Plastic Demystified: An Expert FAQ on Definition, Materials, and Applications
As a seasoned professional in the plastics sector, I’ve often been asked, “What is plastic?” The answer isn’t as straightforward as it seems, and the best responses come from a mix of historical context, chemistry, and industry practice.
While I usually write about specific engineering materials—G10/FR4, nylon, polypropylene, PEEK, TPU, Vespel®—this FAQ breaks down the core concepts that define everything we call plastic.
1. What does the word “plastic” mean, and where does it come from?
The term originates from the Greek word plastikos, meaning “capable of being shaped or molded.” In modern usage, it broadly refers to any material that can be formed into a desired shape, whether synthetic or natural. This inclusive definition explains why plastics encompass such a diverse range of substances.
2. What are plastics made of?
Plastics derive from a wide spectrum of organic and inorganic compounds. Most are synthetic and stem from petrochemicals, though many contain natural components. “Bioplastics”—made from renewable biomass such as vegetable fats, oils, corn starch, or even bacteria—are engineered to biodegrade. For more on sustainable plastic technologies, see our recent post on this topic.
3. Are plastics produced from oil?
While oil is a common feedstock, the majority of U.S. plastics are actually produced from natural gas, especially following the rise of fracking technology. At present, plastics account for roughly 5 % of total oil consumption, making natural gas a more economical source.
4. What are the primary categories of plastic?
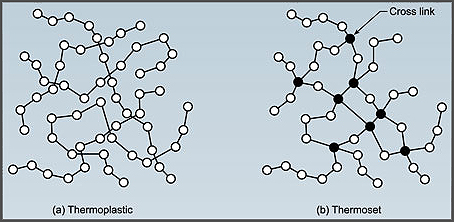
Plastics fall into two main groups: thermoplastics and thermosetting polymers. Thermoplastics retain their chemical structure when heated, allowing them to be melted and reshaped repeatedly—think PET and HDPE. Thermosetting polymers, such as epoxy and phenolic resins, undergo a permanent chemical change upon curing, so they can be molded only once. Both categories can be recycled: thermoplastics by re‑melt, thermosets by grinding into fillers.
5. What’s the difference between a monomer and a polymer?
In chemistry, a monomer is a small molecule that can link to others via polymerization, forming a larger polymer. Although many polymers are plastics, not all polymers qualify as such; natural polymers like cellulose or amber also exist.
6. How do homopolymers differ from copolymers?
Homopolymers consist of a single type of monomer—examples include polyethylene and polystyrene. Copolymers combine two or more distinct monomers and can be arranged as alternating, periodic, or block copolymers. Common copolymers such as ABS and nylon demonstrate how the composition influences properties. When selecting a material, verify whether it is a homopolymer or copolymer, as this impacts performance.
7. What are the main uses of plastic?
Plastics are ubiquitous—used in clothing, food packaging, electronics, automotive parts, medical devices, and countless other products. Their versatility stems from the ability to tailor mechanical, thermal, and chemical properties to specific applications.
Have more questions? Drop a comment below or explore our free Glossary of Plastic Manufacturing Terms for deeper insight.

Resin
- Is Plastic Manufacturing Sustainable? Innovations Reducing Its Environmental Footprint
- Understanding 10 Key Plastic Manufacturing Terms—A Practical Guide
- Plastic Manufacturing: Evolution, Current Trends, and Future Outlook
- Biodegradable Plastics: Benefits, Challenges, and the Path Forward
- 6 Key Ways Plastic Materials Transform Modern Home Construction
- How to Identify the Type of Plastic in Your Everyday Items
- Polyurethane vs. Plastic: A Technical Comparison
- Current Trends and Innovations Shaping the Plastics Industry
- How Plastic Machinery Shapes Everyday Products
- Understanding Plastic Resin: Composition, Production, and Applications



