Gold: History, Properties, Mining, and Future Applications
Background
Gold, with its unmistakable yellow hue, is one of humanity’s oldest metals. Archaeological evidence shows that Neolithic people harvested gold from riverbeds, while the earliest documented mining dates back to 3500 B.C. when Egyptians and Sumerians fashioned jewelry, religious artifacts, and goblets from the metal.
Gold’s combination of beauty and physical resilience has made it a prized resource for millennia, often sparking conflict and adventure—from the collapse of the Aztec and Inca empires to the gold rushes that reshaped America’s frontier.
The world’s largest gold reserve lies in South Africa’s Precambrian Witwatersrand Conglomerate—a vast, multi‑mile‑deep deposit that accounts for roughly two‑thirds of global production. Major producers also include Australia, the former Soviet Union, and the United States, where gold is mined in Arizona, California, Colorado, Montana, Nevada, South Dakota, and Washington.
About 65 % of processed gold goes into the arts sector, primarily jewelry. Industrial uses—electrical, electronic, and ceramic—now represent an estimated 25 % of the market. The remaining gold is fashioned into “purple of Cassius,” a ruby‑colored glass used on office building windows to reduce summer heat, and mirrors for space‑flight and electron‑microscopy that reflect the infrared spectrum.
Physical Characteristics
Gold (chemical symbol Au) is renowned for its malleability, ductility, and resistance to oxidation. Malleability allows gold to be hammered into sheets a thousand times thinner than paper; these sheets are then used in infrared‑reflective coatings, dental fillings, or plated surfaces. Ductility enables gold to be drawn into fine wire, essential for integrated circuit connections, orthodontic appliances, and even jet‑engine components.
Because gold is relatively soft, it is usually alloyed with silver, copper, platinum, or nickel. Alloys are expressed in karats; 24‑karat gold is pure, while 18‑karat gold contains 18 parts gold to 6 parts metal.
Extraction and Refining
Gold is typically found in pure form, but it can also be recovered from silver, copper, lead, and zinc. While seawater contains trace amounts of gold, the concentration is far too low to be commercially viable. The metal occurs in two main deposit types: lode (vein) and placer. Extraction methods vary accordingly. After mining, ore is pulverized and refined through one or more of four primary processes: flotation, amalgamation, cyanidation, or carbon‑in‑pulp.
Gold is generally found in two types of deposits: lode (vein) or placer deposits.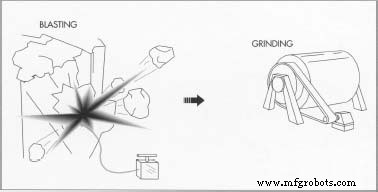
Mining
- In lode deposits, gold is embedded in quartz veins within host rock. Miners drill, blast, or shovel to extract the ore, often deep underground. The ore is then transported to a mill for refinement.
- Placer deposits contain nuggets and grains that have been transported by water and are usually mixed with sand or gravel. Hydraulic mining, dredging, and power shoveling are the principal techniques, all relying on gravity for separation. These methods are increasingly regulated or banned due to environmental impacts.
Grinding
- Once mined, the ore is washed and ground into smaller fragments, typically in a ball mill that uses steel balls to pulverize the material.
Separating the Gold from the Ore
- Flotation separates gold by adding a frothing agent and a collector that binds gold to air bubbles, which then rise to the surface for skimming.
- Cyanidation dissolves gold in a weak cyanide solution, after which zinc is added to precipitate the metal. The gold is recovered by filtration.
- Amalgamation involves passing ore over mercury‑covered plates to form a gold‑mercury amalgam; heating the amalgam releases mercury, leaving pure gold.
- Carbon‑in‑pulp uses activated carbon to adsorb gold from a cyanide‑treated pulp, then removes the carbon in a heated, caustic solution.
- Smelting heats gold with flux, which bonds contaminants and floats as slag, allowing the clean metal to solidify in molds.
Floatation, cyanidation, and the carbon‑in‑pulp method are 3 processes used to refine gold. They can be used alone or in combination with one another.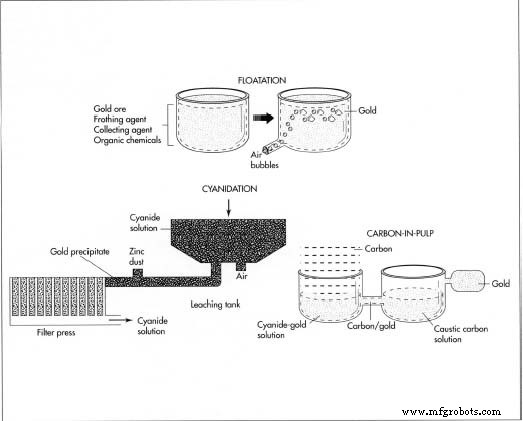
Two other methods of gold refining are amalgamation and smelting.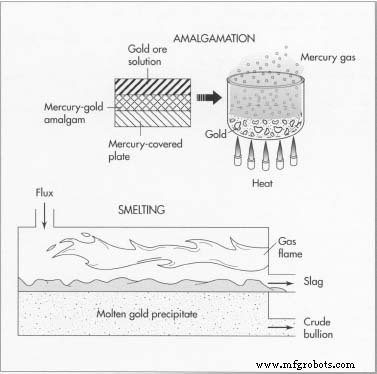
The Future
As a finite resource, gold’s long‑term supply is limited, yet its demand in jewelry and high‑tech industries—particularly electronics—remains strong. Recent advances focus on extracting gold from sulfide ores, traditionally more challenging than oxide ores. Bioleaching, which employs bacteria to break down sulfide minerals, has emerged as a cost‑effective and environmentally friendly method.
Manufacturing process
- Titanium: Key Properties, Applications, and Industrial Uses
- Castanets: History, Craftsmanship, and Performance
- The History, Production, and Future of Glue: From Ancient Adhesives to Modern Innovations
- Thread: Types, History, Production, and Quality Control
- Acetylene: Production, Uses, and Handling – A Comprehensive Overview
- Comprehensive Overview of Asbestos: From Ancient Uses to Modern Health Risks
- The History, Design, and Manufacturing of Dice: From Ancient Tools to Modern Gaming
- Plywood: History, Production, and the Path Ahead
- Tin: From Bronze Age to Modern Industry – Uses, Production, and Future
- Key Insights About Gold: What You Need to Know



