Rare‑Earth Fluoride Nanoparticles Trigger Tumor Cell Proliferation through Electrical Dipole‑Mediated Integrin Activation
Abstract
Mechanical and electrical cues at the cell–extracellular matrix interface are increasingly recognized as drivers of oncogenic signaling. In this study, we demonstrate that nanometer‑scale lanthanum fluoride (LaF3) and praseodymium fluoride (PrF3) particles, dispersed in DMEM+FBS, stimulate proliferation of three human carcinoma lines (A549, SW837, MCF‑7). Size analysis (DLS, AFM, TEM) revealed a dominant sub‑10 nm population. Cell viability assays, combined with Western blotting for AKT and ERK phosphorylation, confirm activation of canonical growth pathways. A theoretical dipole‑interaction model shows that, for the required piconewton force range, only nanoparticles below ~10 nm can exert sufficient electrostatic traction on ligand‑binding sites of integrins and EGFR. These findings illuminate a distinct mechanotransduction route by which rare‑earth fluorides can influence tumor biology.
Background
Tumorigenesis arises from a complex interplay between genetic mutations and biomechanical cues from the extracellular matrix (ECM). Integrin‑mediated adhesion and receptor tyrosine kinases such as EGFR, VEGFR, and NGFR translate mechanical stress into intracellular signaling cascades that can remodel the cytoskeleton and activate transcriptional programs. The magnitude of the mechanical force required to trigger conformational changes in these receptors is on the order of piconewtons (pN), well below nanonewton scales, suggesting that nanoscale perturbations can be biologically meaningful.
Nanoparticles (NPs) can influence cellular signaling through size‑dependent surface chemistry, charge distribution, and the ability to form protein coronas in biological media. Previous work has shown that silica, gold, and carbon nanotubes can selectively promote or inhibit tumor cell growth depending on size. However, the interaction of rare‑earth fluorides with cancer cells remains unexplored, despite their widespread use in optics, catalysis, and lighting.
Lanthanum fluoride (LaF3) and praseodymium fluoride (PrF3) are low‑toxicity, highly crystalline materials that can be engineered to sub‑10 nm dimensions. Their ionic composition suggests strong electrostatic interactions with charged residues in ligand‑binding domains of integrins and EGFR, potentially enabling mechanical activation without direct membrane penetration.
Results
Size and Structure of NPs
Dynamic light scattering (DLS) measured mean hydrodynamic radii (MHR) of 5–10 nm for both LaF3 and PrF3 in DMEM+FBS. Atomic force microscopy (AFM) and transmission electron microscopy (TEM) confirmed a narrow size distribution centered at 8–12 nm, with a minor fraction of larger aggregates (20–80 nm). X‑ray diffraction (XRD) confirmed the hexagonal crystal phase, and 2‑D FFT analysis of AFM topography revealed isotropic surface roughness, indicative of a homogeneous core‑shell structure with a protein corona.
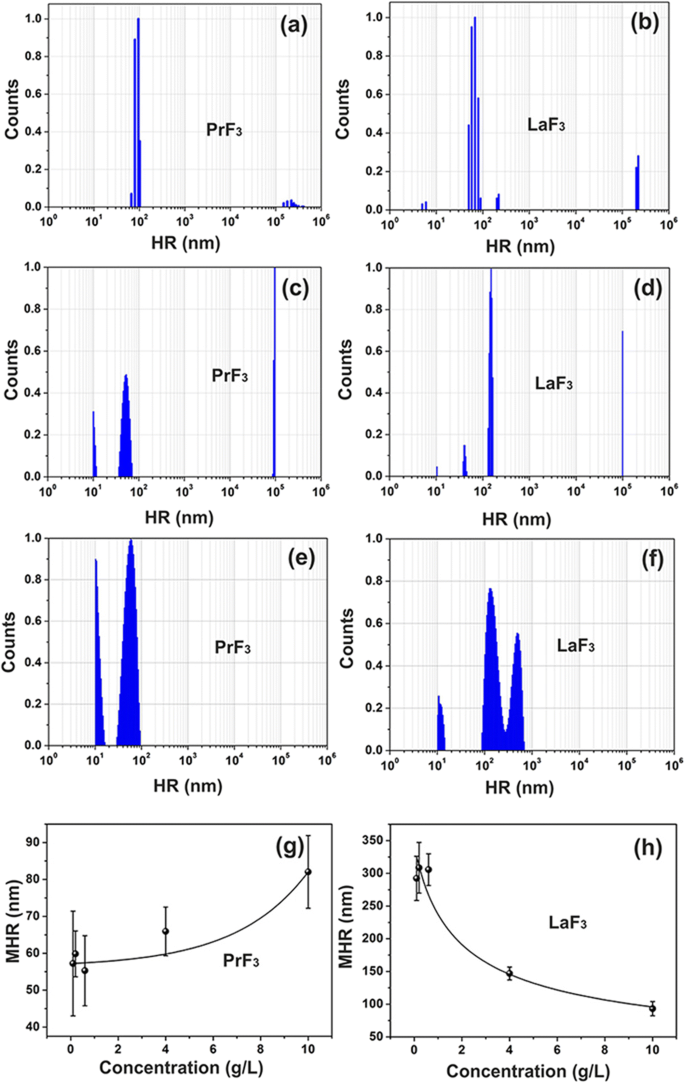
DLS size distribution spectra of RE suspensions. a, b PrF3 and LaF3 NPs (5 g/L) in water. c, d PrF3 and LaF3 NPs (5 g/L) in DMEM+FBS. e, f PrF3 and LaF3 NPs (0.1 g/L) in DMEM+FBS. g, h Mean hydrodynamic radius (MHR) with standard deviation of PrF3 and LaF3 NPs in DMEM+FBS at different concentration levels
Cell Viability
Water‑soluble tetrazolium salt (WST) assays revealed a concentration‑dependent increase in viability for all three cell lines at 5 mM NPs, with SW837 showing the greatest response (≈ 86 % increase). Lower concentrations (0.5–1 mM) produced modest, non‑significant changes. The data were statistically significant (p < 0.05) and exhibited a linear trend across the tested range.
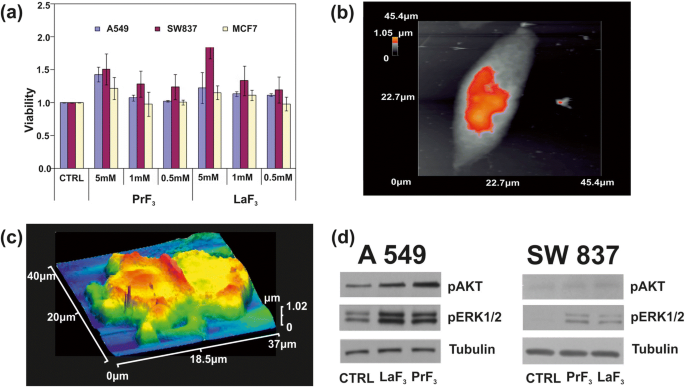
a WST viability assay histograms of three different cancer cell lines (A549, SW837, MCF7) treated with different concentrations of PrF3 and LaF3 NPs in biological media. b AFM image of a single A549 cancer cell. c AFM image of a divided A549 cancer cell in RE‑NPs in DMEM+FMS. d Wb phosphorylation analysis of the A549, SW837 cells with AKT and ERK pathways
Phosphorylation Assays
Western blotting of cells exposed to 5 mM NPs showed robust phosphorylation of ERK1/2 and AKT in A549 and SW837, whereas MCF7 exhibited negligible changes. These results align with the observed proliferation patterns and support activation of the MAPK/PI3K pathways.
Theoretical Dipole Interaction
Using a dipole‑dipole model, we estimated the force exerted by a single RE‑NP on a ligand‑binding site (LABS) as a function of particle radius and separation distance. The polar component of the force scales inversely with the square of the NP radius, explaining why only particles below ~10 nm generate forces within the 10−12–10−9 N window necessary for integrin activation. The model also predicts that the required surface charge density is attainable with the measured ionic content of La3+ and Pr3+ in the fluorides.
Discussion
The convergence of experimental and theoretical evidence points to a mechanistic pathway where sub‑10 nm LaF3 and PrF3 nanoparticles, via electrostatic dipole forces, activate integrins and EGFR, thereby triggering AKT/ERK signaling and promoting cell proliferation. The lack of cellular internalization, as confirmed by TEM, underscores that surface interactions alone are sufficient to modulate growth.
Size‑dependent effects mirror findings with other metal‑based NPs, reinforcing the principle that nanoscale mechanical cues can override chemical toxicity. The data also suggest that the protein corona formed in DMEM+FBS plays a modulatory role, possibly tuning the effective charge and distance between NP and receptor.
These insights carry implications for nanomedicine safety, particularly for rare‑earth fluorides employed in industrial and biomedical contexts. Regulatory guidelines should consider the dual role of NPs as both potential therapeutic agents and inadvertent growth promoters.
Conclusions
We demonstrate that rare‑earth fluoride nanoparticles of sub‑10 nm diameter can stimulate tumor cell proliferation by activating integrin and EGFR signaling through piconewton‑scale electrical dipole forces. The findings highlight a novel, size‑dependent mechanotransduction pathway that may inform both cancer biology research and nanomaterial safety assessment.
Methods
Synthesis of RE‑NPs
LaF3 and PrF3 nanoparticles were synthesized by co‑precipitation from La2O3/Pr2O3 and NaF in 10 % nitric acid, followed by pH adjustment to 4 and washing. Suspensions were air‑dried for structural analysis or retained in aqueous form for biological assays. Working concentrations ranged from 0.5 to 5 mM in DMEM+FBS.
Size Distribution
DLS, AFM, TEM, XRD, 2‑D FFT, and VUV spectroscopy were employed to characterize particle size, crystal structure, and surface chemistry. Data were consistent across techniques, confirming a narrow sub‑10 nm core with a protein corona.
Cell Culture and Viability
Human carcinoma lines A549, SW837, and MCF‑7 were cultured in DMEM+FBS or RPMI+FBS. WST assays measured metabolic activity after 24–48 h exposure to NPs. Western blots probed phosphorylated ERK1/2 and AKT using standard protocols.
Abbreviations
- 2D‑FFT
Two‑dimensional fast Fourier transform
- AFM
Atomic force microscopy
- AKT
Protein kinase B
- DMEM
Dulbecco’s modified Eagle’s medium
- EGFR
Epidermal growth factor receptor
- ERK
Extracellular signal‑regulated kinase
- LABS
Ligand‑binding adhesion site
- NP
Nanoparticle
- RE‑NP
Rare‑earth fluoride nanoparticle
- WST
Water‑soluble tetrazolium salt
- XRD
X‑ray diffraction
Nanomaterials
- Semiconducting Nanoparticles: Quantum Confinement and Advanced Synthesis Techniques
- Effects of 15‑nm Gold Nanoparticles on Proliferation, Apoptosis, and Spheroid Formation in HT29 Colon Carcinoma and SPEV Embryonic Kidney Cells
- Amphiphilic Hyperbranched Polyglycerol Enables Size‑Controlled, Stable Gold Nanoparticles for Hydrocarbon‑Based Nanofluids
- Gold Nanoparticle–Modified 6‑Mercaptopurine and Rabies‑Derived Peptide Enhance SH‑SY5Y Neural Cell Proliferation and Neurite Outgrowth
- Impact of Gold Nanoparticle Size and Concentration on Root Development in Arabidopsis thaliana
- Optimized Mitoxantrone Delivery Using Cholesterol‑Modified Pullulan Nanoparticles: Size‑Dependent Antitumor Efficacy Against Bladder Cancer
- Goblet Cell–Mediated Clearance of Metal Nanoparticles: An In Vivo Pathway Accelerated by Chinese‑Herb‑Induced Diarrhea
- Nanoparticle‑Induced Modulation of Cellular Mechanics: Comparative Analysis of SiO₂ and TiO₂ Effects on Caco‑2 and A549 Cells
- How Growth Directors Shape Hematite Nanorods: A Hydrothermal Synthesis Study
- Plasmon‑Enhanced Fluorescence of Rhodamine 6G at Controlled Distances from Gold Nanoparticles



