How Digitisation Powers the Future of Medical 3D Printing
2020 was a watershed moment for healthcare. Hospitals, overwhelmed by COVID‑19, faced unprecedented shortages of ventilators, face shields, masks, and test swabs. With global supply chains strained, many turned to local 3D‑printing service bureaus to design, evaluate, and manufacture critical components in days, rather than weeks. The pandemic spotlighted 3D printing’s capacity for rapid, flexible production. Yet the medical community had already recognized this potential. Over the past decade, hospitals and device manufacturers have employed 3D printing for anatomical models, surgical instruments, implants, and prostheses, illustrating the technology’s breadth. Today, additive manufacturing (AM) in medicine is a $1 billion industry, projected to reach $6 billion by 2027. To realise this growth, firms must overcome data management hurdles, regulatory compliance, and production consistency. Below we examine each challenge and show how digitisation and MES software can resolve them.
Managing Data in Medical Additive Manufacturing
Capturing and maintaining complete, accurate data throughout the AM lifecycle is critical. Unfortunately, data collection remains largely manual, increasing the risk of errors and audit gaps.
When separate systems handle project tracking, production scheduling, and quality control, information becomes fragmented. Each system maintains its own “truth,” and reconciling them consumes valuable time.
Digitising and centralising production data eliminates these silos. A modern Manufacturing Execution System (MES) provides a unified digital workflow that captures, enriches, and shares consistent, up‑to‑date information across the enterprise.
With an MES, every part receives a unique serial number that tracks every step—post‑processing, inspection, and more. Integration with AM hardware allows real‑time monitoring of printers and collection of key parameters such as print time, temperature, and laser intensity. Analyzing this data identifies the conditions that yield the highest quality and flags failures before they impact patients.
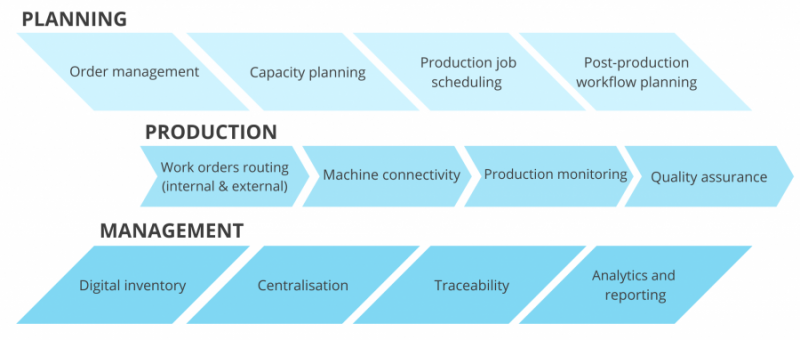
Ensuring Repeatable Production
AM technology advances rapidly, but achieving repeatability remains challenging. Material variations, scheduling errors, part orientation, and machine calibration can all affect consistency.
Medical device manufacturing demands that every step and parameter be defined, tracked, and controlled. Workflow automation within an MES enforces these controls by automatically assigning jobs to the appropriate machine and orientation based on predefined rules.
Automation eliminates manual scheduling errors, reduces variability, and supports scalability. It also enables rapid identification and replication of successful processes while correcting those that fail.
Discover more about AM automation: Where Are You in Your Additive Manufacturing Automation Journey?
Meeting Regulatory and Quality Requirements for 3D‑Printed Medical Devices
Regulators such as the FDA and EMA expect manufacturers to embed quality into every stage of AM, rather than merely inspecting at the end. Achieving this without a specialised digital system is nearly impossible.
A modern additive MES validates processes, logs data with full traceability, and enforces procedures. When auditors demand evidence that AM processes are in control, the MES delivers instant, comprehensive documentation—dramatically reducing audit time and resource demands.
The MES’s automatic data capture supports compliance with ISO 13485, 21 CFR Part 11, and 820, providing regulators confidence that quality standards remain met over time.
Enable the Digital Thread in Medical Additive Manufacturing
Beyond regulatory support, a future‑ready MES unlocks a continuous digital thread that unites all AM operations. This integration fuels data‑driven decision‑making, streamlines standardised workflows, and accelerates scalability.
Learn how AMFG can help you start collecting data, standardise, and automate processes with MES today
3D printing
- 3D Printing: A Cornerstone of Industry 4.0
- How 3D Printing Is Revolutionizing the Medical Field: 4 Key Applications & Future Trends
- How 3D Printing Is Revolutionizing Defence – Benefits, Challenges, and Future Applications
- How 3D Printing is Revolutionizing Automotive Production – 2021 Insights
- 10 Expert Predictions Shaping the Future of 3D Printing
- How 3D Printing Is Transforming Manufacturing, Healthcare, Aerospace, and Education
- Cutting 3D Printing Costs: Proven Strategies from Design to Post-Processing
- How 3D Printing Will Transform Manufacturing in the Next Decade
- How 3D Printing is Revolutionizing Modern Manufacturing
- 4D Printing: Revolutionizing Additive Manufacturing for Adaptive Solutions



