Decoding GaN Growth on Exfoliated Graphite: O‑Plasma Defects and Al‑Enhanced Nucleation
Abstract
This study elucidates the nucleation and growth of GaN epitaxial layers on mechanically exfoliated graphite using classic nucleation theory. Oxygen‑plasma treatment introduces surface defects, thereby increasing nucleation density. Incorporation of elemental Al markedly boosts the nucleation rate, leading to dense nucleation layers that promote lateral GaN growth. Field‑emission SEM reveals a 3D‑to‑2D growth evolution, while HR‑TEM and FFT analyses differentiate cubic (zinc‑blend) GaN grains from conventional nucleation layers and hexagonal (wurtzite) GaN films formed with AlGaN nucleation layers. These insights enable the use of highly oriented pyrolytic graphite as a substrate for transferable optoelectronic devices.
Background
Over the past two decades, GaN has risen to the second most critical semiconductor after silicon, owing to its superior optical and electrical properties. It underpins LEDs, lasers, high‑power, and high‑frequency devices [1,2,3,4,5]. Metal‑organic chemical vapor deposition (MOCVD) dominates large‑scale GaN film growth [6,7]. Because large‑scale native substrates are scarce, GaN is typically grown heteroepitaxially on c‑sapphire, SiC, or Si. Lattice and thermal mismatch with these substrates generates extensive threading dislocations, degrading device performance [8,9,10].
Graphite’s layered structure—strong in‑plane σ bonds and weak out‑of‑plane π bonds—offers a van der Waals interface that relaxes lattice‑matching constraints. Mechanically exfoliated graphite, in particular, presents a pristine 2D surface ideal for GaN growth. While many reports describe GaN on CVD‑grown or SiC‑derived graphene, these layers contain step edges and defects that act as nucleation sites [12–15]. Highly oriented pyrolytic graphite (HOPG) yields multilayer graphite with superior crystal quality and photo‑electrical properties, and can be easily detached from epitaxial films, facilitating transferable device fabrication. Few studies have addressed the growth mechanism on pristine 2D graphite; this work investigates the influence of O‑plasma treatment and Al addition on GaN growth on multilayer graphite, guided by classic nucleation theory (CNT).
Methods/Experimental
Preparation of Graphite
Graphite was peeled from HOPG using Scotch tape. The peeled flakes were affixed to a photoresist‑coated glass plate, baked at 80 °C for 3 min, and then repeatedly peeled ten times in the same direction. The final tape, bearing the thinnest graphite layer, was pressed onto a SiO₂ substrate and the tape was slowly removed after 10 min, leaving a 10–20 nm thick graphite film. The film was then exposed to O‑plasma (40 s, 100 mW) to introduce surface defects.
Conventional Two‑Step Growth (Nucleation at 550 °C, Growth at 1075 °C)
Prior to deposition, samples were cleaned in H₂ at 1100 °C for 6 min. GaN nucleation layers were grown at 550 °C for 100 s using trimethylgallium (TMGa) and NH₃ fluxes of 35.7 µmol min⁻¹ and 26,800 µmol min⁻¹, respectively, at 600 mbar. The nucleation layers were annealed at 1090 °C for 2 min, followed by GaN film deposition at 1075 °C for 600 s.
Modified Two‑Step Growth (Nucleation at 1000 °C, Growth at 1075 °C)
After the same cleaning step, AlGaN nucleation layers were formed at 1000 °C for 100 s by introducing NH₃, TMGa, and trimethylaluminum (TMAl) with fluxes of 26,800, 22.4, and 13.3 µmol min⁻¹, respectively, at 100 mbar. The layers were annealed at 1090 °C for 2 min and GaN films were grown at 1075 °C for 600 s. Low pressure during AlGaN growth minimizes TMAl–NH₃ pre‑reactions.
Field‑emission SEM (JSM‑6700F), Raman spectroscopy (Renishaw Invia, 514‑nm laser), cross‑sectional TEM (FIB‑milled, LYRA 3 XMH, TESCAN), HR‑TEM (JEM‑2010), and AFM (SPA‑300HV) characterized surface morphology, defects, and roughness before and after O‑plasma treatment.
Results and Discussion
Defects, atomic steps, and impurities typically serve as nucleation sites by lowering the activation energy for film‑substrate bonding. Pristine graphite lacks dangling bonds, making nucleation challenging. O‑plasma treatment increases surface defects by forming oxygen functional groups, thereby enhancing GaN nucleation.
Raman spectra (Fig. 1a) show G (1582 cm⁻¹) and 2D (2727 cm⁻¹) peaks; I_G/I_2D = 2.2 indicates multilayer graphite. Post‑plasma spectra exhibit a pronounced D peak, confirming increased defects [20]. AFM (Fig. 1b,c) reveals RMS roughness rising from 0.28 nm to 0.39 nm after treatment, reflecting defect density. SEM (Fig. 1d,e) shows that untreated graphite yields sparse nuclei only at wrinkles, while plasma‑treated graphite produces a higher island density with average sizes >200 nm—larger than those on sapphire, likely due to the low migration barrier of group‑III metals on graphite [6,21].
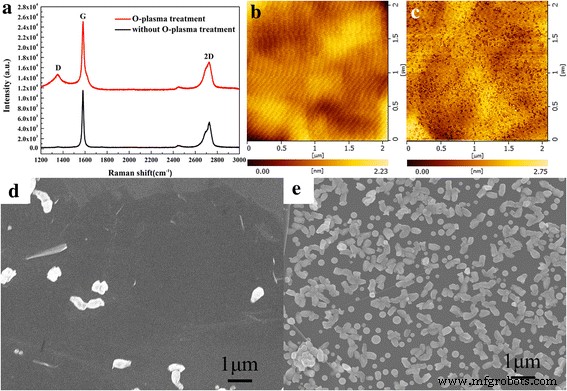
a Raman spectra of untreated (black) and treated (red) graphite. b, c 2 × 2 µm² AFM images of untreated and treated graphite, respectively. d, e FE‑SEM images of nucleation islands on untreated and treated graphite, respectively
Annealed islands (Fig. 2a) and GaN grains at growth termination (Fig. 2b) indicate that conventional two‑step growth yields sparse, small islands. Interrupted‑annealing experiments confirm that island density remains unchanged while size decreases post‑annealing, suggesting decomposition without re‑crystallization due to insufficient nucleation density. Ostwald ripening explains why only islands surpassing a critical radius grow during the high‑temperature growth step.
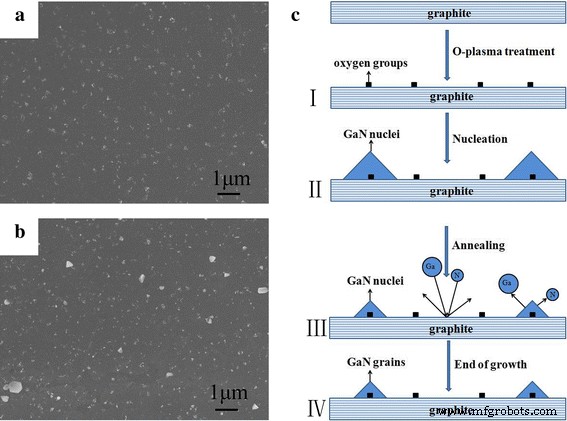
a FE‑SEM of annealed islands. b Surface morphology of GaN grains at growth end. c Proposed schematic of GaN growth mechanism on graphite
HR‑TEM (Fig. 3a–c) reveals 20 nm thick graphite and 20 nm GaN grains. FFT patterns show graphite (002) planes and cubic GaN grains, confirming that nucleation islands decompose without re‑crystallization—contrary to earlier reports that observed mixed cubic/hexagonal phases [23].

a, b TEM and HR‑TEM cross‑section micrographs along c‑GaN [110] showing the GaN/graphite interface. c–e FFT patterns for graphite, GaN grains, and their interface, respectively
High‑temperature nucleation at 1000 °C alone fails to produce GaN on graphite (Fig. 4), because elevated temperatures reduce the nucleation rate and sticking coefficient per CNT [24]. To counteract this, Al—known to have higher adsorption energy (1.7 eV) and lower migration barrier (0.03 eV) than Ga—was introduced via AlGaN nucleation layers. Al atoms remain on graphite and facilitate dense nucleation, enhancing re‑crystallization during annealing.

FE‑SEM images of nucleation layers grown at 1000 °C
AlGaN‑based growth (Fig. 5) produces dense nucleation layers (Fig. 5a), leading to larger islands after annealing (Fig. 5b). These islands coalesce during subsequent growth, yielding quasi‑2D GaN films (Fig. 5c). HR‑TEM (Fig. 6) confirms hexagonal wurtzite GaN aligned along graphite (0002) planes, indicating that Al incorporation promotes hexagonal phase nucleation and subsequent film growth.
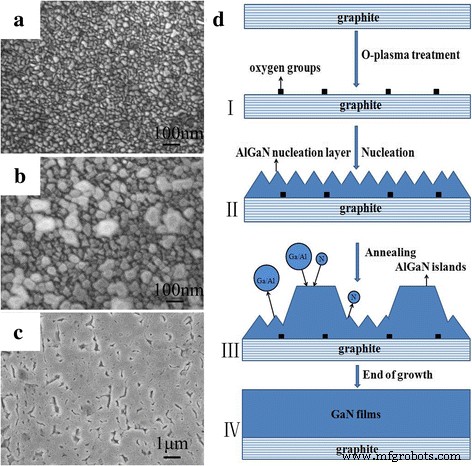
a–c FE‑SEM images of nucleation layers, annealed layers, and epitaxial layers, respectively. d Schematic of GaN growth mechanism using AlGaN nucleation layers

a, b TEM and HR‑TEM cross‑section micrographs along h‑GaN [010] showing the GaN/graphite interface. c–e FFT patterns for graphite, GaN films, and their interface, respectively
Conclusions
O‑plasma treatment introduces surface defects that lower the activation energy for GaN nucleation, increasing nucleation density on pristine graphite. Adding Al elevates the adsorption energy and reduces the migration barrier, yielding dense nucleation layers that promote lateral GaN growth. These findings accelerate the fabrication of optoelectronic devices on high‑purity graphite substrates.
Abbreviations
- 2D:
two‑dimensional
- 3D:
three‑dimensional
- AFM:
Atomic force microscopy
- CNT:
Classic nucleation theory
- CVD:
Chemical vapor deposition
- FE‑SEM:
Field‑emission scanning electron microscopy
- FFT:
Fast Fourier transform
- HOPG:
Highly oriented pyrolytic graphite
- HR‑TEM:
High‑resolution transmission electron microscopy
- MOCVD:
Metal‑organic chemical vapor deposition
Nanomaterials
- Mastering UART: The Universal Asynchronous Receiver/Transmitter Explained
- The Rapid Rise of Plant-Based Milk: Market Growth & Consumer Trends
- How Predictive Maintenance Drives Efficiency and Cuts Downtime
- How a Maintenance Storeroom Drives Reliability and Efficiency
- The 5‑Layer IoT Technology Stack: A Product Manager’s Blueprint
- Infrared Reflectance Analysis of Si-Doped Epitaxial GaN Layers Grown on Sapphire Substrates
- Optimizing Silicon‑Doped Quantum Barriers Boosts Brightness and Efficiency of Eight‑Period InGaN/GaN Blue LEDs
- Optimizing Buffer Layers via Atomic Layer Deposition for High‑Performance Vertically Aligned Carbon Nanotube Arrays
- Milling Machines 101: A Comprehensive Guide to Their Function, Components, and Uses
- Quick Return Mechanism Explained: Function, Applications, and Benefits



