer reorganization, producing small, uniformly sized vesicles with ample internal core for drug loading. UV‑Vis spectroscopy confirmed a strong absorption peak at 600–650 nm (Fig. 1a), attributable to plasmonic coupling between adjacent AuNPs. Dynamic light scattering revealed a narrow size distribution centered at 166 ± 83 nm (Fig. 1b), while SEM/TEM images showed intact, spherical vesicles (~200 nm) with clearly visible AuNPs embedded in the bilayer (Fig. 1c,d). Entrapment efficiency (EE) of ALA reached 20 %, surpassing values reported for conventional ES formulations (<10 %)【27】. These characteristics suggest A/A‑ES is well‑suited for traversing the dense collagen matrix of HS.
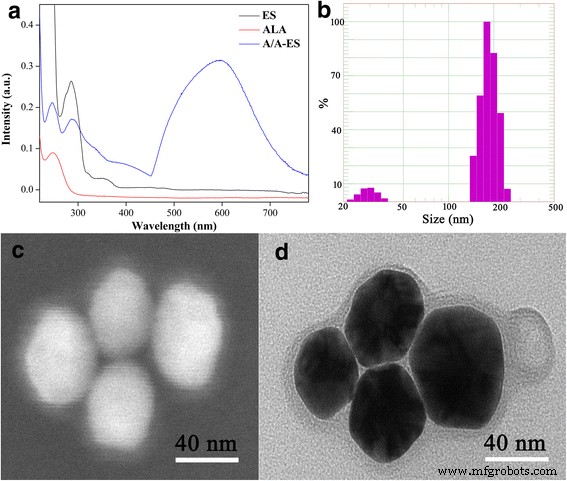
a UV‑Vis spectra of ES, ALA, and A/A‑ES. b Size distribution of A/A‑ES. c, d SEM and TEM images of A/A‑ES
In Vitro Transdermal Penetrability Study of A/A‑ES
Using Franz diffusion cells, we quantified the retention of ALA and AuNPs in freshly excised HS tissue. Both agents peaked at 2 h, reaching 48 % of the applied ALA dose within the tissue (Fig. 2a). The concurrent decline thereafter reflects complete transdermal permeation. TEM imaging (Fig. 2b–d) confirmed intact vesicles in the dermis, followed by release of AuNPs that aggregated in situ—favorable for heat generation upon laser irradiation. These data establish A/A‑ES as an effective carrier for co‑delivery of ALA and AuNPs into HS tissue.
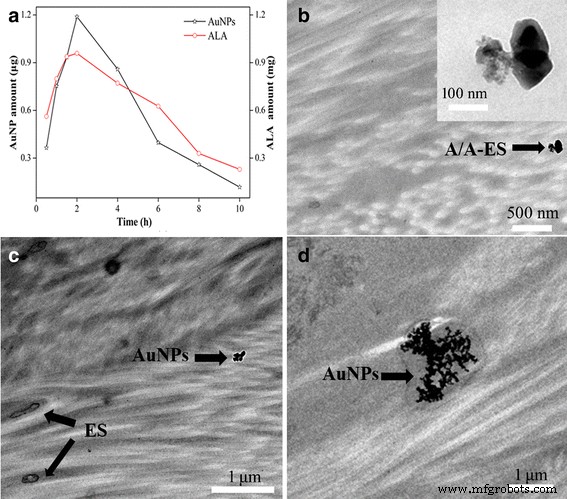
a Retention of ALA and AuNPs. b Distribution of A/A‑ES in HS tissue. c Distribution of ES and AuNPs. d AuNP accumulation in HS tissue
In Vitro PDT/PTT of HSF
Biocompatibility Assay
Cell viability assays revealed no significant cytotoxicity for A/A‑ES at concentrations ≤2.0 mM, with survival >90 % after 12 h (Fig. 3). Higher doses caused only modest reductions, confirming the formulation’s safety profile and guiding the therapeutic concentration to 2.0 mM (≈14 % v/v) for subsequent experiments.
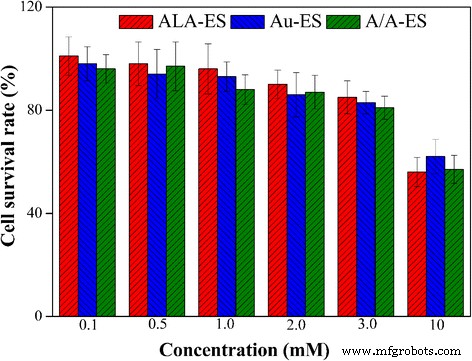
Cell viability of HSF after 12 h exposure to ALA‑ES, Au‑ES, and A/A‑ES in the dark.
PDT/PTT for HSF
Confocal microscopy showed robust protoporphyrin IX (PpIX) accumulation in HSF treated with ALA‑ES and A/A‑ES; Au‑ES produced negligible PpIX (Fig. 4). Upon 632 nm laser irradiation, PpIX fluorescence decreased while ROS signals intensified, especially in A/A‑ES–treated cells—evidence of enhanced ROS generation via AuNP LSPR. Morphological changes, such as membrane blebbing, were pronounced in A/A‑ES–treated cells, indicating cell death. Infrared thermography confirmed that ALA‑ES, Au‑ES, and A/A‑ES elevated local temperature to 41.3 °C (Au‑ES/A/A‑ES) and 36.5 °C (ALA‑ES) under irradiation, with rapid cooling thereafter (Fig. 5). These findings underscore the dual photodynamic and photothermal actions of A/A‑ES.
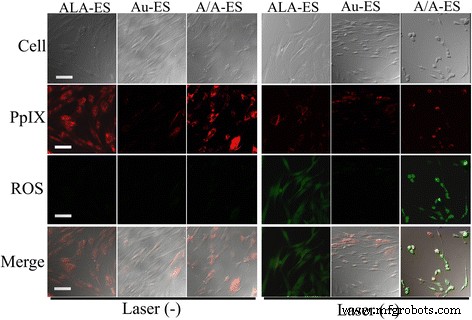
Confocal images of HSF treated with ALA‑ES, Au‑ES, and A/A‑ES before and after 632 nm laser irradiation.
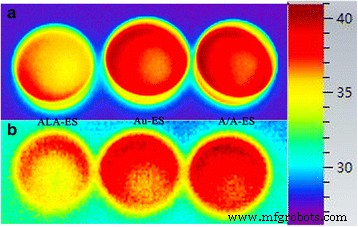
Infrared imaging of HSF treated with ALA‑ES, Au‑ES, and A/A‑ES during and after irradiation.
Apoptosis and Necrosis Assay
Flow cytometry of Annexin V/PI‑stained cells revealed that laser‑treated A/A‑ES induced the highest necrotic fraction (61.8 %) compared with ALA‑ES (47.7 %) and Au‑ES (24.3 %) (Fig. 6). Early and late apoptosis were also elevated in A/A‑ES, reflecting the synergistic effect of ROS amplification and photothermal stress. These results quantitatively demonstrate the superiority of the combined PDT/PTT approach over single modalities.
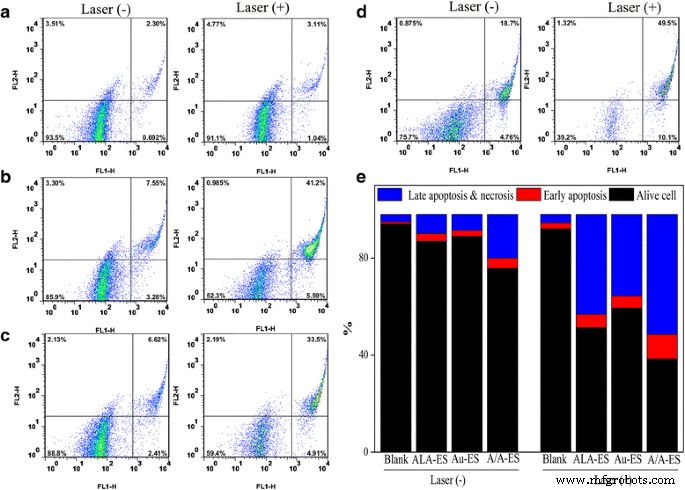
Apoptosis assay of HSF treated with ALA‑ES, Au‑ES, and A/A‑ES with/without laser. Statistical analysis of live, early apoptotic, late apoptotic, and necrotic cells.
Visualization of A/A‑ES in HSF
Transmission electron microscopy revealed extensive AuNP aggregation within the cytoplasm of A/A‑ES–treated HSF, consistent with efficient vesicle fusion and cargo release. Post‑irradiation images showed characteristic membrane blebs, swollen mitochondria, and ruptured outer membranes—hallmarks of cell death (Fig. 7). These ultrastructural changes confirm that A/A‑ES effectively delivers ALA and AuNPs to HSF, leading to synergistic photodynamic and photothermal cell killing.

Light microscopy of HSF before and after irradiation (a,b). TEM images before and after irradiation (c,d).
Conclusions
We have developed a biocompatible, ultrasonically synthesized 5‑ALA/AuNP‑loaded ethosomal vesicle (A/A‑ES) that demonstrates strong 600–650 nm absorption, high ALA entrapment (~20 %), and efficient transdermal delivery to HS tissue. In vitro studies confirm that A/A‑ES enhances ROS production and photothermal heating, yielding superior apoptosis and necrosis of HSF compared with either PDT or PTT alone. These findings support A/A‑ES as a promising topical platform for synergistic photodynamic/photothermal therapy of hypertrophic scars, warranting future in vivo evaluation.
Experiments and Methods
The Preparation of A/A‑ES
Phosphatidylcholine (180 mg) was dissolved in 1.8 mL ethanol, 0.6 mL 10 mM HAuCl₄, and 3.6 mL 0.01 M ALA‑citrate buffer (pH 4.0, 12 mg ALA). The mixture was stirred at 700 rpm for 10 min, then sonicated at 200 W for 30 min until a wine‑red color appeared. Centrifugation (8000 rpm, 20 min) removed unreacted reagents, and the pellet was redispersed in 3 mL 2 mg/mL ALA hydroalcoholic solution. A transmembrane pH‑gradient loading method was applied to entrap ALA within the vesicle core, yielding the final A/A‑ES. For comparison, ALA‑ES and Au‑ES were prepared with identical ALA or AuNP concentrations but lacking the other component.
The Characterization of A/A‑ES
Negative staining with 1.5 wt % phosphotungstic acid followed by TEM (JEOL, 120 kV) assessed morphology. SEM (JEOL, 10 kV) evaluated surface features. Size distribution was measured by DLS (Nicomp 380ZLS). ALA content was quantified via fluorescamine derivatization, and EE was calculated by ultrafiltration. UV‑Vis spectra were recorded on a Varian Cary 50 spectrophotometer.
In Vitro Penetrability Study by Franz Diffusion Cells
HS tissue (≤24 h post‑excision) was mounted on a Franz cell with stratum corneum facing the donor compartment. One milliliter of A/A‑ES was applied, and the receptor was maintained at 37 °C. After defined intervals, tissue was washed, homogenized, and ALA was extracted by dialysis for 24 h. AuNP retention was quantified by ICP‑MS. After 2 h exposure, tissues were processed for ultrathin sectioning and TEM imaging.
In Vitro PDT/PTT for HSF
Cell Culture
HSF were isolated from patient HS biopsies using collagenase I digestion and cultured in DMEM supplemented with 10 % FBS at 37 °C, 5 % CO₂. Cells were used at passages 2–3.
Biocompatibility Assay
HSF (2 × 10³ cells/well) were exposed to serial dilutions of ALA‑ES, Au‑ES, or A/A‑ES in FBS‑free medium for 12 h. Cell viability was assessed with CCK‑8.
PDT/PTT Procedure
HSF (4 × 10⁴ cells/well) were incubated with 14 % v/v A/A‑ES or controls for 6 h, then washed and irradiated with a 632 nm, 40 mW/cm² He‑Ne laser for 20 min. Post‑irradiation, cells were cultured in DMEM with 10 % FBS for 24 h before downstream assays.
Intracellular PpIX and ROS Generation Assay
PpIX and ROS were visualized by CLSM (Leica TCS SP5). DCFH‑DA staining quantified ROS, while PpIX fluorescence was captured at 405 nm excitation/635 nm emission.
Apoptosis and Necrosis Assay
Annexin V‑FITC/PI double staining followed by BD FACSCalibur flow cytometry determined apoptosis and necrosis rates. Data were analyzed with FlowJo 7.6.
Statistical Analysis
Data are presented as mean ± SD. Student’s t‑test determined significance (P < 0.05).
Abbreviations
- A/A‑ES:
5‑Aminolevulinic acid/Au nanoparticle‑loaded ethosomal vesicle
- ALA:
5‑Aminolevulinic acid
- ALA‑ES:
ALA‑loaded ES
- ALA‑PDT:
ALA‑based PDT
- Au‑ES:
AuNP‑loaded ES
- AuNPs:
Au nanoparticles
- CLSM:
Confocal laser scanning microscopy
- DLS:
Dynamic light scattering
- DMEM:
Dulbecco’s Modified Eagle Medium
- EE:
Entrapment efficiency
- ES:
Ethosomal vesicles
- FBS:
Fetal bovine serum
- HS:
Hypertrophic scar
- HSF:
Hypertrophic scar fibroblasts
- ICP‑MS:
Inductively coupled plasma‑mass spectrometry
- LSPR:
Localized surface plasmon resonance
- PDT:
Photodynamic therapy
- PDT/PTT:
Photodynamic/photothermal therapy
- PpIX:
Protoporphyrin IX
- PTT:
Photothermal therapy
- ROS:
Reactive oxygen species
- SEM:
Scanning electron microscopy
- TEM:
Transmission electron microscope
Nanomaterials
- Bovine Serum Albumin–Coated Gold Nanorods Enable Low‑Dose NIR‑II Photothermal Therapy
- Polypyrrole‑Coated FePt Nanoparticles: A Multifunctional Platform for Photothermal Therapy and Photoacoustic Imaging
- High‑Loading 5‑Aminolevulinic Acid–Squalene Nanoparticles: Enhanced Tumor Photodetection and Therapy in Cell Models
- Antibody‑Functionalized Silica‑Coated Gold Nanorods for Rapid Diagnosis and Photothermal Treatment of Cryptococcus neoformans
- 5‑Minute Microwave‑Assisted Synthesis of Polydopamine‑Passivated Carbon Dots with 5 % Quantum Yield and 35 % Photothermal Efficiency for Cancer Therapy
- Carbon Quantum Dot–Platinum Porphyrin Composite: A Dual‑Function Theranostic Agent for Enhanced Photodynamic Cancer Therapy
- Targeted MoSe2 Nanodots in Albumin Nanospheres: A Biocompatible Dual‑Modality Agent for Synergistic Photothermal Radiotherapy
- Her2‑Functionalized Gold‑Nanoshelled Magnetic Hybrid Nanoparticles: Dual‑Modal US/MR Imaging and Targeted Photothermal Therapy for Breast Cancer
- Ultrasound-Enhanced Multifunctional Nanoparticles: A Novel Platform for Imaging and Synergistic Therapy of Metastatic Breast Cancer
- CoFe₂O₄ Quantum Dots: Dual Photothermal/Photodynamic Therapy for NSCLC—Inducing Apoptosis via PI3K/AKT Modulation



