Acrylic Plastic: Properties, Production, and Future Trends
Background
Acrylic plastic is a family of synthetic polymers derived from acrylic acid. The most common member, polymethyl methacrylate (PMMA), is sold under brand names such as Plexiglas, Lucite, Perspex, and Crystallite. PMMA is a tough, highly transparent material with exceptional resistance to ultraviolet light and weathering. Its versatility—coloring, molding, cutting, drilling, and forming—makes it ideal for airplane windshields, skylights, automobile taillights, outdoor signage, and even the iconic double‑insulating panels that line the Houston Astrodome’s ceiling.
Polymers are long chains of repeating molecular units. The term originates from Greek words meaning “many parts.” When these units link, the resulting material exhibits properties far beyond those of its individual components, a phenomenon that can be likened to shuffling raw materials into a box and emerging with a functional product such as a color television set.
The first polymer, celluloid, was created in 1869 from cellulose nitrate and camphor, and found early use in photographic film, combs, and men’s shirt collars. In 1909, Leo Baekeland introduced Bakelite—phenol‑formaldehyde resin—which could be machined, molded, and colored, becoming a staple in radio, telephone, and electrical equipment as well as kitchen countertops, buttons, and knife handles.
Acrylic acid dates back to 1843, with methacrylic acid—its derivative—formulated in 1865. Reacting methacrylic acid with methyl alcohol produces methyl methacrylate, the monomer that, when polymerized by German chemists Fittig and Paul in 1877, yields PMMA. Commercial acrylic sheets first appeared in 1936, and during World War II the material was used in submarine periscopes, aircraft windshields, canopies, and gun turrets.
Raw Materials
The foundational monomer is methyl methacrylate (CH2=C(CH3)COOCH3), depicted in Figure 1. The double bond between the central carbons breaks during polymerization, linking chains into PMMA. Figure 2 illustrates other monomers—methyl acrylate and acrylonitrile—that can copolymerize with methyl methacrylate to tailor properties such as elasticity.
Methyl methacrylate can be produced by reacting acetone (CH3COCH3) with sodium cyanide (NaCN) to form acetone cyanhydrin, which then reacts with methyl alcohol (CH3OH). Copolymerization with other monomers allows precise control over the resulting plastic’s characteristics.
The Manufacturing Process
Acrylic polymers form when a monomer like methyl methacrylate reacts with a catalyst—often an organic peroxide—initiating a chain reaction that proceeds without incorporating the catalyst into the final polymer.
Products are available as flat sheets, rods and tubes, or molding powders. Molding powders are typically produced via suspension polymerization, creating grains with tightly controlled molecular weight suitable for extrusion or molding.
Sheet production employs bulk polymerization. Two primary methods exist: batch cell and continuous. Batch cell is the most common due to its simplicity and adaptability for sheets ranging from 0.06 to 6.0 inches (0.16–15 cm) in thickness and widths from 3 ft (0.9 m) up to several hundred feet. Continuous polymerization, faster and less labor‑intensive, is reserved for thinner, smaller sheets.
Below we detail both processes.
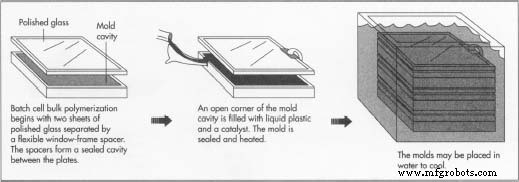 Batch cell bulk polymerization is the preferred method for creating acrylic sheets of varied thickness.
Batch cell bulk polymerization is the preferred method for creating acrylic sheets of varied thickness.
Batch Cell Bulk Polymerization
- Two polished glass plates are separated by a flexible “window‑frame” spacer that allows the mold cavity to shrink, compensating for material contraction during polymerization. In some setups, polished metal plates replace glass. Multiple plates can be stacked, with the upper surface of one becoming the bottom of the next cavity. Spring clamps secure the assembly.
- Each cavity receives a pre‑measured liquid syrup of methyl methacrylate and catalyst, occasionally supplemented with a prepolymer—short chains that accelerate the reaction. The syrup fills the cavity entirely.
- The mold is sealed, and heat is applied to activate the catalyst.
- The exothermic reaction generates heat, which is managed by air ovens or a water bath. A programmed temperature cycle ensures proper cure time while preventing monomer vaporization and bubble formation. Thinner sheets cure in 10–12 hours; thicker sheets may require several days.
- Upon curing, the mold cools, opens, and the sheets are removed. The plates are cleaned and reassembled for the next batch.
- Sheets may undergo annealing—heating to 284–302 °F (140–150 °C) for several hours—to relieve residual stresses that could cause warping.
- Excess material, or flash, is trimmed. Masking paper or plastic film is applied for protection during handling and shipping, often stamped with brand name, size, and handling instructions, and indicating compliance with safety or building code standards.
Continuous Bulk Polymerization
- The continuous process parallels batch cell but targets thinner, smaller sheets, shortening cycle times. Syrup of monomer and catalyst is introduced at one end of parallel stainless‑steel belts; belt spacing determines sheet thickness.
- Belts move the reacting mixture through sequential heating and cooling zones following a programmed temperature cycle to cure the material.
- Electric heaters or hot air anneal the material as it exits the belts.
- Sheets are cut to size and covered with masking paper or plastic film.
Quality Control
Throughout manufacturing, chemicals are stored, handled, and processed under controlled environmental conditions to prevent contamination and ensure safety. Temperature control is critical: the monomer and catalyst are pre‑conditioned, and the reacting mixture’s temperature is continuously monitored and regulated during polymerization. Periodic laboratory analyses confirm the finished acrylic’s physical, optical, and chemical properties.
Toxic Materials, Safety Considerations, and Recycling
Producing acrylic plastic involves highly toxic substances. The polymerization process can trigger explosions if not properly monitored and generates toxic fumes. Modern regulations mandate closed‑system processing and complete capture or neutralization of fumes before atmospheric release.
Acrylic is classified as a group 7 plastic and is rarely collected for recycling. Large, undamaged pieces can sometimes be reformed, but this represents only a small fraction of waste. In landfills, acrylic is non‑biodegradable and can be highly flammable, requiring careful storage away from combustion sources.
The Future
Annual consumption of acrylic plastics has risen at roughly 10% per year, with a projected growth rate of around 5% for the coming decade. Despite being one of the earliest plastics, acrylic continues to offer unrivaled optical clarity and outdoor durability, securing its position as the material of choice across diverse applications.
Manufacturing process
- Amber: From Ancient Resin to Scientific Treasure – History, Properties, and Modern Uses
- The Guillotine: History, Design, and Legacy
- Silicon: Properties, Production, and Market Outlook
- The Ukulele: From Portuguese Roots to Modern Craftsmanship
- The Complete Guide to the U.S. Chicken Industry: History, Production, and Quality Control
- Compost: The Ultimate Soil Enrichment Solution
- Gelatin: From Ancient Protein to Modern Applications – Production, Uses, and Safety
- The Evolution and Manufacturing of Modern Pin‑Tumbler Locks
- Vodka: From Ancient Distillation to Modern Production
- Premium Plastic Food Wrap: Preserve Freshness & Seal Odors



